3.3.10.2 electrophilic substitution
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
define an electrophile
electron pair acceptor
what mechanism does benzene undergo?
electrophilic substitution
what are the two electrophiles we need to know?
⁺NO₂ (nitronium ion)
RCO⁺ (acylium ion)
[R−C≡O +]
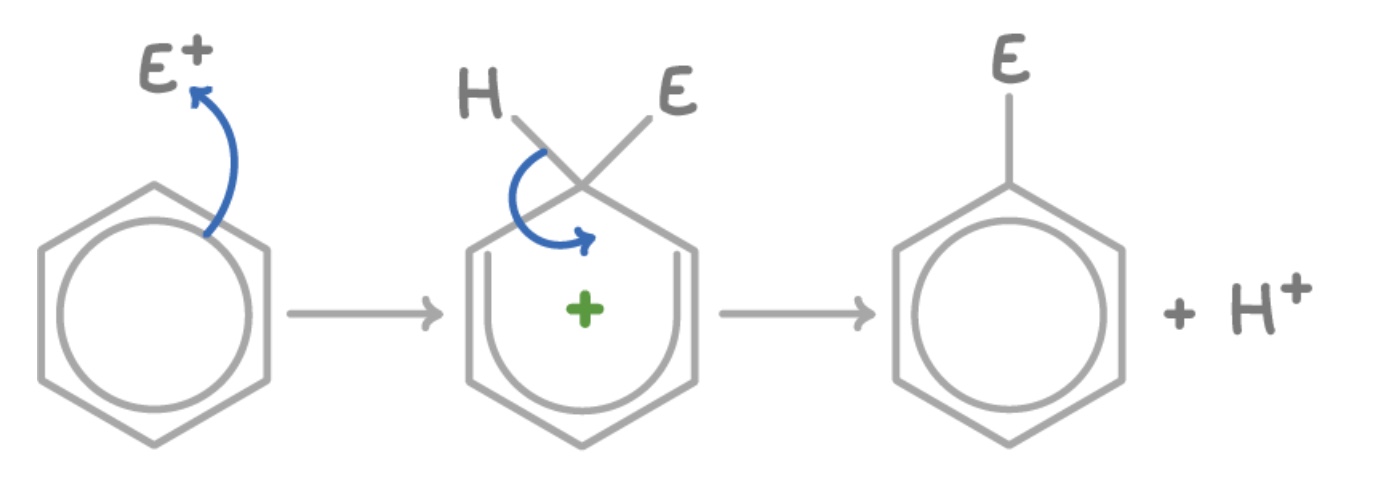
outline the steps of the general mechanism
(generation of electrophile): a catalyst is used to produce the electrophile, E⁺
electrophilic substitution
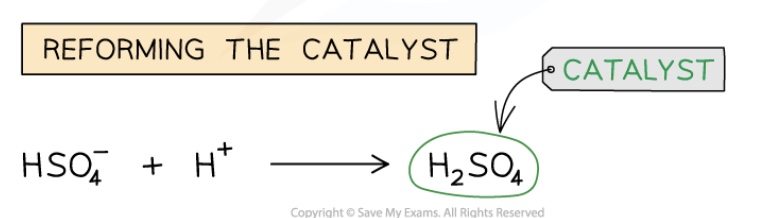
(regeneration of catalyst): the H⁺ liberated from the benzene reforms the catalyst
draw + outline the general electrophilic substitution for benzene
2 electrons from the delocalised ring form a new bond with the E⁺
2 electrons from C-H bond go back to the ring to complete it
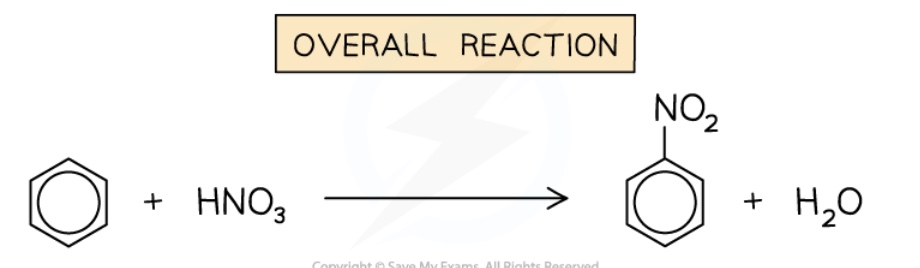
outline nitration of benzene
benzene reacts with a mixture of concentrated HNO₃ + concentrated H₂SO₄ (catalyst) at 50℃ to form nitrobenzene
