Topic 2: Chemical Bonding
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
Primary bonding
Strong interactions
eg. Covalent, ionic, and metallic bonding.
Secondary bonding
Weak interactions between atoms and molecules
eg. Dispersion forces and dipole-dipole interactions.
Covalent bonding
Valence electrons are shared between nuclei to form a bond (2 electrons).
Molecules are held together by covalent bonds: This bond forms stable molecules!
When bonding occurs the energy of the molecule is lower than that of the separate atoms.
Lewis structure
A simple bonding model involving the arrangement of valence electrons in a molecule.
The octet rule
In most stable compounds, each atom has an octet (4 pairs) of valence electrons.
There is an intrinsic stability associated with having 8 valence electrons
ie. inertness of the noble gases (group 18).
Ne 1s2 2s22p6 = 8 valence electrons (blue)
Ar 1s2 2s22p6 3s23p6 = 8 valence electrons
Drawing lewis structure
ICl-4 electronic config
36 valence e-

Drawing lewis structure steps
Find the total valence e- in a moelcule
A single covalent bond contains 2 e-
- some atoms can have multiple bonds (eg. C,N,O,P,S)Apply the octet rule for every atom except H (bc 2 is enough)
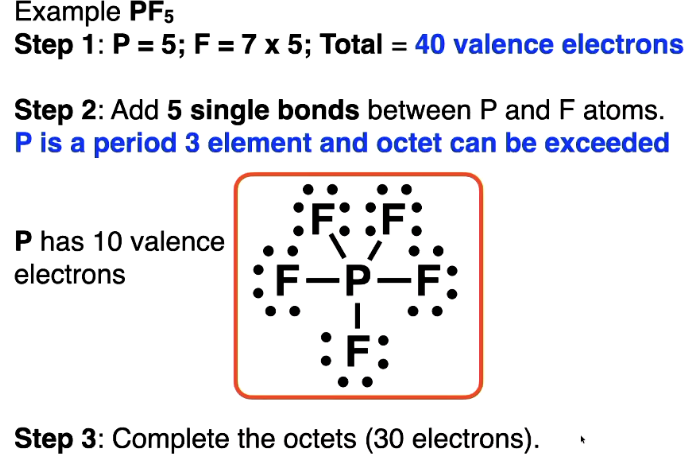
- use lone pairs and multiple bonds (double, triple)Place left over e- on atoms with available d orbitals (3rd period + beyond)
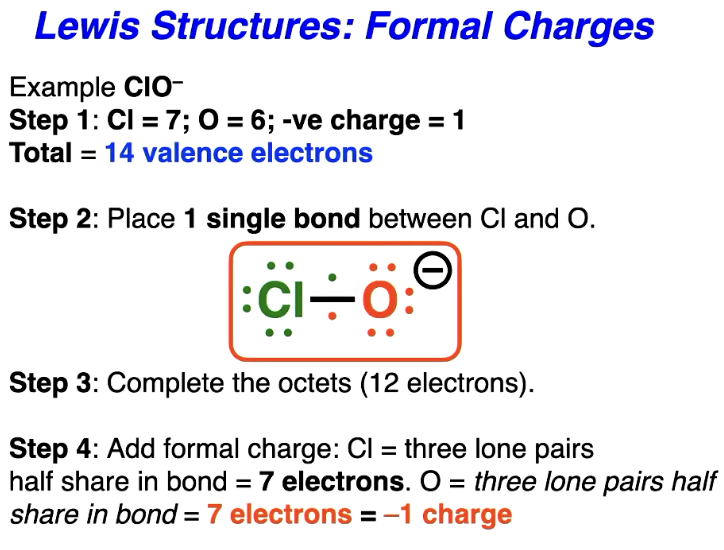
Check atoms for ‘formal’ charges

CF4 lewis structure
central atom must be the least electronegative

Lewis structures with molecules with formal charge
Exceptions to the octet rule - lewis diagrams
Exceptions to the octet rule - neon gases
Phosphorus, sulfur etc. will do this

Multiple bonds lewis diagram

oxygens in O3 charges
when oxygen forms a single bond it usually has a single negative charge (starts with 6 and shares 1 → so one more than when started so so neg charge)
The other oxygen has a lone pair for 2 and then shares 3 in the bond so 5 e- → therefore one less than when it started so +1
Other no charge bc 6 e- as when it began

