biochem 2k26
1/207
Earn XP
Description and Tags
"term" is question, "definition" is answer
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
208 Terms
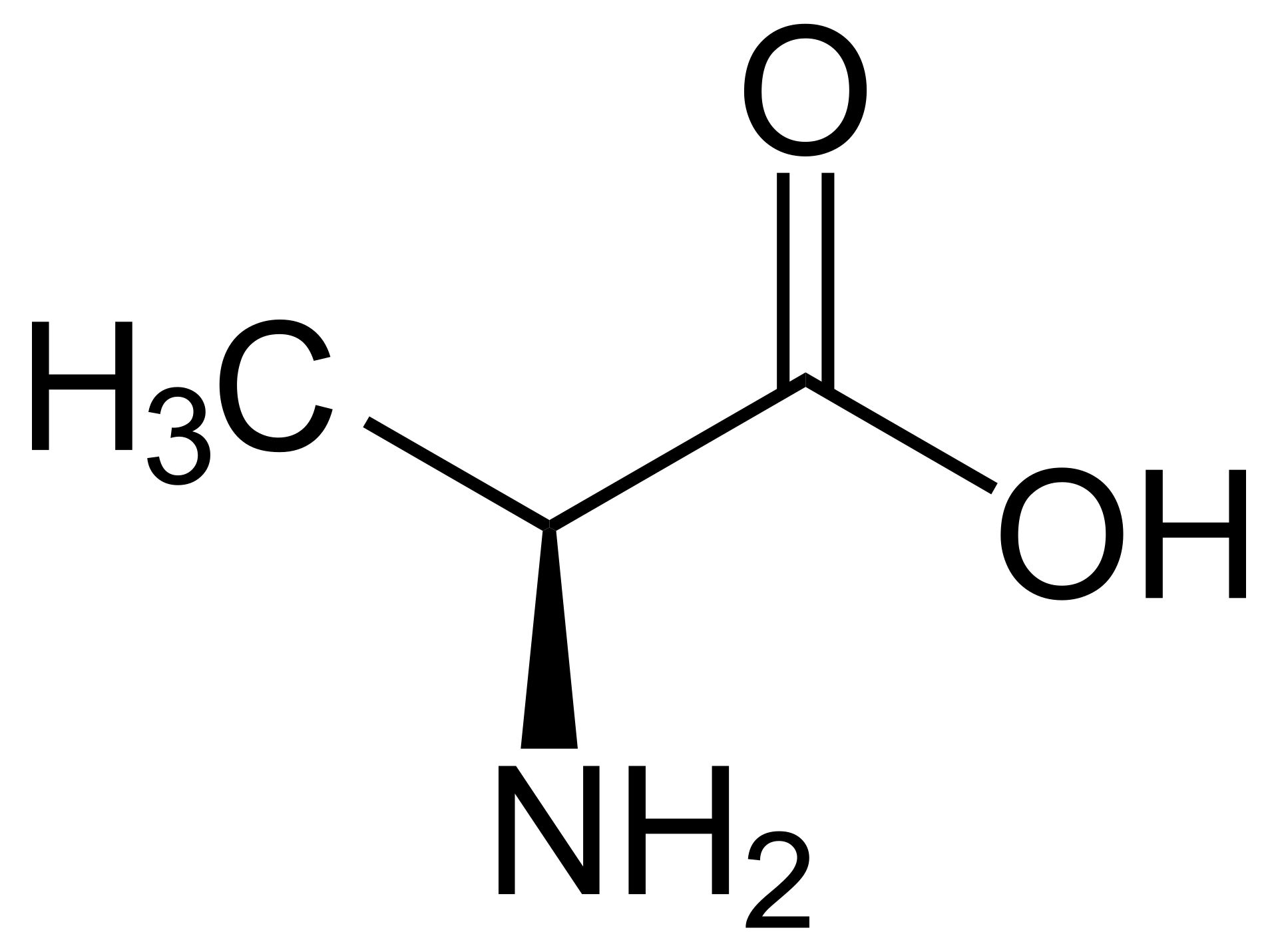
name this amino acid