mechanism is electrophilic substitution
give the overall equation for the nitration of benzene
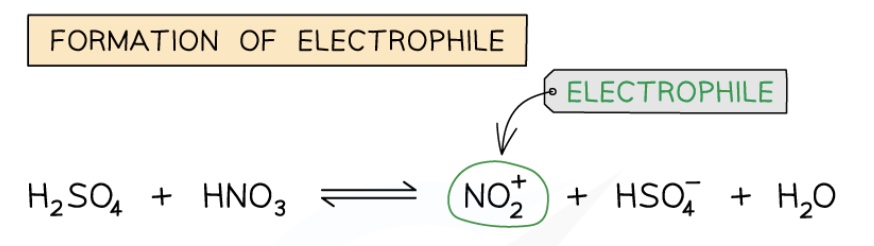
describe step 1 (generation of electrophile) of nitration of benzene + give the 2 equations
H₂SO₄ protonates the HNO₃ as it is a stronger acid
H₂SO₄ + HNO₃ → HSO₄⁻ + H₂NO₃⁺
the protonated nitric acid breaks down + forms the nitronium ion (nitric cation) + water
H₂NO₃⁺ → NO₂⁺ + H₂O
combine the 2 equations of step 1 to give an overall equation for the generation of the electrophile
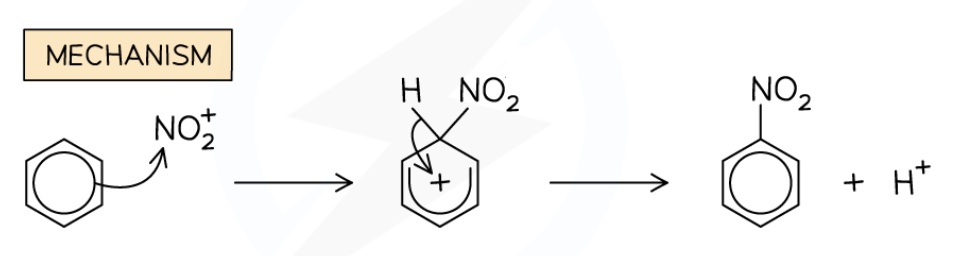
draw the mechanism for the nitration of benzene
give the equation for the regeneration of the catalyst
give 2 uses of nitrobenzene
synthetic uses
making TNT
nitrobenzene can be used in the preparation of what?
azo dyes
describe how nitrobenzene is used in the preparation of azo dyes
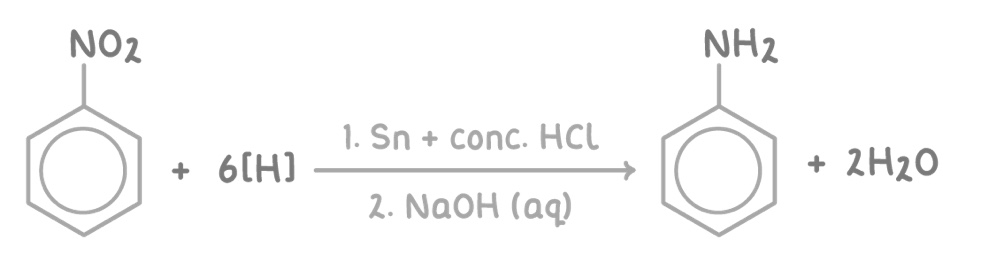
give the overall equation
nitro group is reduced to an amine
reagent: HCl
condition: Sn catalyst
nitration can occur more than….
