Biochem exam 3
1/123
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
124 Terms
How are liposomes used in drug delivery?
Carriers in drug delivery (COVID mRNA vaccine packaged into liposomes)
What type of molecules can easily cross the membrane?
small, non-polar molecules (Gases like O2)
What travels faster through the membrane, small polar molecules (H2O, urea) or larger non-polar molecules (Trp)?
Small polar molecules! Larger non-polar molecules are slow due to size
What molecules have difficulty crossing the membrane?
Ions and charged molecules
How do polar molecules and ions cross the membrane?
Membrane proteins
Different cells have different % of proteins (20-75)
How can you infer what biochemistry is occurring inside of the cell?
The types of membrane proteins
The expression of membrane transporters
What is an example of a cell type with relatively low membrane protein content?
Schwann cells
Schwann cell membrane structure
lipid rich (serves as an insulator/fast transmission)
Low protein = flexibility (allows wrapping on the axon)
What is Multiple sclerosis?
an example of a demyelination disease, impairing myelin assembly or damaging existing myelin.
Integral membrane proteins
Embedded in the hydrocarbon membrane
Peripheral membrane proteins
Bound to the polar heads or exposed surfaces of an integral membrane protein
sometimes attached by hydrophobic moiety inserted in the membrane
Example of integral protein
Porin
Outside surface is hydrophobic and interacts with membrane interior
Inside surface is polar as it faces water.
Example of peripheral membrane protein
Prostaglandin H2 Synthase-1
How is Prostaglandin H2 Synthase-1 attached to the membrane
Mostly hydrophilic molecule has a small hydrophobic side chain that is embedded in the membrane
What does Prostaglandin H2 Synthase-1 do
Injury → Arachidonate → Goes through Cyclooxygenase → Prostaglandin G2 → Goes through peroxidase → Prostaglandin H2 → starts inflammation
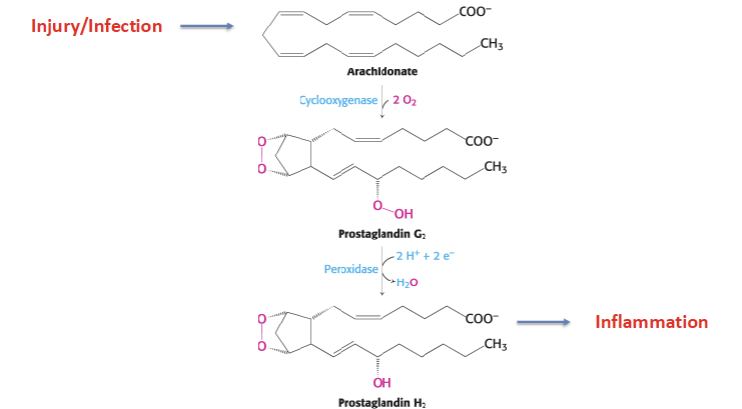
What is the significance of the hydrophobic channel of Prostaglandin H2 Synthase 1
The cyclooxygenase (COX) activity depends on the channel to connect the active site to the membrane interior.
How does Aspirin inhibit COX activity?
By transferring an acetyl group to a serine residue in the hydrophobic channel, obstructing it.
Where would the hydrophobic amino acids of an integral protein reside?
Inside of the transmembrane domain (The part that passes through the phospholipid bilayer)
Do Prokaryotes or Eukaryotes contain compartments bound by internal membranes?
Eukaryotes
What is receptor-mediated endocytosis?
Cells acquire molecules from their environment by separating and fusing parts of the membrane (makes vesticles)
What is exocytosis?
Fusion of internal membranes with plasma membrane to allow the release of molecules such as neurotransmitters
What does Clathrin do?
Helps internalize receptors bound to their cargo (used in endocytosis, coats vesicles)
What are the 4 ways molecules can pass through the membrane?
Diffusion, Facilitated diffusion, passive transport, Active transport
What are the 3 classes of pumps?
ATP-driven pumps, carriers, and channels
How do hydrophobic/lipophilic molecules pass through the membrane?