alanine
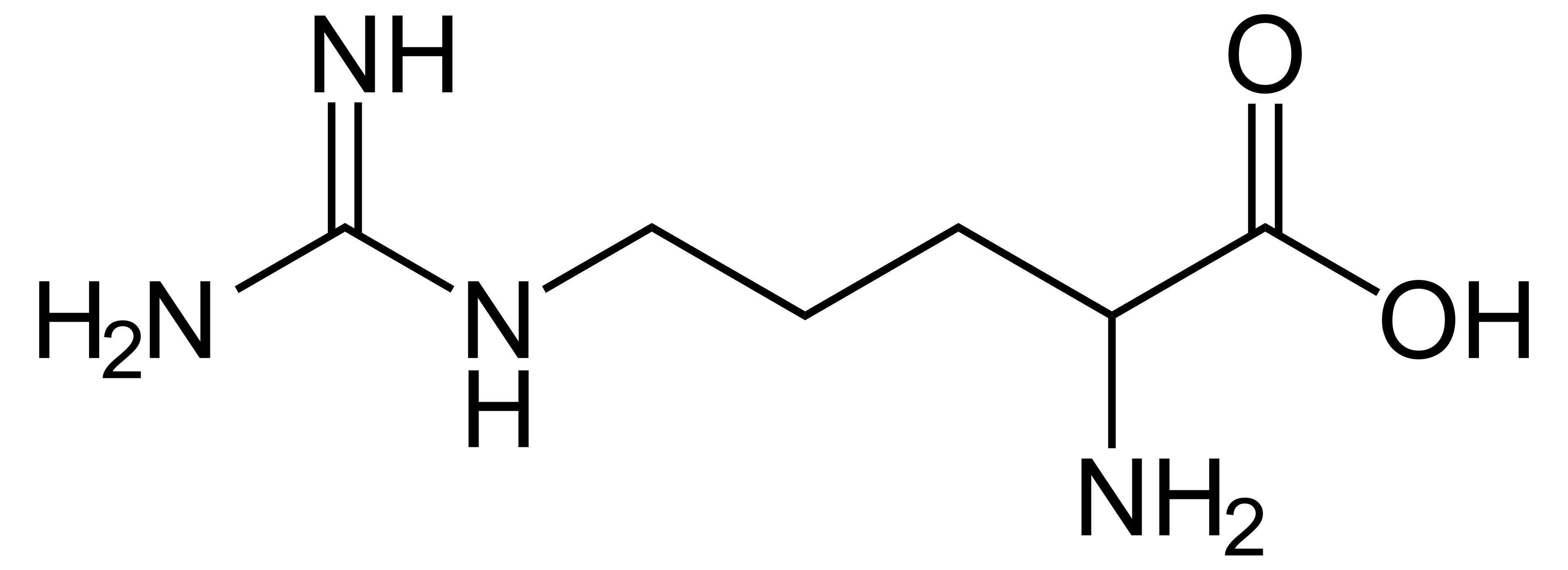
name this amino acid
arginine
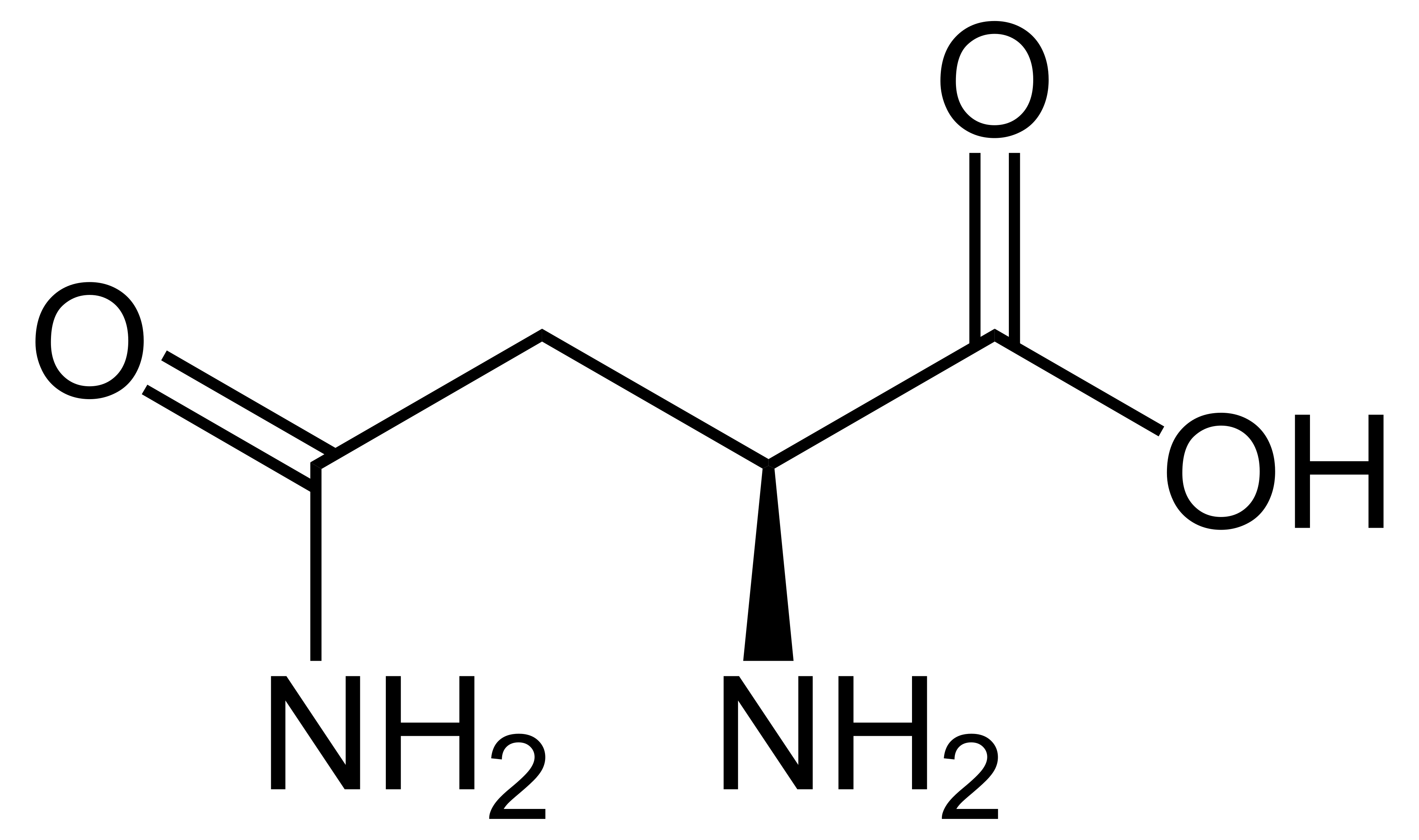
