Unit 21 Organic Chemistry
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
80 Terms
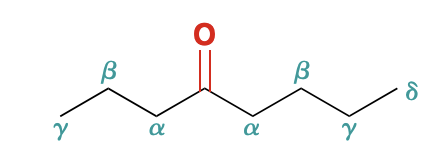
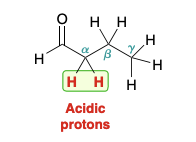
Alpha Carbon
Carbon adjacent to the carbonyl group
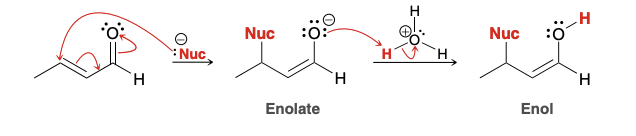
Enol
Enolate
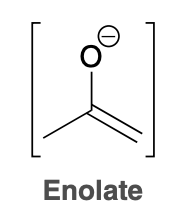
Ketone to Enol formation
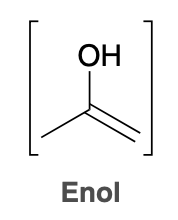
Recall that the ketone and enol shown are tautomers—rapidly interconverting constitutional isomers that
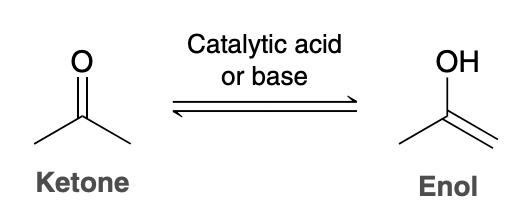
Does the position of equilibrium favor the ketone or enol
In general, the position of equilibrium will significantly favor the ketone, as seen in the following example
Exception to enol vs ketone equilibrium position favoring
1) The enol has a conjugated π system, which is a stabilizing factor (see Section 16.2), and (2) the enol can form an intramolecular H-bonding interaction between the hydroxyl proton and the nearby carbonyl group
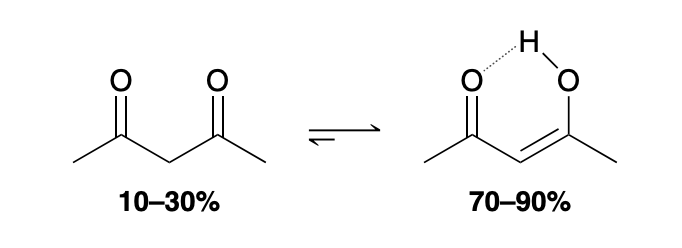
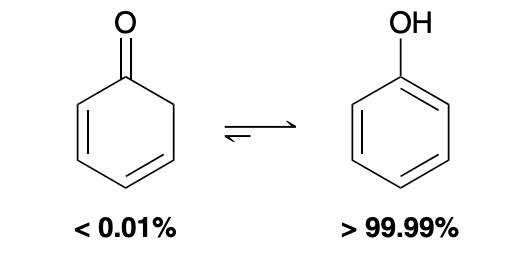
Extreme exception to enol vs ketone equilibrium position favoring
The aromatic enol is much more significant
Acid catalyzed tautomerization (ketone—> enol)
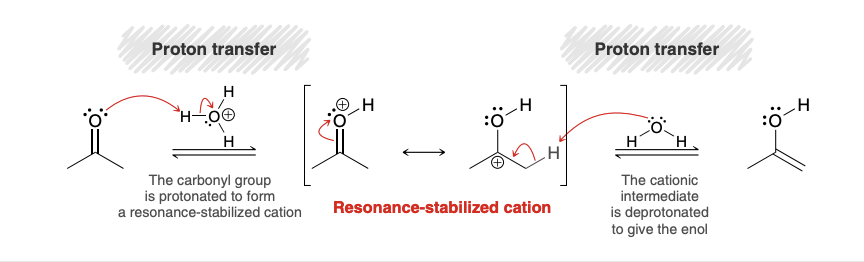
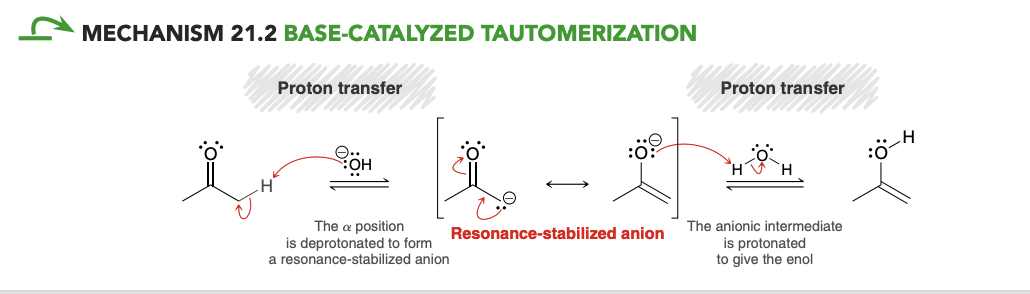
Base catalyzed tautomerization (ketone—> enol)
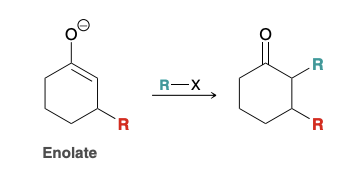
Unique feature of Enolates
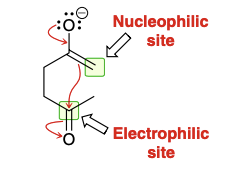
Enolates are ambident nucleophiles containing two nucleophilic sites; oxygen can attack an electrophile (O attack) and alpha carbon can attack an electrophile (C attack)
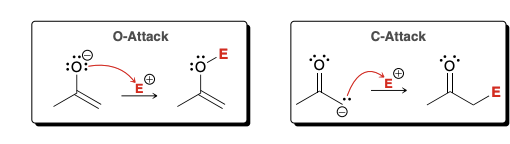
Is C attack or O attack more common?