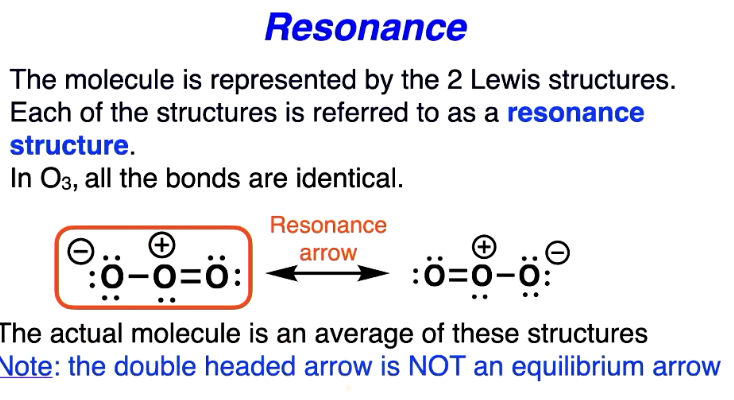
Resonance
The molecule is actually resonating between the two forms either with double bond on the left or right
same molecule on each side but rotated
neither of the structures depicted are exactly right
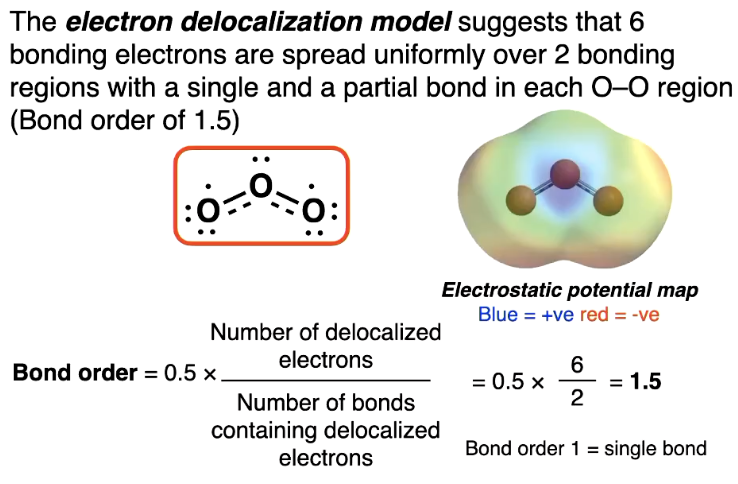
Electron delocalisation model
the diagram here is weird so if asked for lewis structure draw the other one
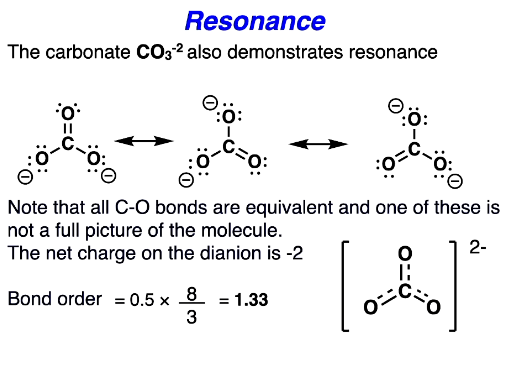
More complex resonance