name this amino acid
asparagine
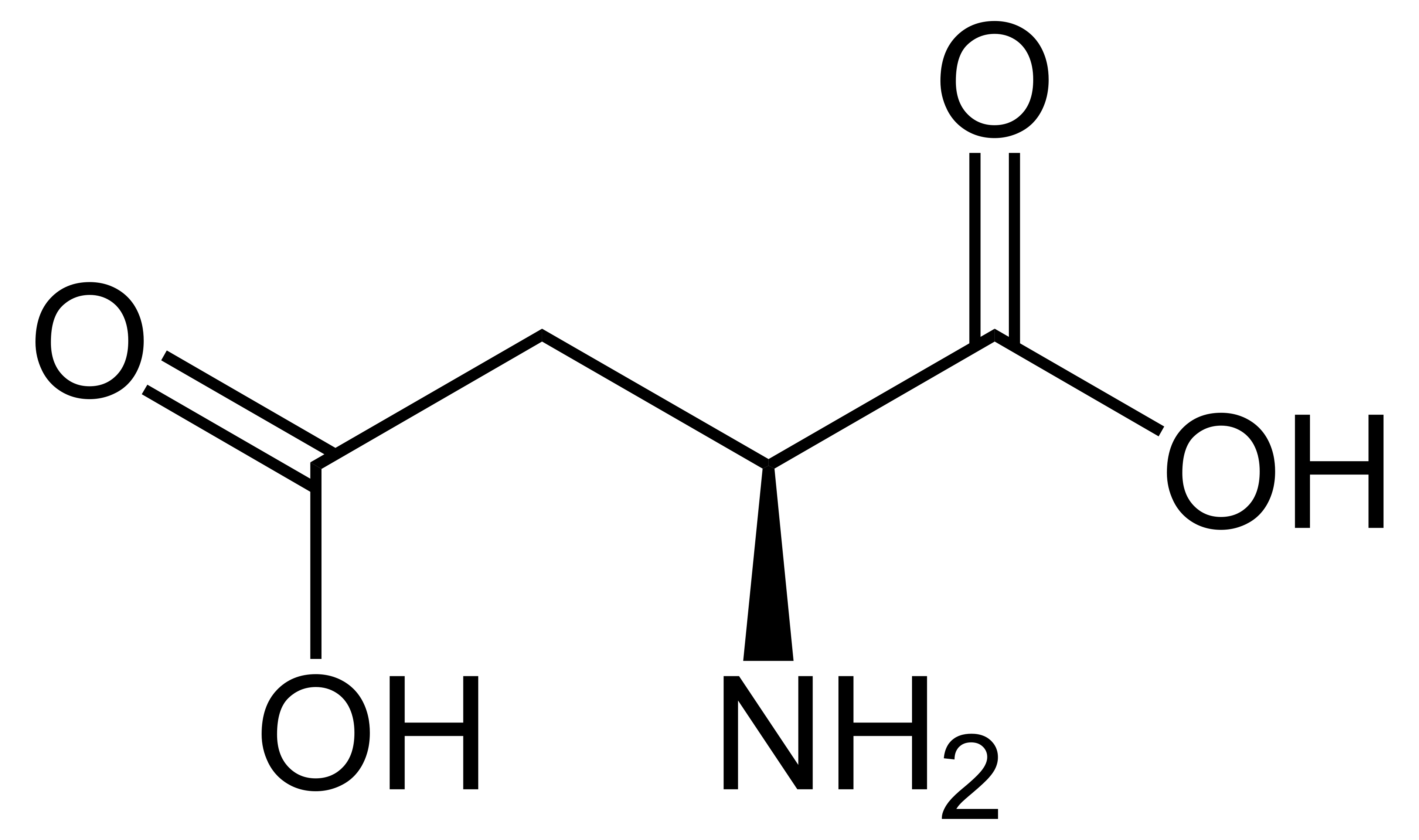
name this amino acid and its anionic form
