Patho Lecture 13: Acid-Base Balance
1/143
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
144 Terms
7.35 - 7.45
what is the normal pH range in the bloodstream?
20 parts to a base to every 1 part of an acid
how many parts base are there to parts of acid?
hydrogen
the main component that determines acidity and alkalinity
acid
releases H+; substance that dissociates into H+ ion and conjugate base
base
substance that takes up H+ ions
- greater the H+ = more acidic = lower pH
- lower the H+ = more alkaline = higher pH
what is the relationship between the H+ concentration and pH
HCO3 (bicarbonate)
what is considered the most important base in the body?
-protein metabolism
-carbohydrate metabolism
-fat metabolism
body acids are formed as end products of what 3 types of metabolism?
-lungs
-kidneys
-bone
what are some major organs involved in regulating the acid-base balance?
1. volatile (weak acids)
2. nonvolatile (strong acids)
what are the 2 forms of body acids?
carbonic acid (H2CO3)
what is an example of a volatile acid?
eliminated as CO2 gas, which we can exhale
what can carbonic acid (H2CO3) be eliminated as?
-sulfuric (H2SO4)
-phosphoric (H3PO4)
-lactic acid
what is an example of a nonvolatile acid?
can be eliminated by kidneys/urination
how are nonvolatile acids eliminated by the body?
metabolic acid
general term that includes all acids except carbonic acid
during aerobic metabolism
when is carbonic acid (H2CO3) formed?
CO2 and H2O
what is carbonic acid (H2CO3) derived from?
buffered and excreted by kidneys
how are metabolic acids eliminated from the body?
buffer
minimizes changes in pH when extraneous acids or bases are added to the solution; a pair of chemicals (weak acid and base)
circulates throughout the body
where does bicarbonate circulate in the body?
buffers metabolic acid produced by cells --> can release H+ if too little acid present (reverse if there is too much)
what is the role of bicarbonate in the body?
bicarbonate circulates in body --> picking up H+ when we need it --> takes it to the kidney --> attaches it to ammonia --> ammonia is excreted
what is the relationship between bicarbonate and ammonia?
excretes carbonic acid (gas form)
what type of acids does the respiratory system excrete?
excretes all other metabolic acids
what type of acids do the kidneys excrete?
not overwhelmed by amount of acid generated
when optimal acid-base balance occurs, what is true of buffers?
-carbonic acid
-metabolic acid
what types of acids can be excreted via acid excretion?
body excretes more acid
during hyperventilation, what happens to carbonic acid?
body holds onto more acid
during hypoventilation, what happens to carbonic acid?
chemoreceptors
receptors that focus on CO2 and H+ level and adjust rate and depth accordingly
exhaled through the lungs
where is carbonic acid exhaled through?
excreted out of the kidneys
where are metabolic acids excreted out of?
H+ combines with NH3 --> becomes NH4+ --> excreted in urine
how is ammonia excreted and where is it excreted?
-respiratory system
-renal system
-phosphate system
-plasma protein system
what are some of the most prominent buffer systems in the body?
phosphate buffer system
buffer system that functions similarly to bicarbonate system; maintains intracellular pH; in extracellular fluid
plasma protein system
buffer system that allows hemoglobin and other plasma proteins to buffer H+; maintains intracellular pH
tissue perfusion
must have this to bring blood, oxygen, and nutrients to the cells and get rid of waste products
-ensure blood flow carries away CO2
-ensure oxygen/nutrients get to cells to maintain aerobic metabolism
-preventing accumulation of lactic acid
what is tissue perfusion essential for in the body?
potassium and calcium
changes in H+ concentration impact which minerals that ultimately impact smooth muscle contraction in vessels?
↓ cardiac contractility (cannot pump effectively)
what effect does acidosis have on cardiac contractility?
- decreases cardiac contractility
- ↑ vasoconstriction of blood vessels
what effect does alkalosis have on cardiac contractility?
alters ability of hemoglobin to bind and transport and release O2
what effect does H+ concentration have on hemoglobin?
describes the unloading/unbinding of O2 from hemoglobin
what is the purpose of the oxyhemoglobin dissociation curve?
-shift to right
-decreased affinity for O2
-doesn’t hold onto O2 very well
describe the effect that acidosis has on the oxyhemoglobin dissociation curve
-shift to the left
-↑ affinity for O2
-hemoglobin doesn't like to let go of O2 and holds onto it
describe the effect that alkalosis has on the oxyhemoglobin dissociation curve
more H+ binds to protein
↓
fewer sites open for calcium to bind
↓
hypercalcemia in the bloodstream
in a state of acidosis, what is the relationship between H+ and calcium?
blocks sodium channels (in nerves & muscles) --> muscle weakness
when there is increased levels of calcium in the bloodstream as a result of acidosis, what is the effect on the body (neuromuscular)?
calcium is able to bind everywhere to the protein sites
↓
H+ is not
↓
hypocalcemia in the bloodstream
in a state of alkalosis, what is the relationship between H+ and calcium?
associated with positive symptoms (hyperactive muscle response)
when there is decreased levels of calcium in the bloodstream as a result of alkalosis, what is the effect on the body (neuromuscular)?