number of bonds isnt 4 in the eq. its the ‘connectivities’
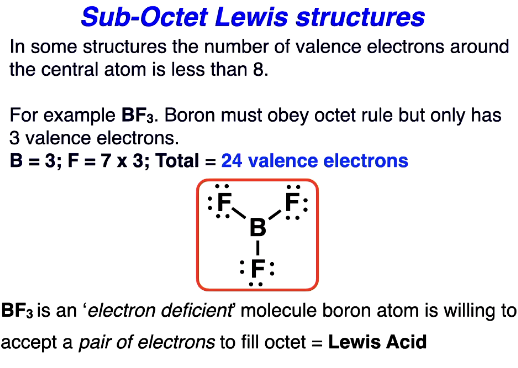
Sub-octet lewis structure - electron deficient molecule
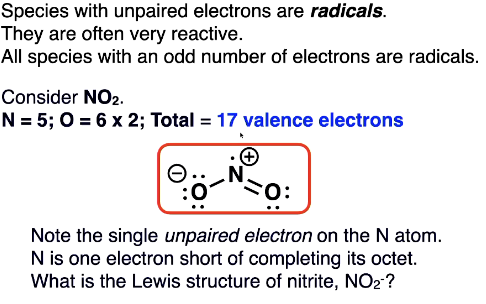
Sub-octet lewis structures: radicals
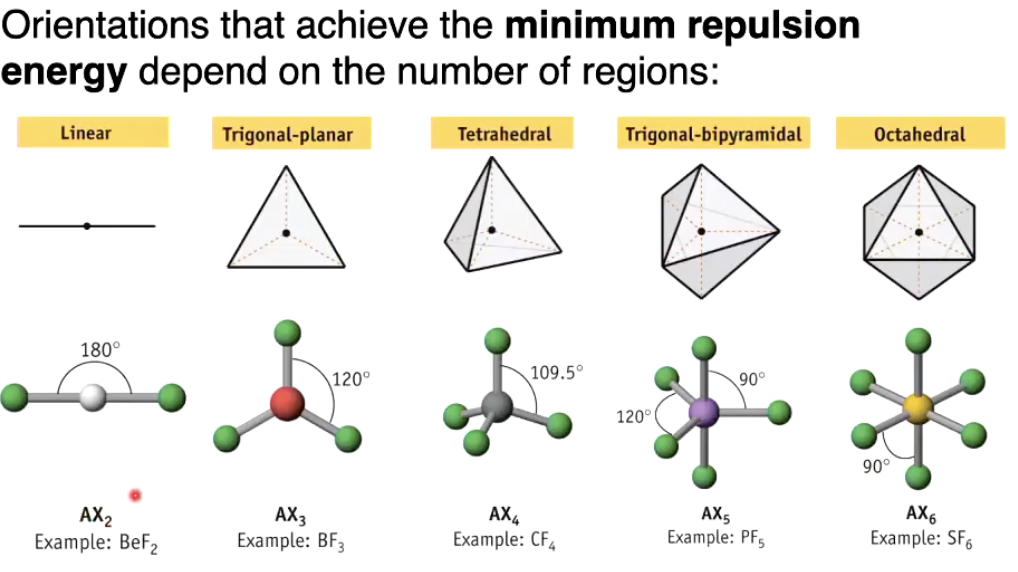
VSEPR Model - spatial arrangement of molecules
Valence electron pairs will arrange themselves to be as far apart as possible
bc like charges repel
Predicting the VSEPR structure

