CHEM 150 FINAL
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
John Dalton
created atomic theory
atomic theory
matter consists of atoms
atoms of the same element have the same properties
atoms combine in small, whole-number ratios
atoms aren’t created or destroyed, simply rearranged (in chem rxns)
JJ Thomson
discovered the electron, determined it carried a negative electrical charge, measured its charge/mass ratio
Robert Millikan
measured charge on the electron and calculated its mass using Thomson’s ratio
Ernest Rutherford
discovered the nucleus; disproved plum pudding model in favor of nuclear model; discovered protons
James Chadwich
discovered neutron
monoatomic gas
all noble gases (group 18)
Diatomic gas
H2,N2,O2,F2,Cl2
monoatomic liquid
Hg
diatomic liquid
Br2
monoatomic solid
all other elements
Wilhelm Röntgen
discovered x-rays
Antoine Becquerel
discovered that uranium was radioactive
Dmitri Mendeleev
organized modern periodic table
RMIVUXG
wavelength spectrum from lowest frequency/energy to highest; Raging Martians Invaded Venus Using X-ray Guns
Johannes Rydberg
came up with an equation to find wavelengths a hydrogen atom emitted; Rydberg’s constant
Niels Bohr
determined electrons are in quantized energy levels; potential energy
Max Planck
energy is quantized; called a quantum of energy a photon, photons travel as waves; Father of quantum theory; E = hv h=Planck’s constant
Albert Einstein
proved Planck’s theory that energy is quantized by explaining the photoelectric effect
Louis de Broglie
proposed matter should have wave-like properties; equation for the wavelength of a piece of matter
George Thomson
proved the wave-like nature of electrons when he showed electrons exhibited diffraction
Werner Heisenberg
devised the Uncertainty Principle —> we can never know the exact position & momentum of a small particle
Arnold Sommerfield
introduced 2 other quantum numbers: l & m,
ROYGBV
order of colors of visible spectrum
reflection
waves bounce off a smooth surface, in one direction; direction is changed (think of a mirror).
scattering
waves bounce off an uneven or rough surface, or tiny particles suspended in air, in multiple directions.
refraction
waves bend when they pass from something with one density to something with another density; for example, light bends when it goes from air to water. A prism spreads out light into its component wavelengths because each wavelength bends a different angle when going into and out of the prism.
diffraction
waves passing through a slit spread out on the other side. If two slits are close together, light spreads out from each one; these new waves overlap with each other and cause constructive and destructive interference
Wolfgang Pauli
discovered 4th quantum number, ms
Erwin Schrödinger
developed a wave equation involving 4 quantum numbers
linear
180°
trigonal planar
120°
tetrahedral
109.5°
trigonal bypyramidal
90°; 120°
octahedral
90°; 180°
steric no.: 2; 0 lone pair
linear

steric no.: 3; 0 lone pair
trigonal planar
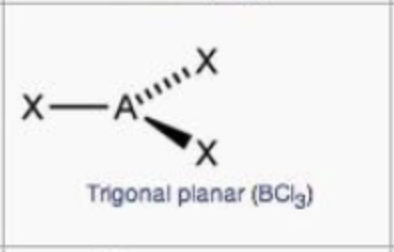
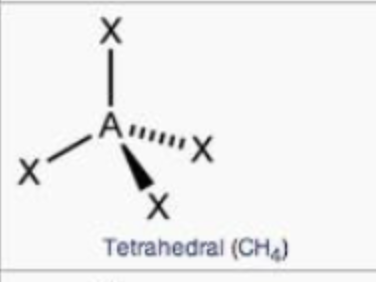
steric no.: 4; 0 lone pair
tetrahedral
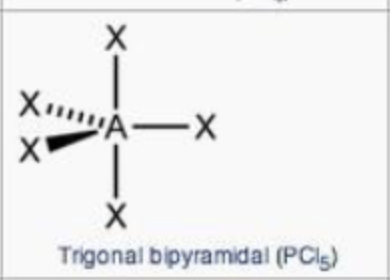
steric no.: 5; 0 lone pair
trigonal bypyramidal
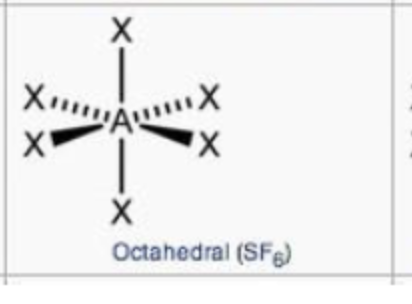
steric no.: 6; 0 lone pair
octahedral
steric no.: 3; 1 lone pair
bent
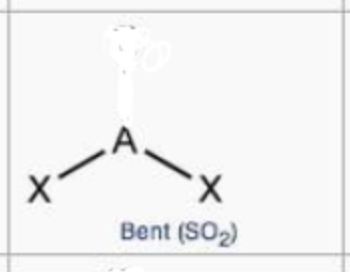
steric no.: 4; 1 lone pair
trigonal pyramidal
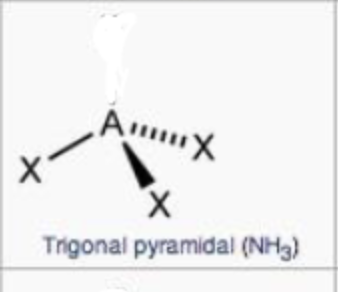
steric no.: 5; 1 lone pair
seesaw
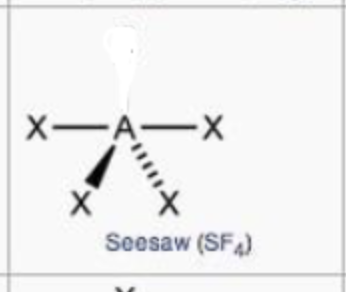
steric no.: 6; 1 lone pair
square pyramidal
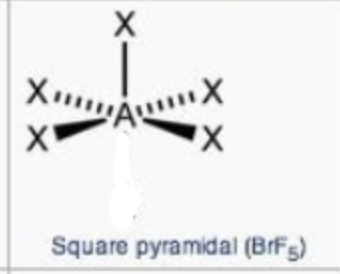
steric no.: 4; 2 lone pairs
bent
steric no.: 5; 2 lone pairs
T-shaped
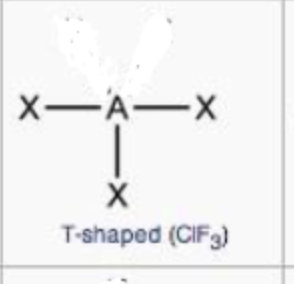
steric no.: 6; 2 lone pairs
square planar
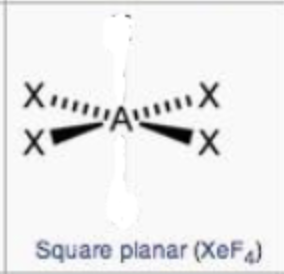
steric no.: 5; 3 lone pairs
linear
trigonal pyramidal
107°
bent
104.5°