C attack
Why are Enolates more useful than Enols
enolates possess a full negative charge and are therefore more reactive than enols and (2) enolates can be isolated and stored for short periods of time, unlike enols, which cannot be isolated or stored
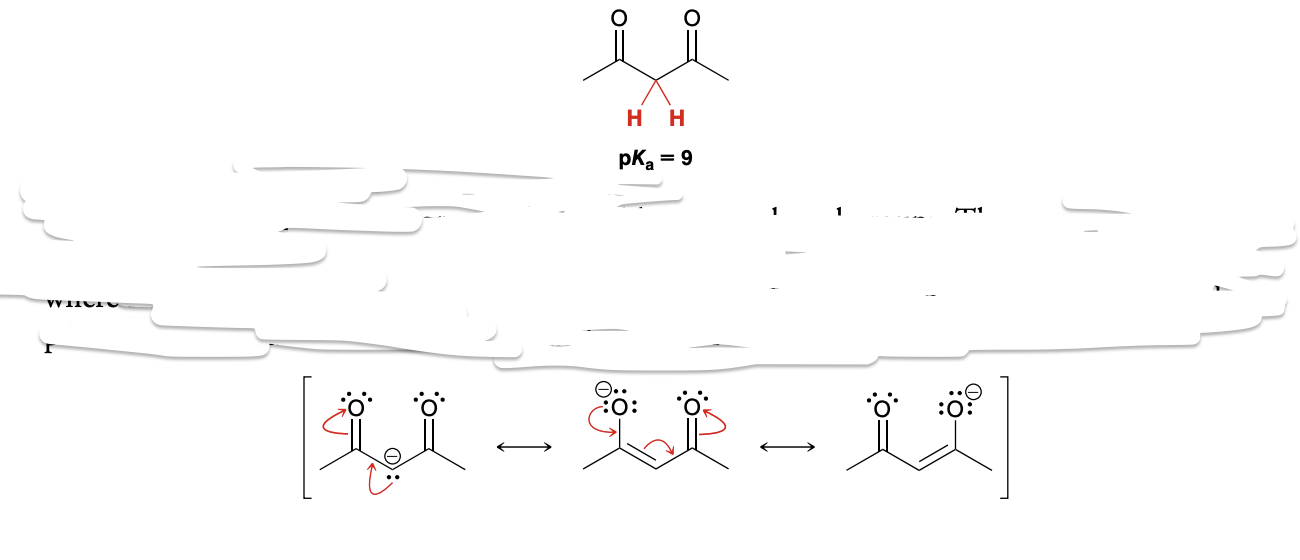
Which protons are acidic in aldehydes and ketones
only the α protons are acidic in aldehydes and ketones (formation of resonance stabilized anion).
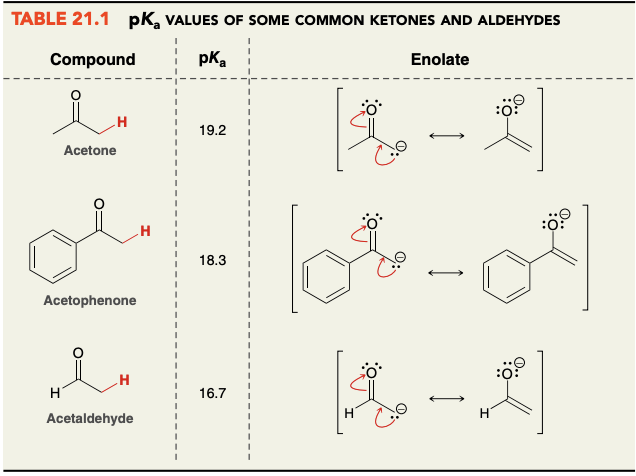
pKa values of common ketones/aldehydes(acetone, acetophenone, acetaldehyde)
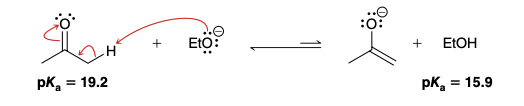
Deprotonation of acetone with NaOEt result
Equilibrium greatly favors the reactants (i.e acetone does not get greatly deprotonated); pKa of acetone is higher than the pKa of EtOH(formed as a result of acetone deprotonation), meaning that a EtOH is more likely to donate an H than acetone, and the more stable outcome is the presense of -OEt + acetone.