-Chvostek's sign
-Trousseau's sign
-paresthesia
-convulsions
-laryngospasm
-tetany
what are some positive symptoms that result from decreased levels of calcium in the bloodstream (in a state of alkalosis)?
Chvostek's sign
spasm of the facial muscles produced by sharply tapping over the facial nerve; suggestive of hypocalcemia
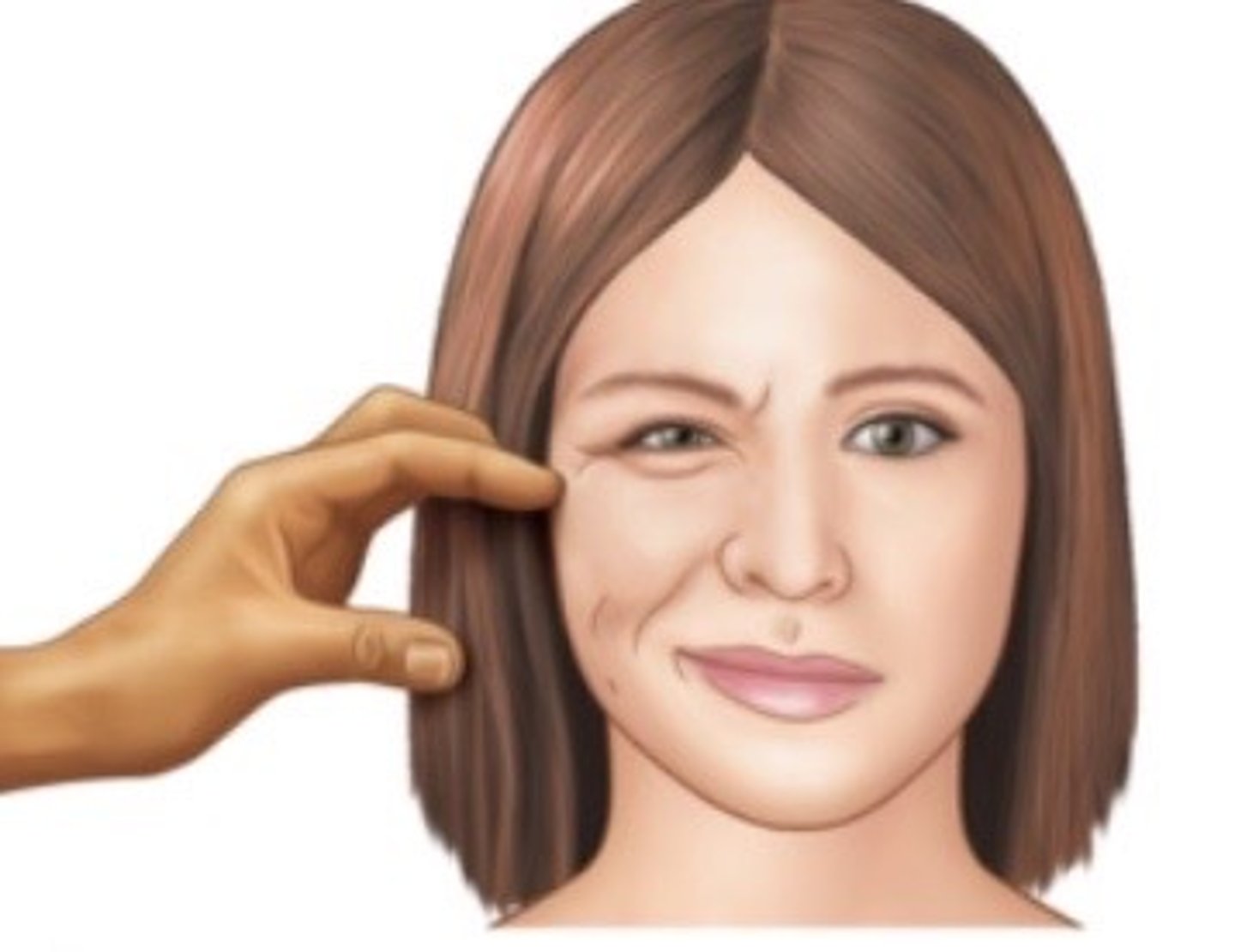
Trousseau's sign
hand/finger spasms with sustained blood pressure cuff inflation; suggestive of hypocalcemia

-acidosis = cerebral vasodilation
-alkalosis = cerebral vasoconstriction
what effect does acidosis and alkalosis have on the CNS?
cerebral vasodilation --> ↑ blood flow --> ↑ ICP
(try to get more O2 and blood flow to the brain)
explain what happens during cerebral vasodilation as a result of acidosis
cerebral vasoconstriction --> ↓ blood flow --> ↓ O2 delivery to brain
(brain is getting more than enough O2 so blood flow is decreased)
explain what happens during cerebral vasoconstriction as a result of alkalosis
H+ likes to swap places with K+
regarding electrolyte levels, what does H+ like to switch places with in a state of acidosis?