Diffusion, down their concentration gradient (don’t need proteins)
How do polar/ charged molecules pass through the membrane?
Passive transport of facilitated diffusion down their concentration gradient.
What is Active transport
Moving a molecule across the membrane against its concentration gradient
How do pumps pump molecules across the membrane
ATP hydrolysis
What is an example of a membrane pump?
Na+-K+ pump (ATPase) through Active transport to keep membrane potential
How much of the ATP we produce is expended by Na/K pumps (cells and nerve cells)
30%
up to 70% for nerve cells
What are the two conformations of a pump?
Ion binding site facing into the cell
Ion binding site facing out of the cell
Why is Digitalis purpurea useful/dangerous? (foxglove)
It has cardiotonic steroids that inhibit Na/K ATPase dephosphorylation
Can be used to treat congestive heart failure
What are two examples of cardiotonic steroids?
Digitoxigenin and Ouabain
What does Digitoxigenin do?
Decrease in Na+ gradient = slower removal of Ca+ from cell (increased Ca+ in cell) = enhanced contractile ability of the heart
What is Multidrug-resistance protein?
ATP-dependent pump that extrudes small molecules out of the cell.
What family of transporters in Multidrug-resistance protein from?
ABC transporters
What is the common domain of ABC transporters?
APT-binding cassette (ABC)
What is an example of an ABC protein?
Multidrug resistance protein in bacteria
ABC transporter mechanism
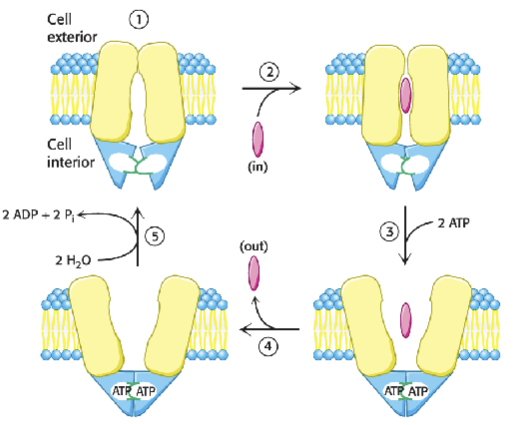
What do secondary transporters / cotransporters do?
They use energy of one gradient to power the formation of another
What are the 3 types of transporters?
Symporters, Antiporters, Uniporters
Symporters
Transport molecule B against its concentration gradient by coupling its movement with molecule A down its concentration gradient
Both molecules move the same direction
What is an example of a Symporter?
Sodium glucose symport
Antiporters
Molecule B exits while molecule A enters
What is an example of an Antiporter
Sodium calcium exchanger
Uniporters
Transports a single molecule either direction
what is an example of an Uniporter
glucose transporter
What do Ion channels do?
Allow rapid movement of ions across membranes down their concentration grandient
What does the potassium channel do?
Ion channel selectively and rapidly transports K+ across the membrane
Bigger ions = too big
Smaller ions = can’t interact
What does epinephrine bind to? What does it do?
Beta-Adrenergic receptor
Energy-store mobilization
What does Insulin bind to? What does it do?
Insulin receptor
Increased glucose uptake
What does Epidermal growth factor bind to? What does it do?
EGF receptor
Expression of growth-promoting genes
Signal cascade common components
Stimuli
Release primary message
Relay message through second messenger
Activate effector
Terminate signal cascade
Is the signal of second messenger equal to initial signal?
Second messenger is amplified
What are common second messengers?
cAMP, cGMP, Calcium ion, Inositol 1,4,5-triphosphate (IP3)
What type of receptor is a GPCR? (beta-adrenergic receptor)
Seven-transmembrane-helix
How does the binding of a ligand affect b-adrenergic receptor
It induces structural changes inside of the cell and initiate fight or flight response
How does epinephrine signal transduction cause changes in a cell?