The different spatial arrangements we need to know
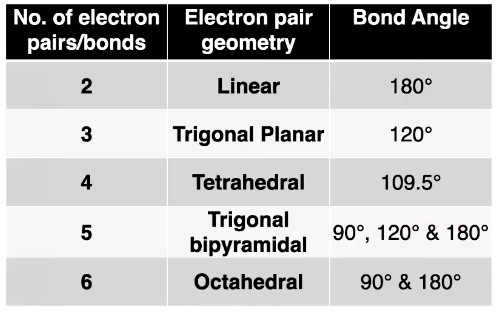
Number of e- pairs/bonds, geometrical shape + bond angles of all 5 types
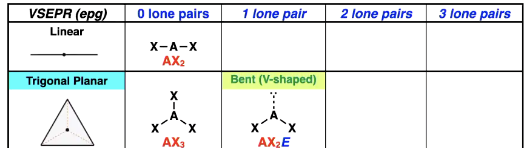
Are electron pair geometry and shape the same
No necessarily, can be but can also be different based on lone pairs
electron pair geometry is for instance tetrahedral
but if 1 lone pair then the shape is trigonal pyramid but still same electron pair geometry
Trigonal planar possible shapes
E stands for electron pair
Tetrahedral shape options
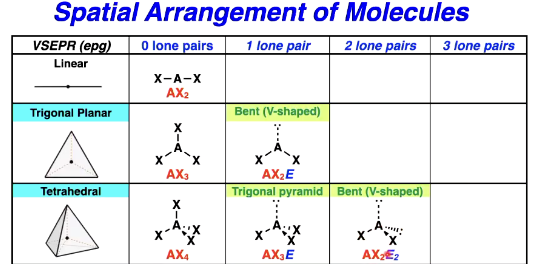
How to tell the difference between bent trigonal planar and tetrahedral
The angle between the Xs is slightly different as per the angles from before
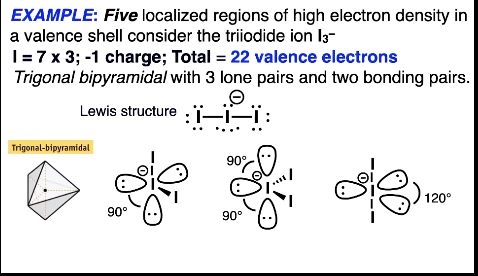
Trigonal bipyramidal shape options
GOAL: get the biggest angle possible between the electron pairs
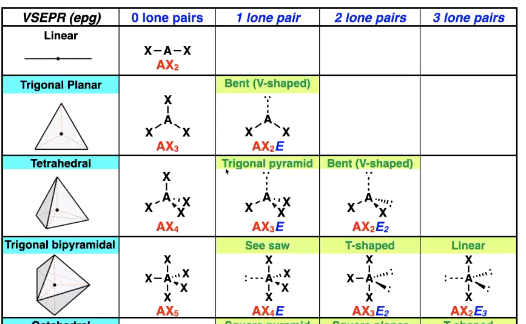
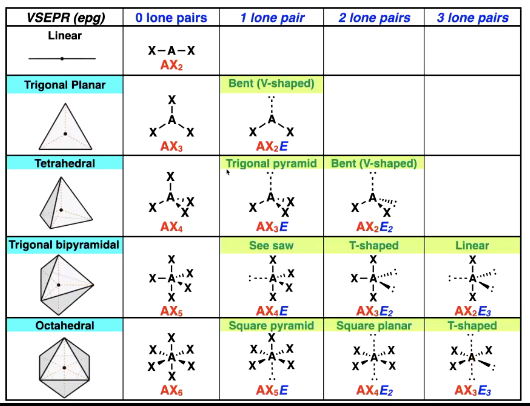
Octrahedral shape options
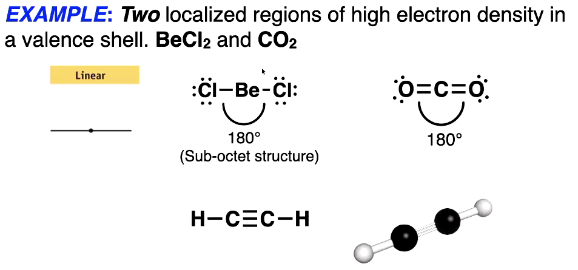
2 localisesd regions of high electron density in a valence shell
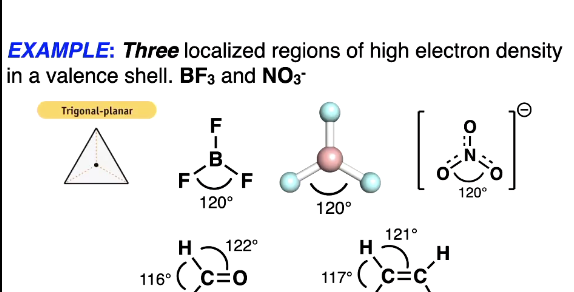
Three localised regions of high electron density
some are perfectly same angle if the same atoms but if diff atoms then angles may be slightly diff like the ketone
WHY: Still trigonal planar, The oxygen is bigger than the 2 hydrogens so the hydrogens get pushed together
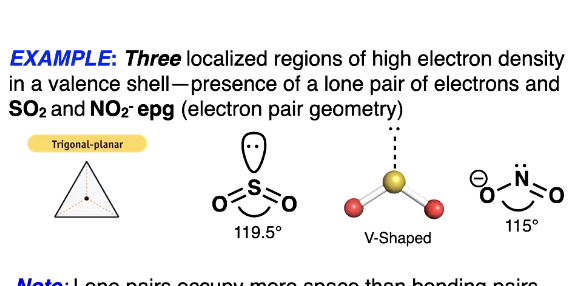
3 localised regions but we add a lone pair
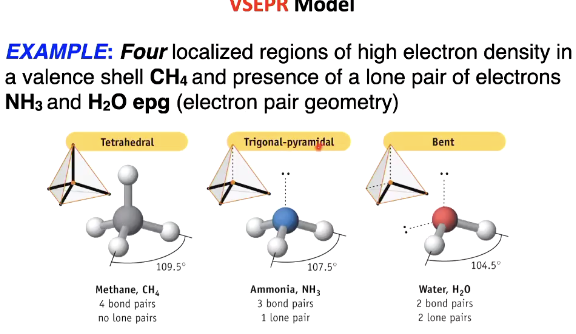
Four localised regions of high e- density and diff lone pairs
Slightly different angles
Five localised regions of high e- density and diff lone pairs