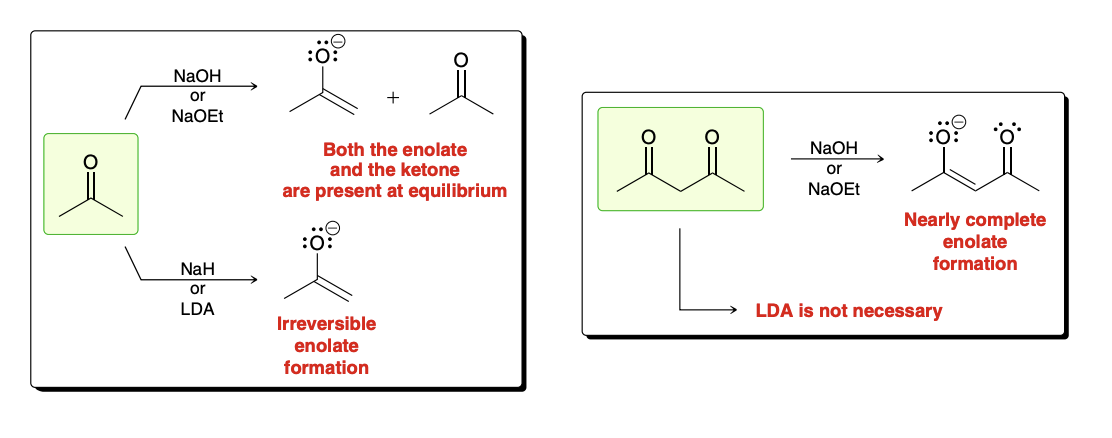
Irreversible deprotonation reagents
NaH (-H super unstable adn will form H2 gas)
LDA
High acidic/stabalized ketone
EtO- can be used to form enolate
choosing a base to form an enolate ion
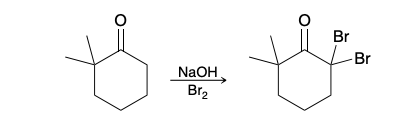
Alpha Halogenation of Enols and Enolates
Does not work with fluorine
The rate of halogenation is found to be independent of the concentration or identity of the halogen, indicating that the halogen does not participate in the rate-determining step
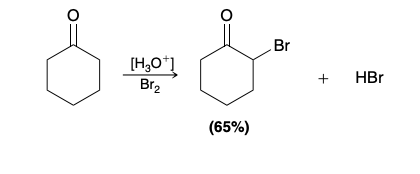
Acid-Catalyzed Halogenation of ketones mechanism
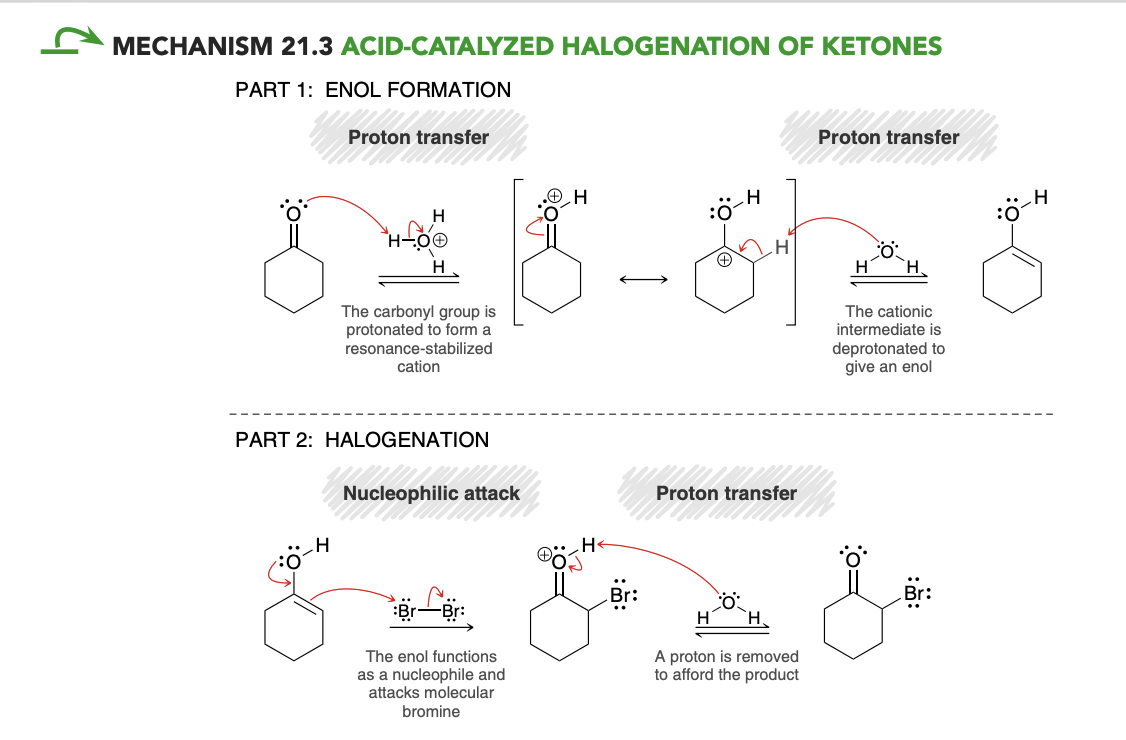
Bromination in unsymetrical ketone
bromination occurs primarily at the more substituted side of the ketone.
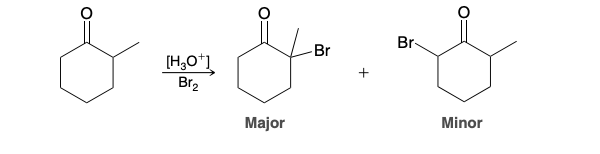
Halogenated ketone upon treatment with a base
pyridine, lithium carbonate (Li2CO3), or potassium tert-butoxide
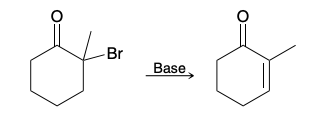
Creating alpha/beta unsaturation in ketone(introduction)
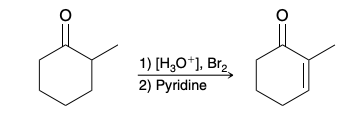
Hell–Volhard–Zelinsky reaction (Alpha Bromination of Carboxylic Acids)
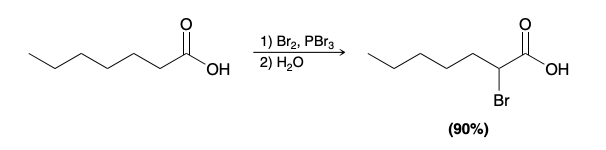
Alpha Halogenation in Basic Conditions (Haloform Reaction)
When more than one α proton is present, it is difficult to achieve monobromination in basic conditions, because the brominated product is more reactive and rapidly undergoes further bromination