aspartatic acid, aspartate
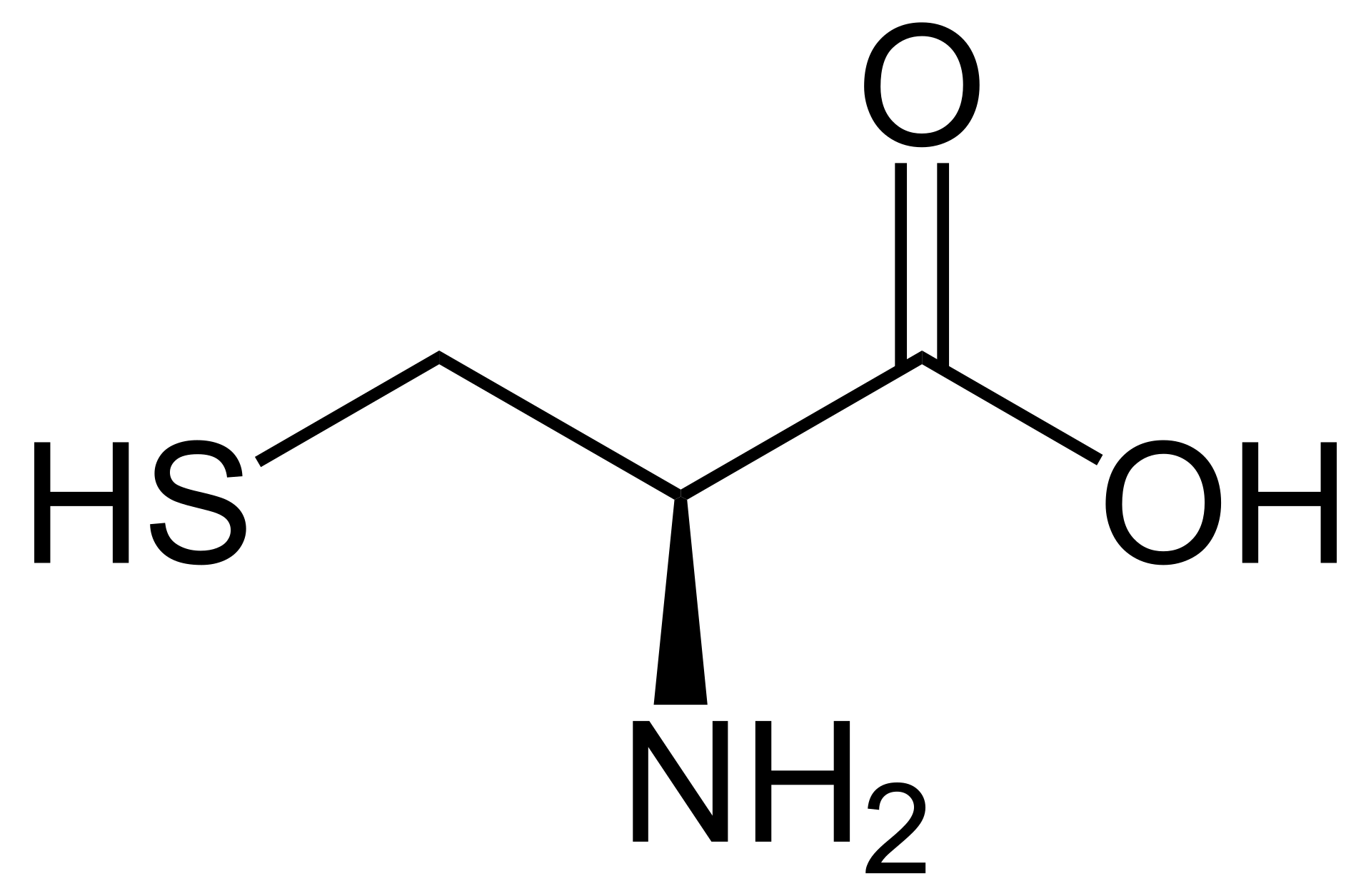
name this amino acid