more than once
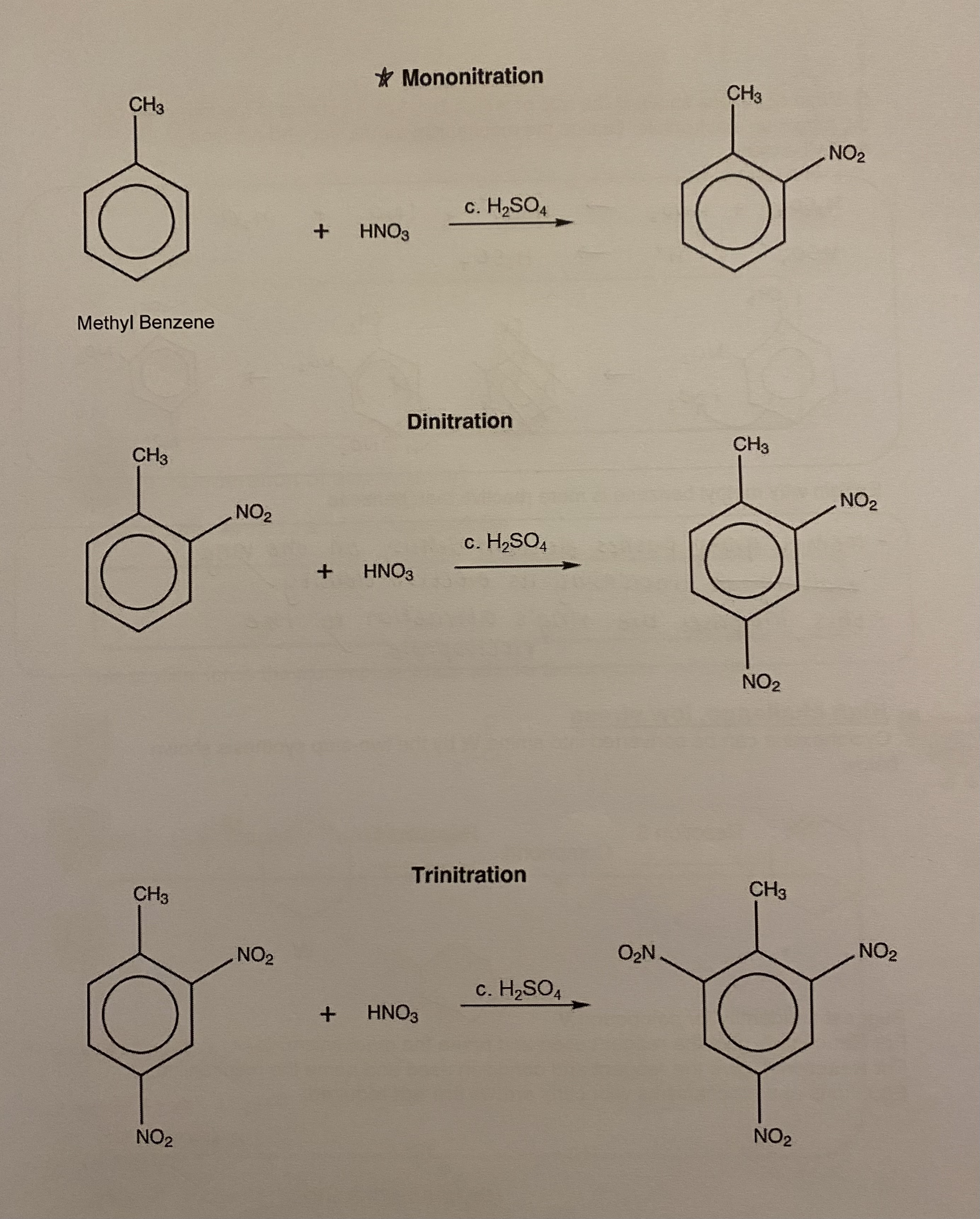
methylbenzene is more ____ than benzene, what does this mean?
reactive
this means that the nitration reaction can be carried out at a lower temperature
why is methyl benzene more reactive than benzene?
methyl group pushes electron density on the ring, increasing its electron density
this increases the ring’s attraction to the electrophile
why does benzene not undergo electrophilic addition instead of substitution?
because this would involve permanently breaking the ring of delocalised electrons, which would make benzene unstable (the ring gives it stability)
give another reaction that benzene undergoes
Friedel-Crafts acylation reactions
what mechanism does Friedel-Crafts acylation involve?