Activates protein kinase A though G-protein pathway
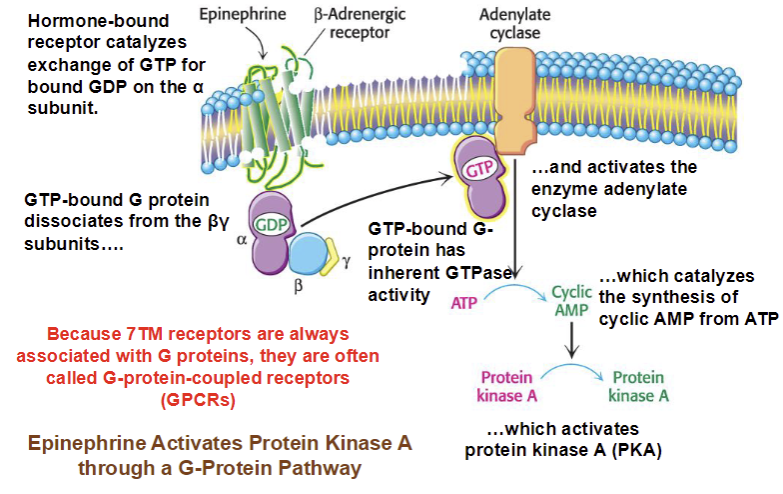
What does epinephrine cause in liver cells?
Epinephrine Promotes glucose release from liver cells
What changes does epinephrine cause in muscle cells?
Promotes calcium release within muscle cells
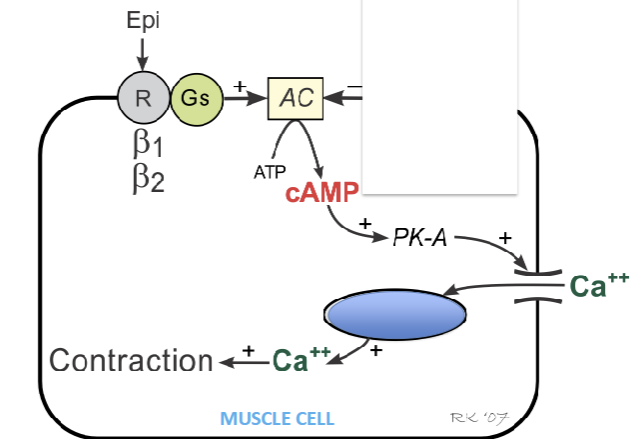
What is an example of a common Ca+ sensor
Calmodulin
How many binding sites does Calmodulin have
4, called EF hands
What does the Ca2+ calmodulin complex activate
Variety of biochemical targets
Plasma membrane Ca+ ATPase
How are Kinase transduction pathways turned off?
Protein phosphatase removes phosphates through dephosphorylation
How are G-protein transduction pathways turned off?
GTPase inactivates after the target has been activated
How are cAMP transduction pathways turned off?
Phosphodiesterases break down cAMP into AMP
What type of receptors do hormones such as insulin bind to?
Receptor Tyrosine Kinase
What happens after the ligand binds to a receptor tyrosine kinase
Sometimes dimers are formed
Are insulin receptors dimers?
Yes, even in the absence of insulin
What is the structure of the insulin receptor hormone binding site?
two alpha subinits outside the sell
What do alpha subunits activate?
2 beta subunits, activate tyrosine kinase to phosphorylate.
Simple insulin→glucose pathway
Insulin in insulin receptor → cross-phosphorylation → activates 2 kinase domains → phosphorylates IRSs to activate IRSs → IRS1 activates PI3 kinase → PIP2 becomes PIP3 → PIP3 activates PDK1 → PDK1 phosphorylates (activates) Akt → Akt stimulates GLUT4 allowing glucose to enter
What does epidermal growth factor activate?
Ras
Epidermal growth factor (EGF) → Ras pathway
EGF binds to EGFR → promotes dimerization/cross-phosphorylation of EGFR → Phosphotyrosine of EGFR attach to SH2+Grb2 → forms complex with Sos → promotes exchange of GDP to GTP → activates Ras
What does Ras do?