cysteine
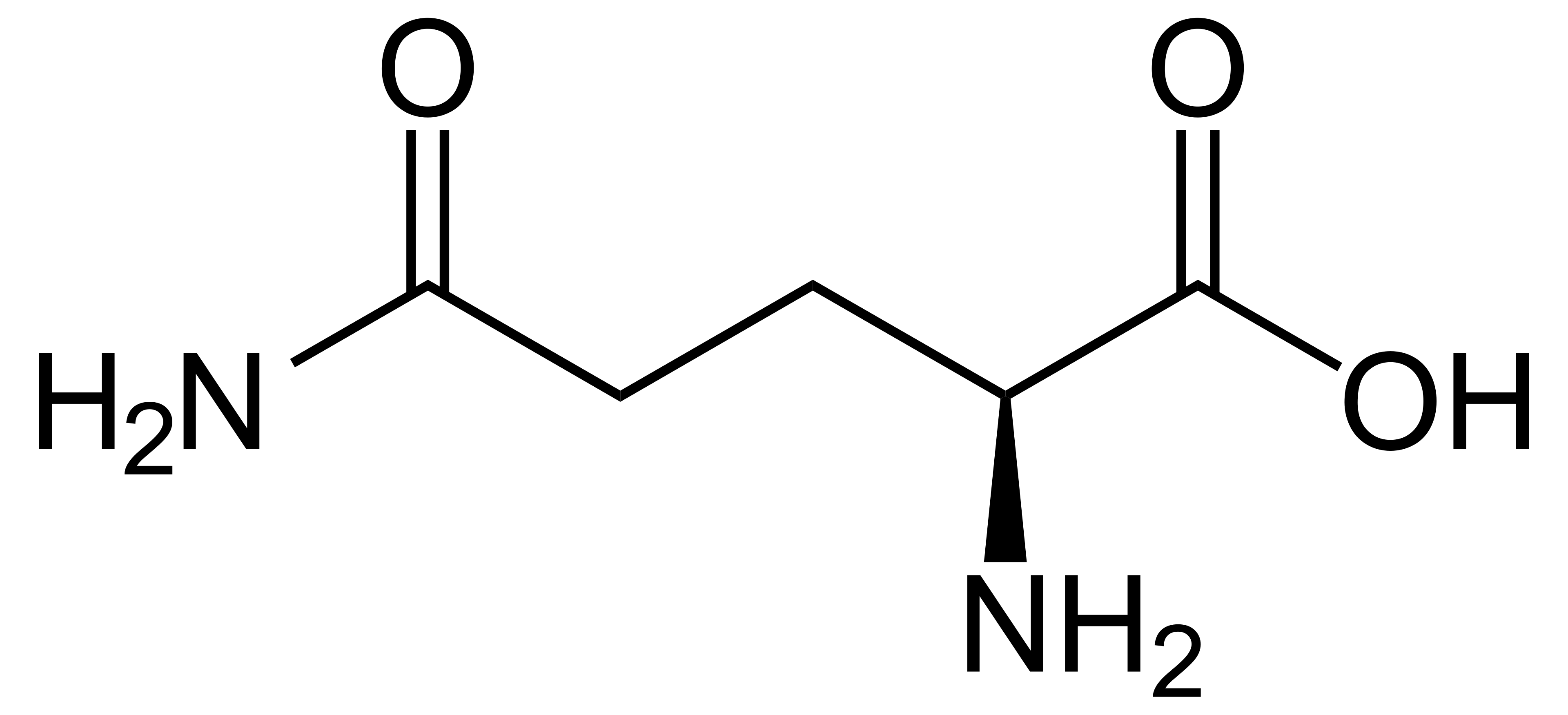
name this amino acid
glutamine
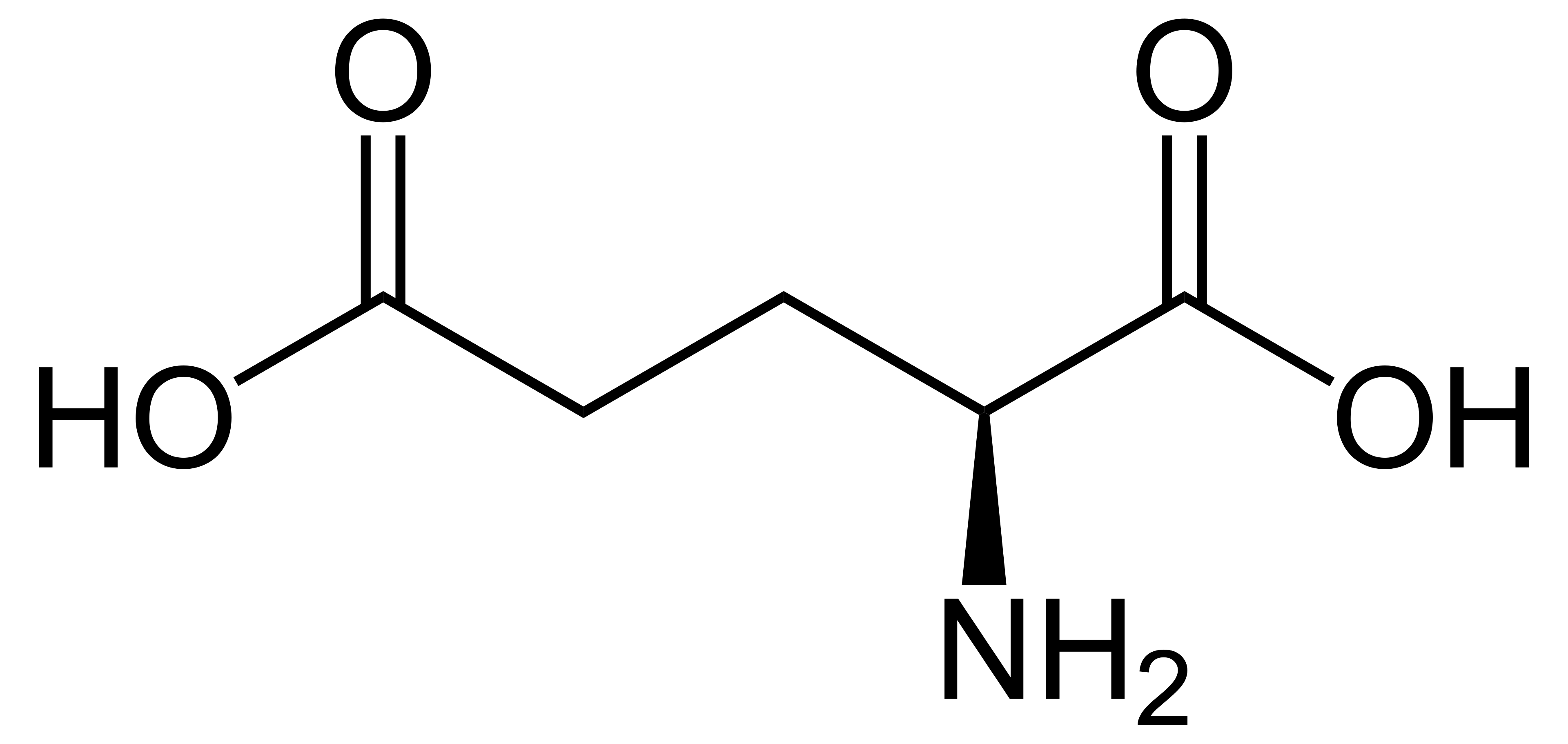