electrophilic substitution
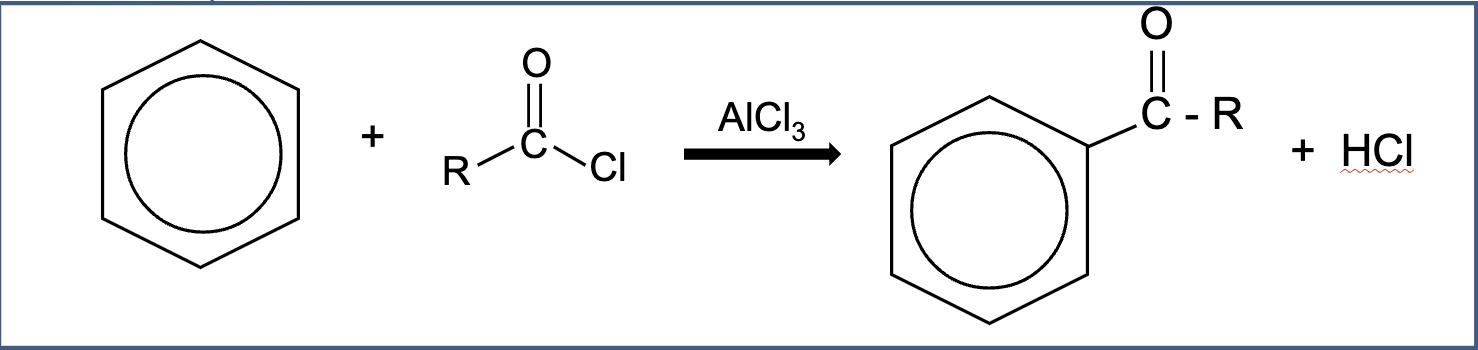
outline what Friedel-Crafts acylation involves
benzene reacts with acyl chlorides (RCOCl) in the presence of AlCl₃ to form a ‘phenyl ketone’
give the overall general equation for the reaction between acyl chlorides and benzene
what are the 3 steps of friedel-crafts acylation?
generation of electrophile
electrophilic substitution
regeneration of catalyst
outline step 1 (generation of electrophile) + give the equation for it
AlCl₃ removes the Cl from the acyl chloride
ROCl + AlCl₃ → RC⁺O + AlCl₄⁻
what is the electrophile involved in this mechanism? give its structure
the acylium ion is the electrophile which attacks the benzene molecule
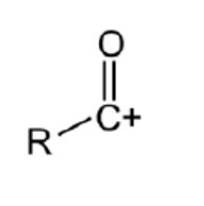
outline the general mechanism of electrophilic substitution for acylation
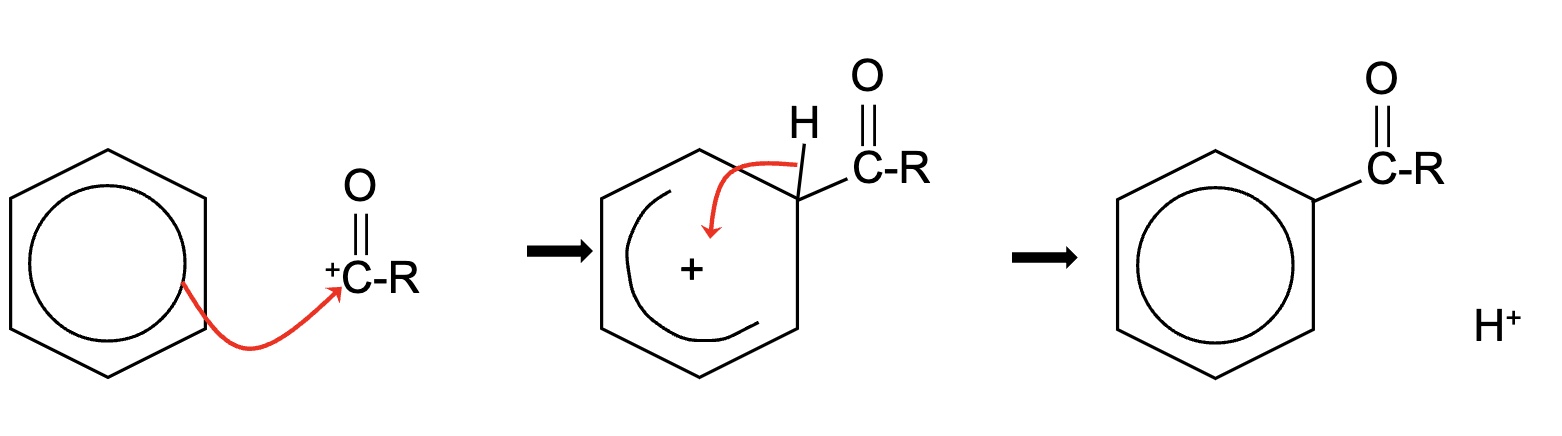
give the equation for step 3 (regeneration of the catalyst)