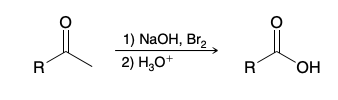
Carboxylic acid production from methyl ketone
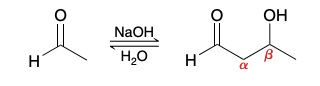
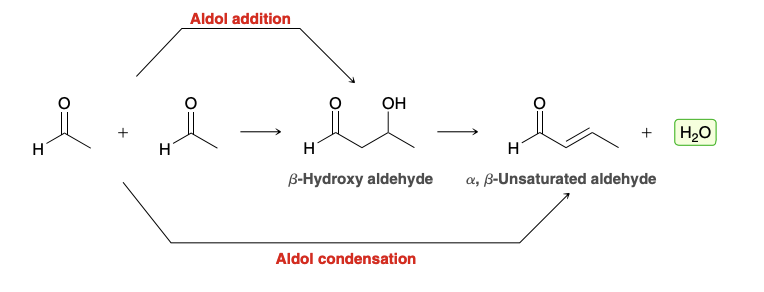
Aldol addition reaction
ald for “aldehyde” and ol for “alcohol”
product of an aldol addition reaction is always a β-hydroxy aldehyde or ketone
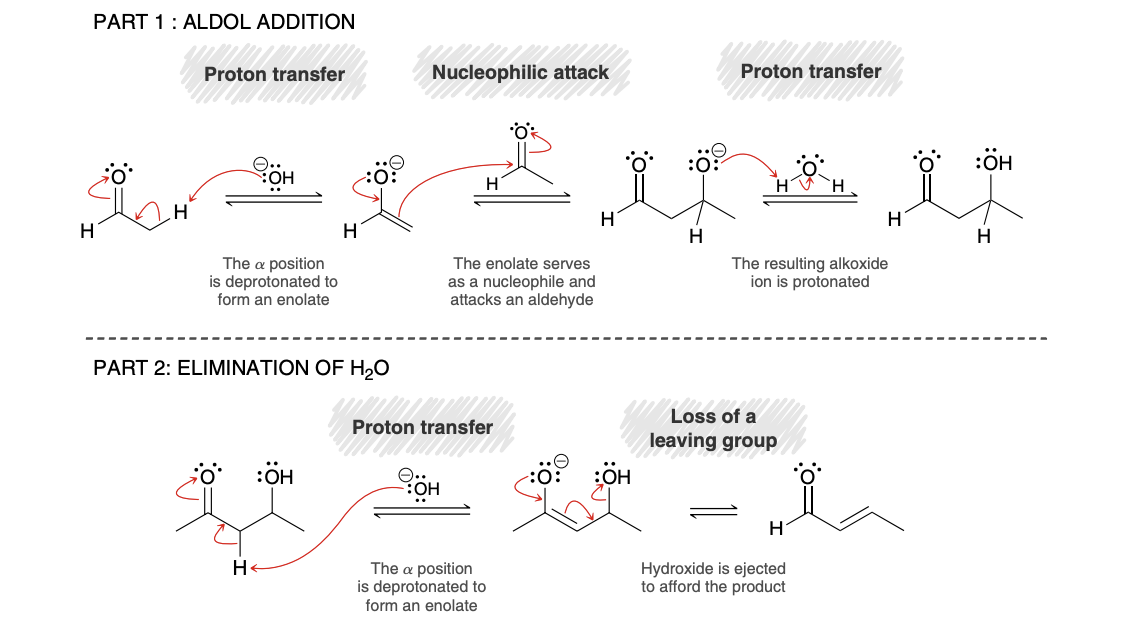
Aldol Addition Mechanism
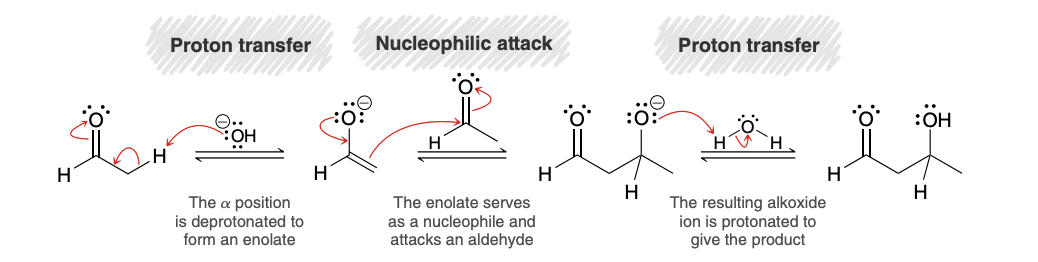
When comparing aldol vs simple aldehyde, which one does equilibrium favor?
For most simple aldehydes, the position of equilibrium favors the aldol product.
When comparing aldol vs simple ketones position, which one does equilibrium favor?
, for most ketones, the aldol product is not favored, and poor yields are common.
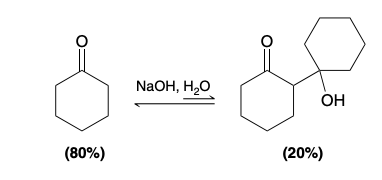
retro-aldol reaction
β-hydroxy ketone is converted back into cyclohexanone more readily than the forward reaction
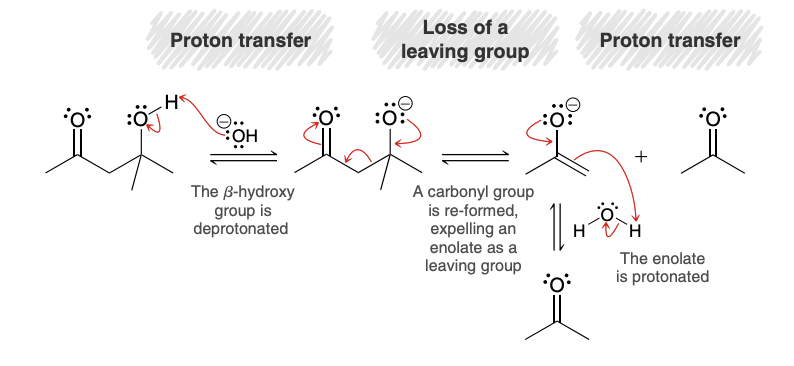
Retro-Aldol Reacction mechanism
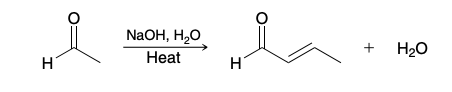
Aldol condensations (aldehyde to α,β-unsaturated aldehyde)
Overview of Aldol Addition/Condensation
All treated with H+/OH-
Aldol Condensation mechanism
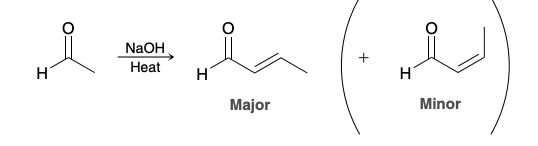
Stereochemistry in certain aldol condensation reactions
the product with fewer steric interactions is generally the major product.