H+ enters cells and K+ moves out --> results in hyperkalemia
what happens in a state of acidosis with H+ and K+?
mainly affects the heart
what part of the body is mainly affected by potassium imbalances?
↓ cardiac contractility
what effect does potassium have on the heart when the body is in acidosis?
some H+ moving out of cells with K+ moving in --> results in hypokalemia
what happens in a state of alkalosis with H+ and K+?
-cardiac dysrhythmias
-contractility will ↑, but then will rapidly drop
what effect does potassium have on the heart when the body is in alkalosis?
↓ in contractility
what is the relationship between acidosis and cardiac contractility?
↑ contractility (up to pH 7.7), then rapidly ↓
what is the relationship between alkalosis and cardiac contractility?
something going on w/ the lungs
in respiratory types of acidosis/alkalosis, what is the main part of the body impacted?
something wrong w/ the bicarbonate level
in metabolic types of acidosis/alkalosis, what is the main part of the body impacted?
respiratory acidosis
too much carbonic acid (CO2)
respiratory alkalosis
too little carbonic acid (CO2)
-cause: hypoventilation (not getting enough O2)
-correction: hyperventilation (brain tries to blow off more CO2 and bring in more oxygen)
what is the cause and correction for the problem of respiratory acidosis?
-cause: hyperventilation
-correction: hypoventilation
what is the cause and correction for the problem of respiratory alkalosis?
- ↑ secretion of H+
- ↑ NH3 (ammonia) production
describe some compensation methods for respiratory acidosis
- ↓ secretion of H+
- ↓ NH3 (ammonia) production
describe some compensation methods for respiratory alkalosis
metabolic acidosis
too much metabolic acid
metabolic alkalosis
too little metabolic acid
-cause: ↑ metabolic acid than kidneys can excrete
-correction: ↑ secretion of H+ and NH3 production
what is the cause and correction for the problem of metabolic acidosis?
-cause: ↑ bicarbonate than kidneys can excrete
-correction: ↓ secretion of H+ and NH3 production
what is the cause and correction for the problem of metabolic alkalosis?
hyperventilation (get rid of CO2 gas)
describe some compensation methods for metabolic acidosis
hypoventilation (hold onto more CO2 bring back balance)
describe some compensation methods for metabolic alkalosis
pH is too low or on lower range of normal
what is characteristic of the pH in acidosis?
-↓ excretion of metabolic acid
-excessive excretion of bicarbonate
-excessive production/intake of metabolic acids
what are some causes of metabolic acidosis?
-renal disease
-oliguria
-kidneys not working as they should
in regard to metabolic acidosis, what happens when there is reduced excretion of metabolic acid?
prolonged diarrhea
what is the main cause associated with the excessive excretion of bicarbonate?
-body cannot keep up
-HHS
-DKA
-Kussmaul breathing
in regard to metabolic acidosis, what can happen in the body when there is excessive production/intake of metabolic acids?
DKA
abnormal cellular metabolism caused by lack of insulin produced ketoacids faster than kidneys able to excrete
hypoventilation
accumulation of too much carbonic acid; usually a result of not getting enough oxygen to the alveoli
1. acute
2. chronic
what are the 2 types of alveolar hypoventilation (respiratory acidosis)?
-pneumonia
-asthma exacerbation
-ARDS
what are some types of acute alveolar hypoventilation?
-COPD
what are some types of chronic alveolar hypoventilation?
look at compensation in ABGs
how do we determine if someone has acute or chronic alveolar hypoventilation?
the alveoli cannot perform gas exchange = holding onto CO2
what happens during acute alveolar hypoventilation?
-chronically retaining CO2
-body has to get used to ↑ CO2 levels
what happens during chronic alveolar hypoventilation?
greater than 45 (hypercapnia)
what is considered a high CO2 level (PaCO2)?
pH is too high or on high range of normal
what is characteristic of the pH in alkalosis?
-massive blood transfusions
-diuretic therapy
-holding onto too much bicarbonate
what are some situations in which we might see high levels of a base in one's body?
-prolonged vomiting
-nasogastric suctioning
-mineralocorticoid excess
what are some situations in which we might see too little metabolic acid in one's body?
a lot bicarbonate in the blood = alkalosis
what is the relationship between high amounts of bicarbonate in the blood and pH?
less than 35 (hypocapnia)
what is considered a low CO2 level (PaCO2)?
-hyperventilation in times of acute hypoxia or pain
what happens during respiratory alkalosis?
in times of acute anxiety or emotional distress
when can respiratory alkalosis occur?
-hypoventilation (cause of problem)
-cardiac dysrhythmias
-↓ LOC, confusion
what are the common clinical findings associated with too much carbonic acid (respiratory acidosis)?
-pH: ↓
-PaCO2: ↑
-HCO3: may be ↑ if compensation is present
what are the common blood gas findings associated with too much carbonic acid (respiratory acidosis)?
-hyperventilation (compensatory mechanism)
-abdominal pain
-nausea/vomiting
-cardiac dysrhythmias
-↓ LOC, confusion
what are the common clinical findings associated with too much metabolic acid (metabolic acidosis)?