Promotes cell division
How does Ras promote cell division?
MAP kinase cascade to activate AP1 (start with 3 kinase, and cut one off each time)
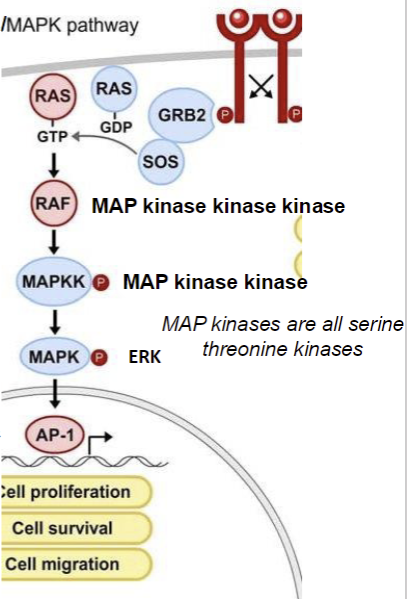
How is EGF terminated
Specific serine/threonine phosphatases
Tyrosine phosphatases
What does uncontrolled activity of Ras/MAP kinase lead to?
Cancer
What is Cetuximab?
Used to treat colorectal cancer
Competitive inhibitor of EGFR (growth factor receptor)
What are mutated genes that can cause cancer called?
Oncogenes
Tumor suppressors
What is an example of an oncogene?
Ras = protooncogene
Mutated Ras = oncogene
What is an example of a tumor supressor gene?
Mutated phosphates that terminate EGF
BRCA1
What is Cholera toxin
secreated by intestinal bacterium vibrio cholerae
Modifies the Gas protein = constantly active
What happens when g protein is constantly active
Continuous production of kinase A = altered ion channels and excessive loss of NaCl & water = diarrhea
How does nitric oxide impact cell signaling?
Nitric oxide diffuses across the membrane and widens blood vessels
What does NO activate?
Protein kinase G → lowered Ca+ levels → smooth muscle relaxation
What are fat-soluble vitamins?
Vitamin K, Vitamin A, Vitamin E, 1,25-Dihydreoxyvitamin D3
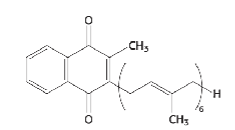
(be able to recognize, not draw)
Vitamin K
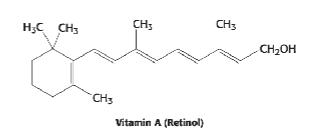
(be able to recognize, not draw)
Vitamin A
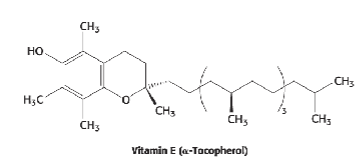
Be able to recognize, not draw
Vitamin E
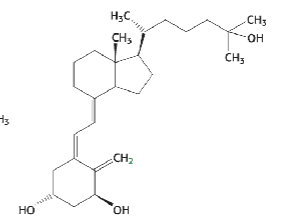
Be able to recognize, not draw
1,25-Dihydreoxyvitamin D3
Why do metabolic pathways have to be regulated?
To maintain homeostasis
How to maintain homeostasis?
Constant monitoring of nutrient levels
Adjusting metabolism to meet cell needs
What are the 3 regulatory strategies of homeostasis
Controlling the amounts of enzymes
Controlling catalytic activity
Controlling the accessibility of substrates
How is the amount of enzyme controlled?
rate of synthesis (long term)
rate of degradation (long term)
How is the catalytic activity of an enzyme controlled?
Allosteric regulation (fast)
Covalent modification (phosphorylation, methylation, ect)
How do hormones coordinate metabolic relations between tissues?
Regulating the reversible modification of key enzymes
Can synthesis and degradation of a reaction occur in different locations? example
Yes
Fatty acid oxidation = in mitochondrial matirx
Fatty acid synthesis = in cytoplasm
What is the function of vitamin A?
Supports vision, growth, reproduction
What happens with vitamin A deficiency?
Night blindness