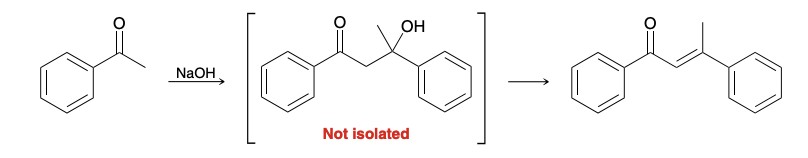
What is the driving force in the aldol condensation
Formation of a conjugated system
It is often hard very difficult to isolate products lacking the conjugated pi systems
Can conditions be manipulated to isolate certain products
Yes, aldol addition products can be isolated in lower temperatures, but at very low yield rate
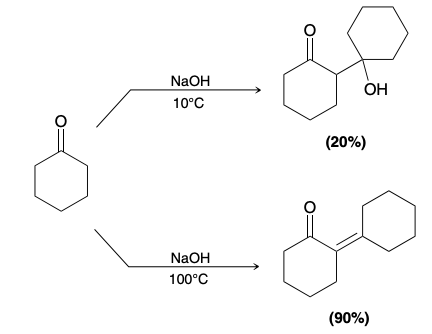
Potential Reactions
This reaction as is will produce a mixture of products which isn’t very useful
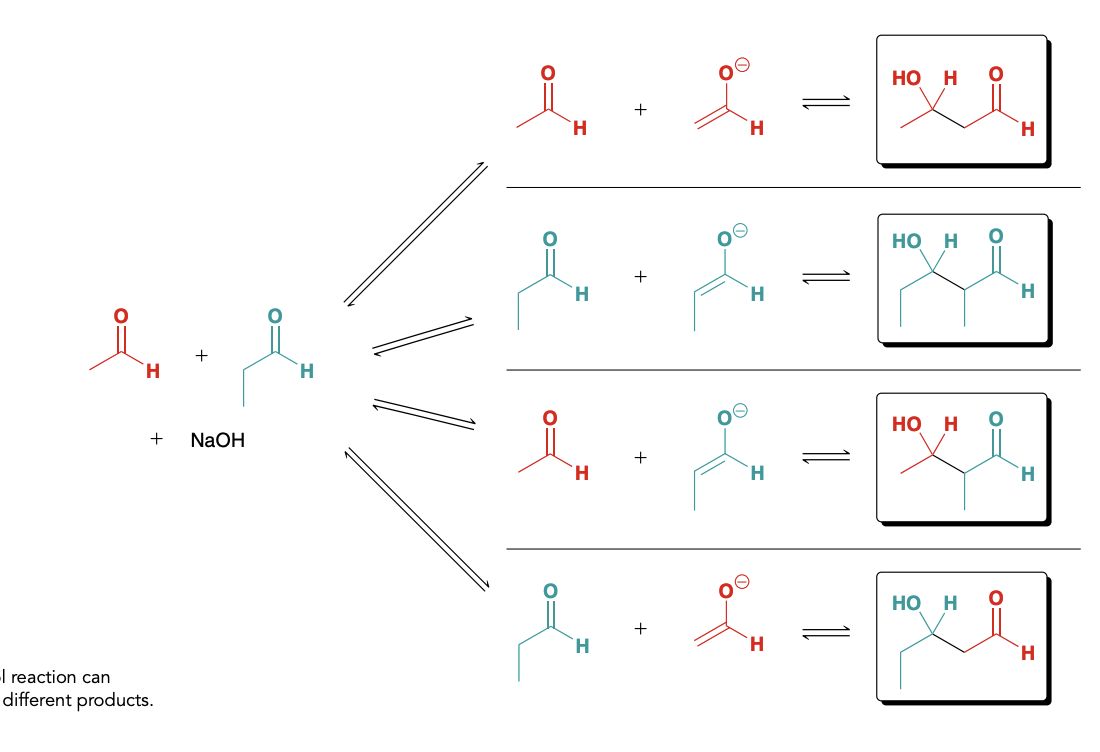
How to prevent a mixture of products from being formed
1)Preforming a crossed aldol reaction with an aldehyde lacking alpha protons and pocessing and unhinered carbonyl group
2)Crossed aldol reactions can also be performed using LDA as a base
Preforming a crossed aldol reaction with an aldehyde(under appropiate conditions)
Using an aldehyde lacking alpha protons and pocessing an unhindered carbonyl group, the other aldehyde is the only one that can turn into an enolate, allowing for the sole product to be a crossed aldol
Can use benzaldehyde and formaldehyde.
If benzaldehyde used, the dehydration step is spontaneous, and the equilibrium favors the condensation product rather than the addition product, because the condensation product is highly conjugated.
