chemistry of statins
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
what are the functions of cholesterol?
steroid hormones
bile acids
biological membranes
cholesterol is the precursor for many hormones including
sex hormones
tissue growth hormones
adrenocortical hormones
what is cholic acid's role
added to emulsify water insoluble foods
What is the role of cholesterol and how does it relate to cardiovascular risk?
Role: Important for biosynthesis and cell membrane structure.
Transported by LDLs (to cells) and HDLs (from cells to liver).
High LDL or low HDL levels → increased mortality.
Can cause fatty plaques → risk of atherosclerosis, clots, stroke, heart attack.
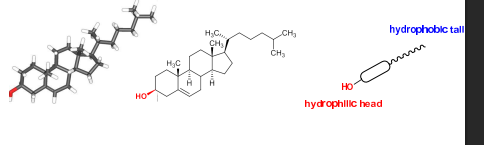
what is the structure of cholesterol?
OH group is polar,hydrophillic so can do hydrogen bonding
alchohol can be HBD+HBA
What are the 3 main phases of cholesterol biosynthesis?
Formation of mevalonic acid.
Conversion of mevalonate → farnesyl pyrophosphate.
Condensation of two farnesyl pyrophosphate units → squalene.
How is cholesterol production regulated in the body?
Key enzyme: HMG-CoA reductase.
Regulation occurs via:
Kinase activity (phosphorylation/inactivation)
Transcription & translation (enzyme synthesis)
Enzyme degradation
Feedback control: Regulated by cholesterol concentration.
How do statins work in cholesterol biosynthesis?
Target enzyme: HMG-CoA reductase (rate-limiting step).
Mechanism: Block the reduction of HMG-CoA → mevalonic acid in Phase 1.
Chemistry: Involves two reduction steps to form mevalonate.
Effect: Lowers cholesterol synthesis in the liver.
But cholesterol can still be obtained from our diet.
How does HMG-CoA bind to its enzyme during catalysis?
Positively charged Lys-735: forms ionic bond with substrate
Hydrogen bonds: Ser-684, Asp-690, Lys-691 stabilize substrate
Hydrophobic interactions: substrate fits into narrow hydrophobic slot
What is the general structure and key feature of Type I statins? Lovasaatin and simvasattin
Structure: Polar “head” + hydrophobic moiety (including decalin ring)
Prodrugs: Lovastatin and Simvastatin contain a lactone ring that is hydrolysed to form the polar head
Why are lovastatin modifications needed in drug design?
Statins are competitive inhibitors, so high levels of HMG-CoA can reduce their effectiveness.
- Modified versions of lovastatin are made to bind more strongly and stay longer in the enzyme’s active site, so the drug works better and lasts longer.
What are the main structural modifications of lovastatin?
Change the side chain (ether side-chain modifications)
Extend the lactone ring (make it slightly bigger)
Convert it to a mevalonate-like form (adjust the shape/stereochemistry at the OH carbon)
What are the main shortcomings of Type I statins?
type 1 stations are liphillhic so cross bbb cns related side effects:sedation. msucle pains . Gi issues
difficult to synthesize, mainly from mould cultures → semi-synthetic
Structural complexity: many asymmetric centres
Type II Statins – How does hydrophobicity affect their action?
Less hydrophobic statins target liver cells more specifically and have lower side effects.
Reason: Less hydrophobic statins don’t cross cell membranes easily, but liver cells have transport proteins for statins.
Why do statins mainly act in the liver?
Majority of cholesterol synthesis occurs in liver cells.
Liver cells express specific transport proteins for statins.
Common side effects of statins
Myalgia (muscle pain) is common.
Severe muscle toxicity = rhabdomyolysis, which can be fatal.
Type I vs Type II statins – structural features & side effects
Feature | Type I Statins | Type II Statins |
|---|---|---|
Origin | Natural / semi-synthetic | Fully synthetic |
Lipophilicity | More lipophilic (hydrophobic) | Less lipophilic |
Cell entry | Passive diffusion across membranes | Transported into liver by proteins |
Targeting | Enter many tissues (including muscle) | More liver-selective |
Side effects | Higher risk (especially muscle toxicity) | Lower risk |
Reason for side effects | Can enter muscle cells easily | Limited entry into non-liver cells |
what makes the differnces in the lipohillicity?
type 1 : Contain a decalin ring or bulky non-polar groups → increases hydrophobic character.+lipophhilic
type 2: fully synthetic;Have larger polar/ionic groups attached to the ring system (like fluorophenyl, pyrimidine).don’t have the decalin ring
These increase hydrophilicity,
Type II statins – structure & side effects
Type II statins are less lipophilic overall than Type I, but they still retain a hydrophobic core (aromatic or cyclic rings) that is essential for HMG-CoA reductase binding.
The difference is that Type II statins have additional polar/ionic groups attached to the hydrophobic core. These reduce overall lipophilicity,
Statins – Mechanism of Action
Compete with HMG-CoA → block the enzyme (HMGR)
Polar head mimics HMG-CoA → binds in the active site
Hydrophobic part adds extra binding → stronger attachment
Bind very tightly but don’t react → act as transition-state analogues
Mimic reaction intermediate (mevalonate-like) → effectively stop the enzyme
Atorvastatin – Binding Interactions
Polar head mimics HMG-CoA → forms:
H-bonds with Ser-565
Ion–dipole interaction with Arg-590
Hydrophobic part → enzyme changes shape to form a pocket
Methylethyl group → fits same site as decalin ring (Type I statins)
Fluorophenyl group → interacts with Arg-590 (important contact)
Rosuvastatin – Binding Interactions
Polar head mimics HMG-CoA → H-bonds with Ser-565
Sulfone group → extra H-bond with Ser-565 + interacts with Arg-568
Hydrophobic part → fits into flexible pocket
Result: stronger binding → high potency
Key structural features of a statin (SAR)
Polar head group → mimics HMG-CoA → essential for binding to HMG-CoA reductase
Hydrophobic core → fits into enzyme pocket → stabilizes binding
Stereochemistry →correct 3,5-hydroxyl positions needed
Ring system & substituents → modulate potency and selectivity
Statin metabolism for atorvastatin, pitavastatin and fluvastatin they have
hydroxylation on aromatic rings
what is the primary metabolite?
why is the OH group the primary site of oxidation
OH group - site of oxidation
The way CYP450 interacts with the drug structure
why is the para- position favoured
it allows for stable intermediate formation as it is electron rich due to resonance effects from the adjacent heterocycle
Atorvastatin – Primary Metabolism & Activity
forms ortho- and para-hydroxy active metabolites
Fluorophenyl: –I/–R effect stabilizes transition state; fits hydrophobic pocket → stronger binding
Pyrrole: conjugated ring, +R effect, planar → optimizes enzyme interactions
Overall: both stabilize binding → potent HMG-CoA reductase inhibition