ch 30- nuclear physics
0.0(0)
Studied by 0 peopleCard Sorting
1/7
There's no tags or description
Looks like no tags are added yet.
Last updated 5:33 PM on 4/29/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
1
New cards
Carbon-12 and Carbon-14 are
chemically identical, are isotopes, differ by number of neutrons
2
New cards
3
New cards
4
New cards
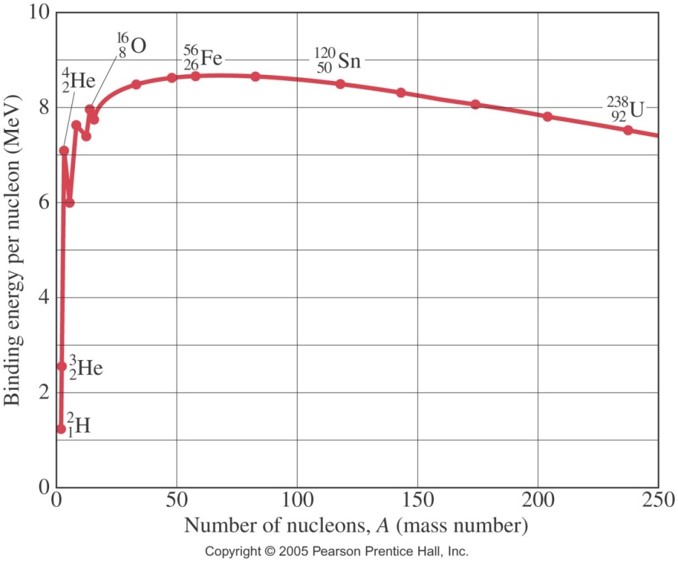
Based on the BE/A curve shown, the most tightly bound nuclei should be near:
Iron (Fe)-56
5
New cards
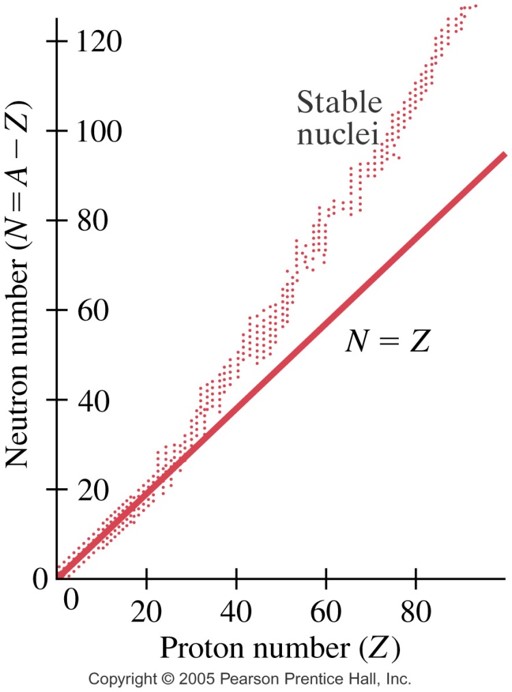
A nuclide that is to the left of the stability line has _________ to be a stable nucleus.
too many neutrons, or too few protons
6
New cards
alpha decay
occurs mostly in larger nuclei, because nucleus is too large
no matter how many nucleons you add its still too large
single particle (helium nucleus) is ejected from parent nucleus
7
New cards
If one sixteenth of a radioactive sample is present after 32 minutes, what is the half life?
8 minutes
1/16= (1/2)^4
T= time/ number of half-lives
T=32/4
8
New cards