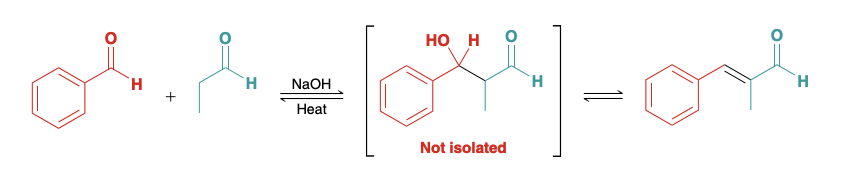
Crossed aldol reactions can also be performed using LDA as a base.
A specific ketone can be deprotonated to form an enolate using LDA, which can then be used to attack a separate aldehyde
it is possible for an enolate ion to function as a base (rather than a nucleophile) and deprotonate a molecule of propionaldehyde. If this process occurs too rapidly, then a mixture of products will result
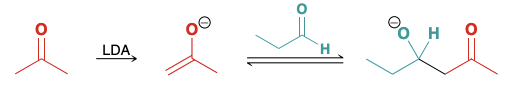
Intramolecular aldol reactions
Compounds containing two carbonyl groups can undergo these reactions
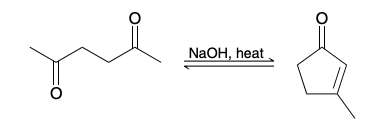
How does Intramolecular aldol reaction work
Must create a minimally strained 5 or 6 ringed product
Claisen Condenation Reaction
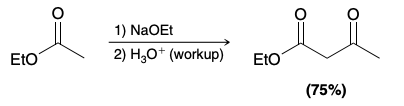
Claisen Condenation Reaction mechanism
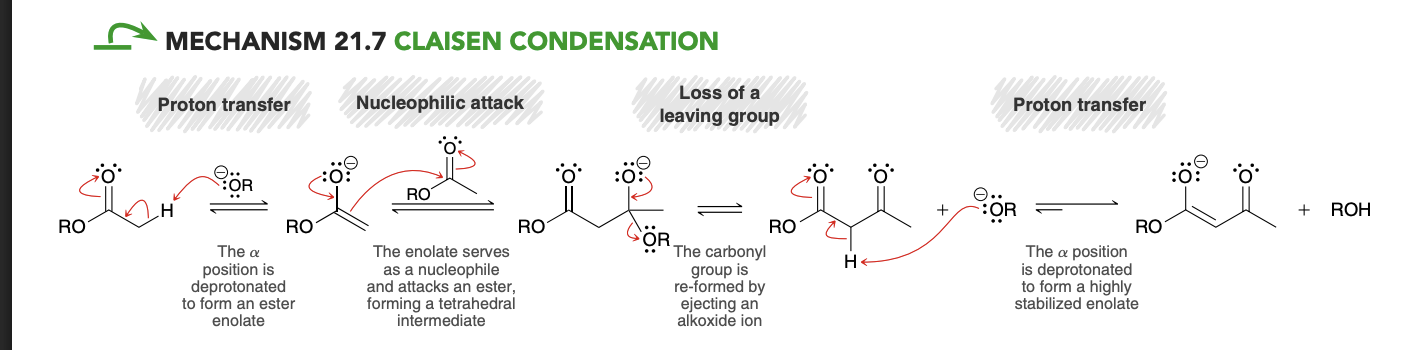
Last step (acidification) of Claisen condensation
In order to avoid the hydrolysis of this ester group (in the presence of H3O+), we must use an extremely mild source of aqueous acid. One common way to produce such conditions is to use a solution of ammonium chloride (NH4Cl) in water. Under these conditions, ammonium ions (NH4 +) are present in solution, rather than hydronium ions (H3O+).
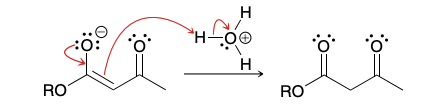
Can hydroxide be used for Claisen condensation
Hydroxide cannot be used as the base for a Claisen condensation because it can cause hydrolysis of the starting ester, as shown here:
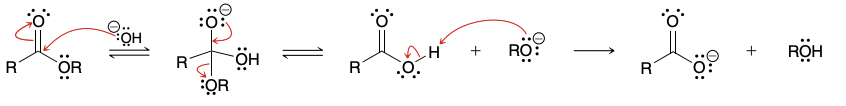
Wha
What strong base can be used in the claisen condensation
alkoxide from the same alkoxy group present in the starting ester(to prevent formation of a different ester
Intramolecular Claisen Condensations (starting material is a 6 carbon molecule with esters on both side)