name this amino acid and its anionic form
glutamic acid, glutamate
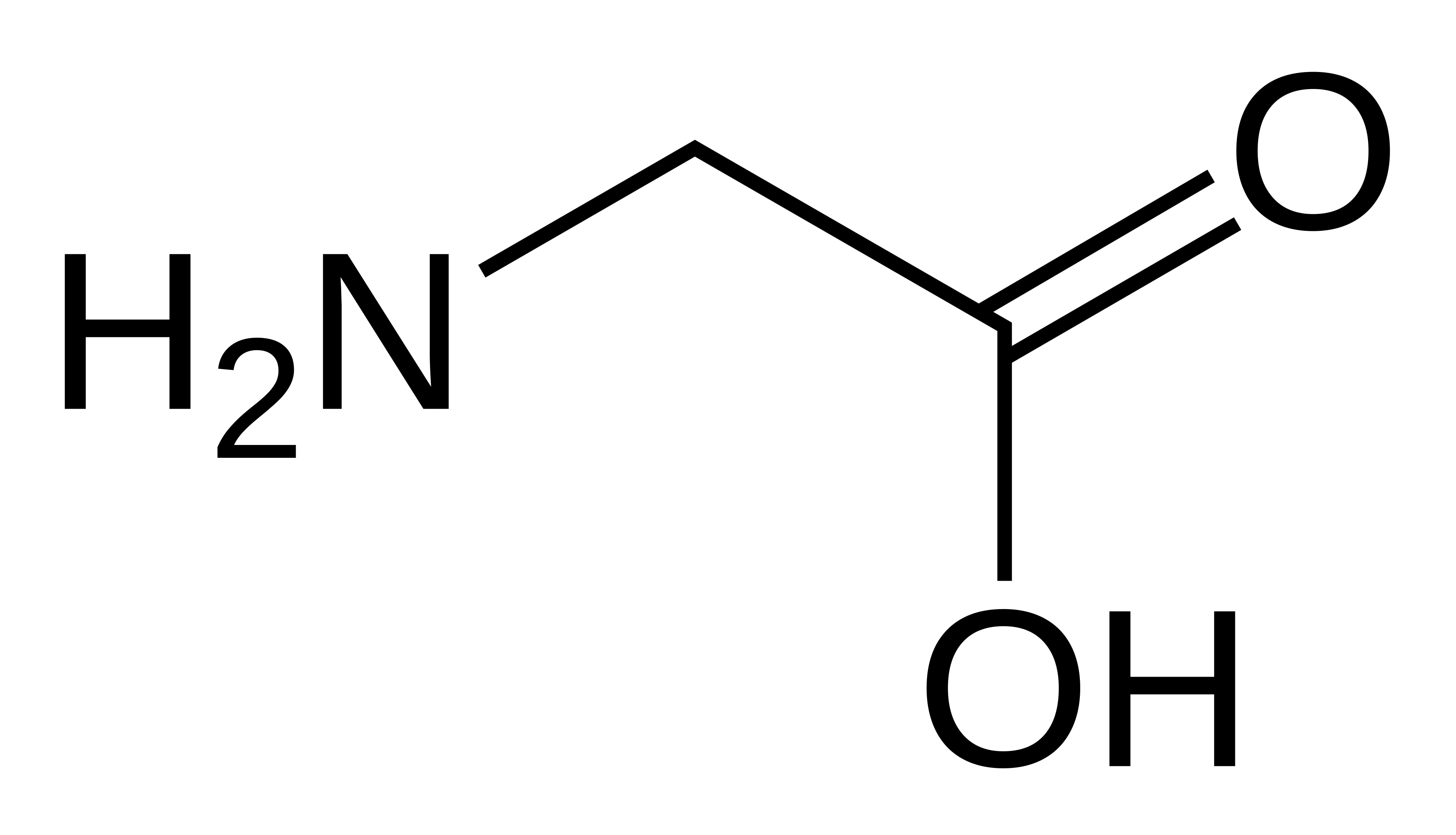
