Organic Chem Reactions to Know
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
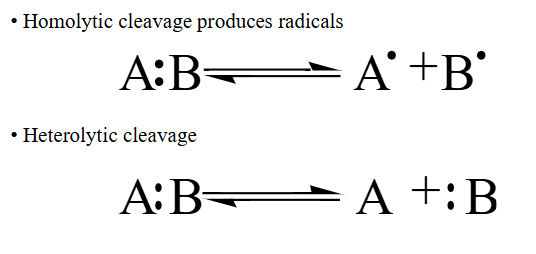
Homolytic vs heterolytic cleavage of bonds
Homolytic: produces two radical species
Heterolytic: Both electrons go to one species → can produce carbanions, carbocations, hydrides and protons
What are the 7 types of reactions we need to know?
Nucleophilic substitution
Aldol condensation
Claisen condensation
Decarboxylation of B-Keto Acids
Rearrangements or Isomerization
Eliminations
Free Radical Reactions
In a nucleophilic sub, what is involved?
Good LG
Good nuc
Electrophile
Usually SN2
SN2 is called an __ mechanism, while SN1 is a __ mechanism
Associative, dissociative
Common nucleophiles have…
High e- density and are sometimes negatively charged
Give an example of some biological nucleophiles
Negatively charged oxygen
Negatively charged sulfhudryl (a deprotonated -SH group)
Carbanion
Amine
Hydroxide
Imidazole
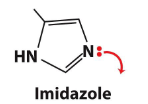
Give some examples of biological electrophiles
Carbonyl C
Phosphorus of phosphate group
Proton
Protonated imine group
Enzymes often create __ leaving groups on substrates
Better
What are two examples of good leaving groups?
Pyrophosphate or phosphate, H2O
How does hexokinase phosphorylate glucose?
There’s a general base as a side chain in the active site that deprotonates the -OH on C6, whose e- then attack the gamma P in ATP.
In glycolysis, we do __ aldol, in gluconeogenesis, we do __ aldol.
Reverse, forward
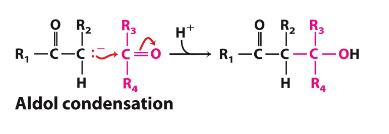
An aldol condensation turns a __ into a __
Ketone into an alcohol
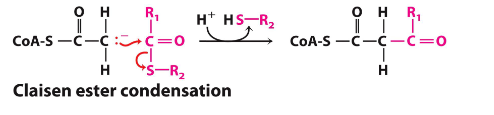
A claisen ester condensation turns an __ into a __
ester into a ketone
Name two significant properties of carbonyls. Similar to carbonyls in this regard are…
The O is very e- withdrawing
They can stabilize adjacent carbanions via resonance
Imines!
List the steps of an aldol condensation reaction
Carbanion formation
Nucleophilic attack
Protonation
What is a biological example of an enzyme that utilizes aldol condensation. What about claisen condensation?
Aldol: Aldolase
Claisen: Thiolase
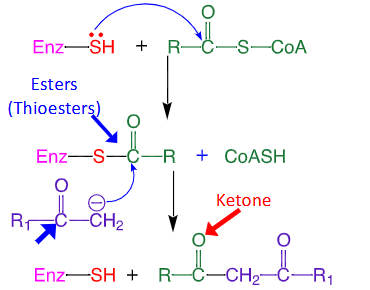
Are thioesters more or less stable than esters?
Less! Bad 3p and 2p S/C overlap
__ is an __ inhibitor of Thiolase, because you can’t hydrolyze off the covalent attachment between the cysteine in thiolase and the C in __.
Iodoacetate is an irreversible inhibitor of thiolase, because it irreversibly covalently attaches to a cysteine residue in the active site
Decarboxylation reactions yield…
Carbon dioxide
What’s an example of an enzyme that catalyzes an isomerization?
Phosphohexose isomerase
Triose phosphate isomerase (enediol intermediate)
What’s an example of an enzyme that catalyzes an elimination?
Enolase (involves metal ion catalysis)
Free radical reactions are often used to…
Break C-C bonds