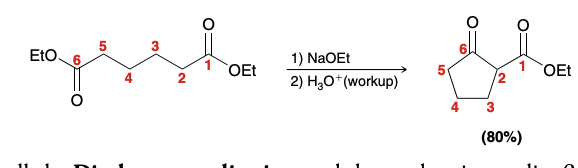
Alkylation via Enolate Ions
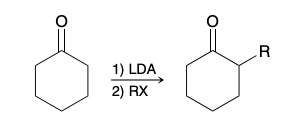
Thermodynamic enolate (unymetrical ketone)
More substituted double bond
More hindered, forms slower, more stable
Kinetic enolate (unymetrical ketone)
Less substiuted double bond
Less hindered to attack, forms quickly, less stable
Thermodynamic enolate vs kinetic enolate energy diagram
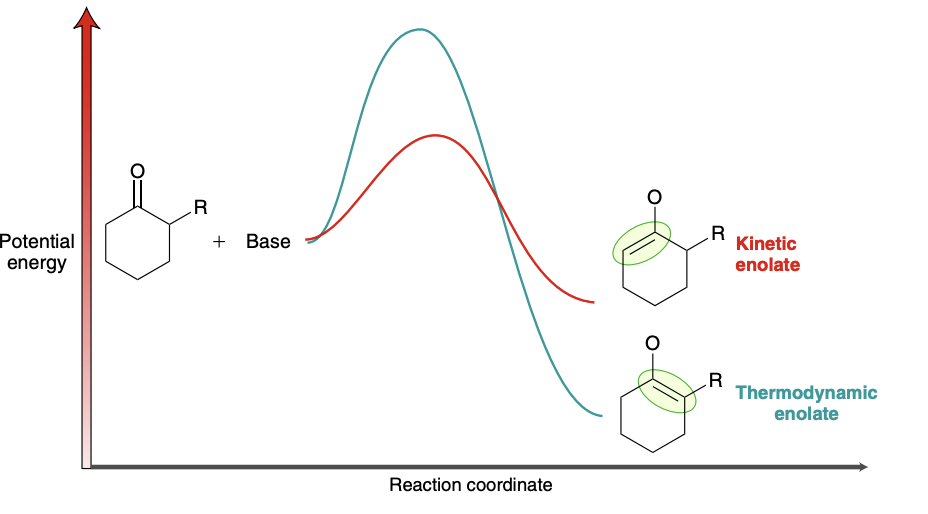
Formation of kinetic enolate
Use LDA at cold temperature
LDA is bulky/sterically hindered and will more readily attack the less hindered side
Formation of thermodynamic enolate
Use NaH at room temperature
NaH is a non-sterically hindered strong base, which mitigates steric hinderance
Thermodynamic enoalte vs kinetic enolate outcome
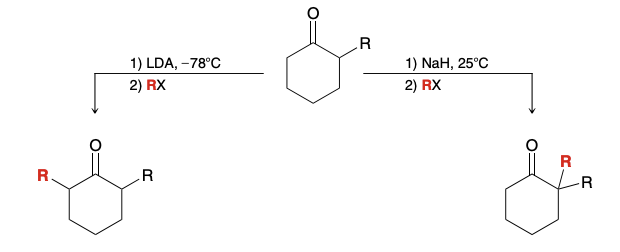
Why is Alkylation via Enolate Ions not a reliable method
in practice, the alkylation of ketones can be challenging and may lead to mixtures of polyalkylated products (
Ethyl acetoacetate
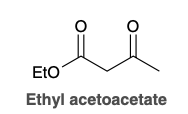
acetoacetic ester synthesis
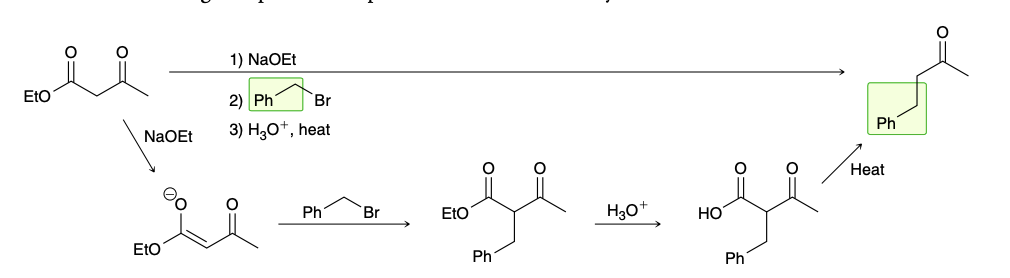
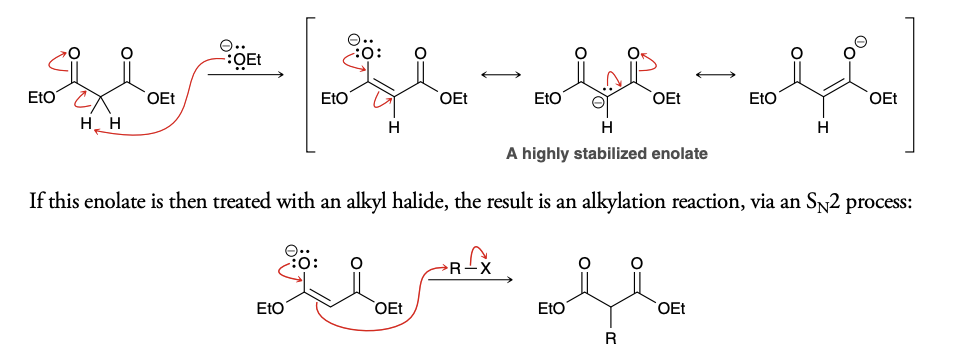
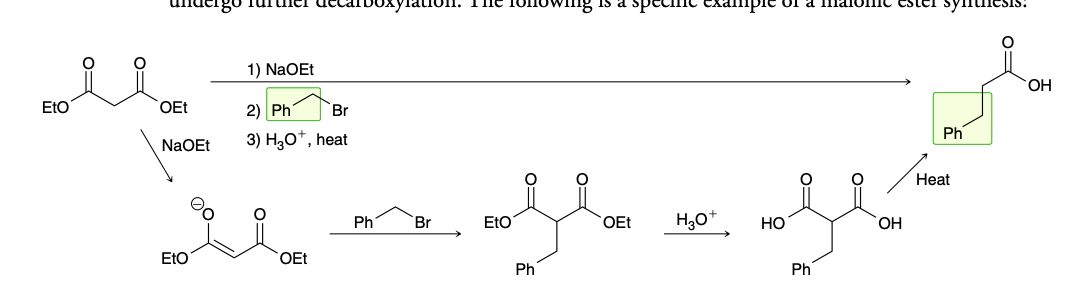
Malonic ester synthesis
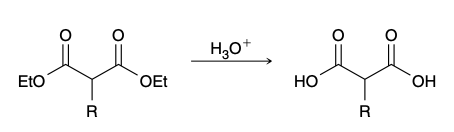
Conversion of ester groups into carboxylic acid
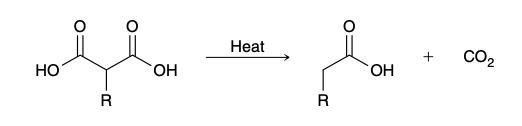
Decarboxylation (malonic ester synthesis)
hydrolysis step is performed at elevated temperatures

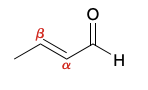
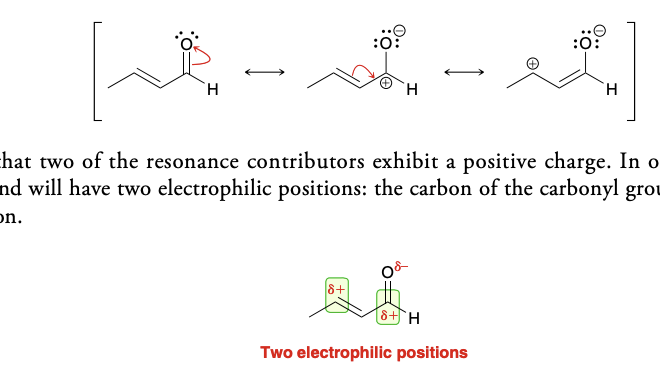
unsaturated alpha beta aldehyde and ketone nucleophilic positions
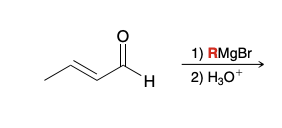
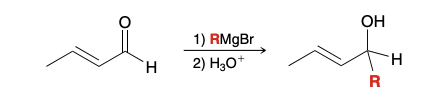
Which nucleophilic position will a Grignard reagent attack in unsaturated alpha beta aldehyde and ketone
Carbonyl
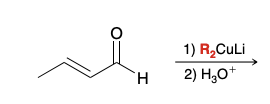
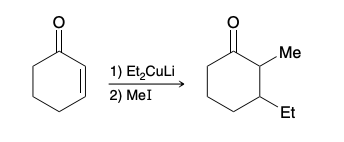
Which nucleophilic position will R2CuLi attack in unsaturated alpha beta aldehyde and ketone
ketone position
conjugate addition
Also called a 1,4 addition, the nucleophile and the proton have added across the ends of a conjugated π system
tautomerization(final product converting to a ketone) is stil a 1,4 addition
Michael reaction
starting diketone is deprotonated to form a highly stabilized enolate ion, which then serves as a nucleophile in a 1,4-conjugate addition
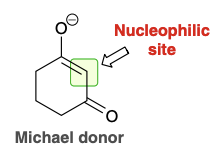
Michael donor
Highly stabilized enolate that attacks α,β-unsaturated aldehyde
Micahel acceptor
Electrophile that gets attacked
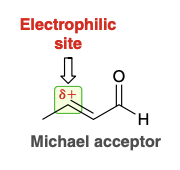
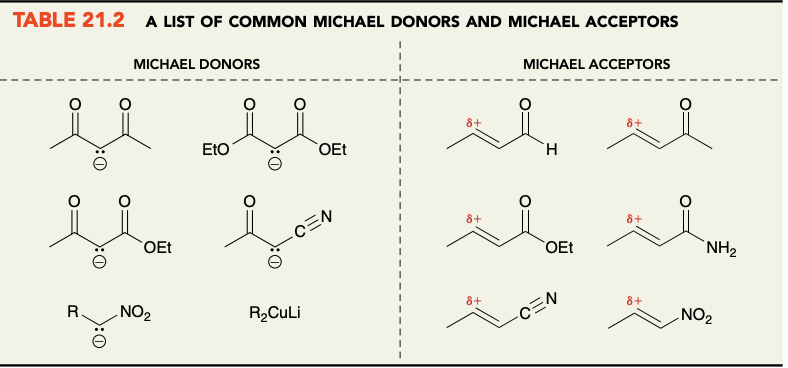
Common michael acceptors and donors
Donors to note: r-CN, r-NO2, and R2CuLi
Acceptors to note: R-CN, R-NO2
Similarity between enolate ion and enamine
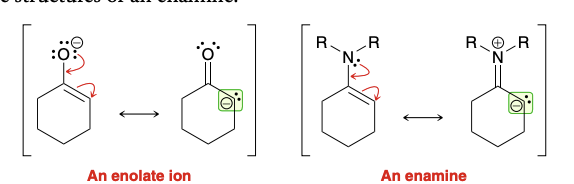
Can an enamine be a michael donor?
Yes. They are less reactive and are effect michael donors that will participate in michael reaction (with suitable Michael acceptor)
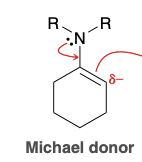
Michael reaction intermediate (enamine)
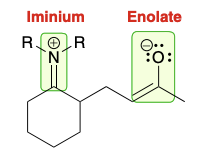
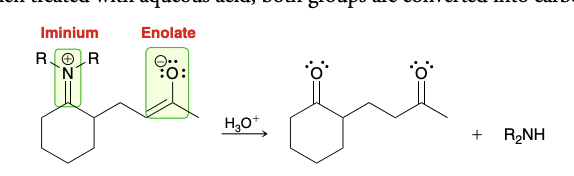
Treatment of michael reaction intermediate (enamine)
Use H3O+; converts both groups into ketones
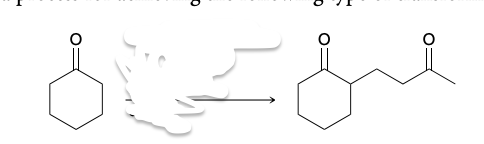
Stork synthesis
(1) formation of an enamine, (2) a Michael addition, and (3) hydrolysis.
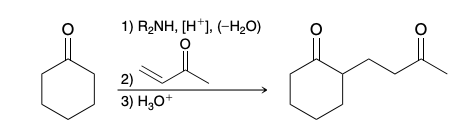
Alkylation of the Alpha and Beta Positions
Intermediate in Alkylation of the Alpha and Beta Positions