name this amino acid
glycine
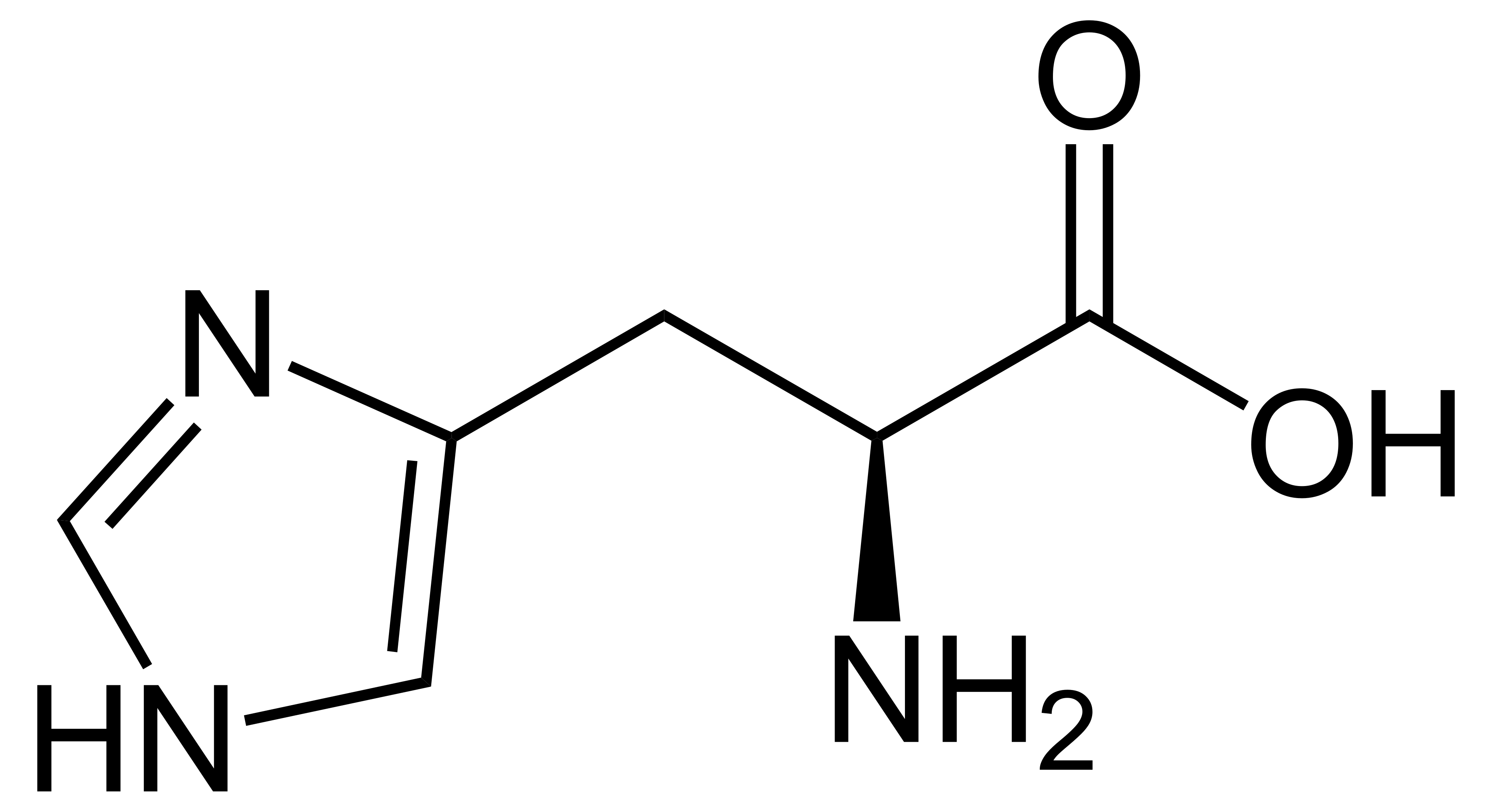
name this amino acid
histidine
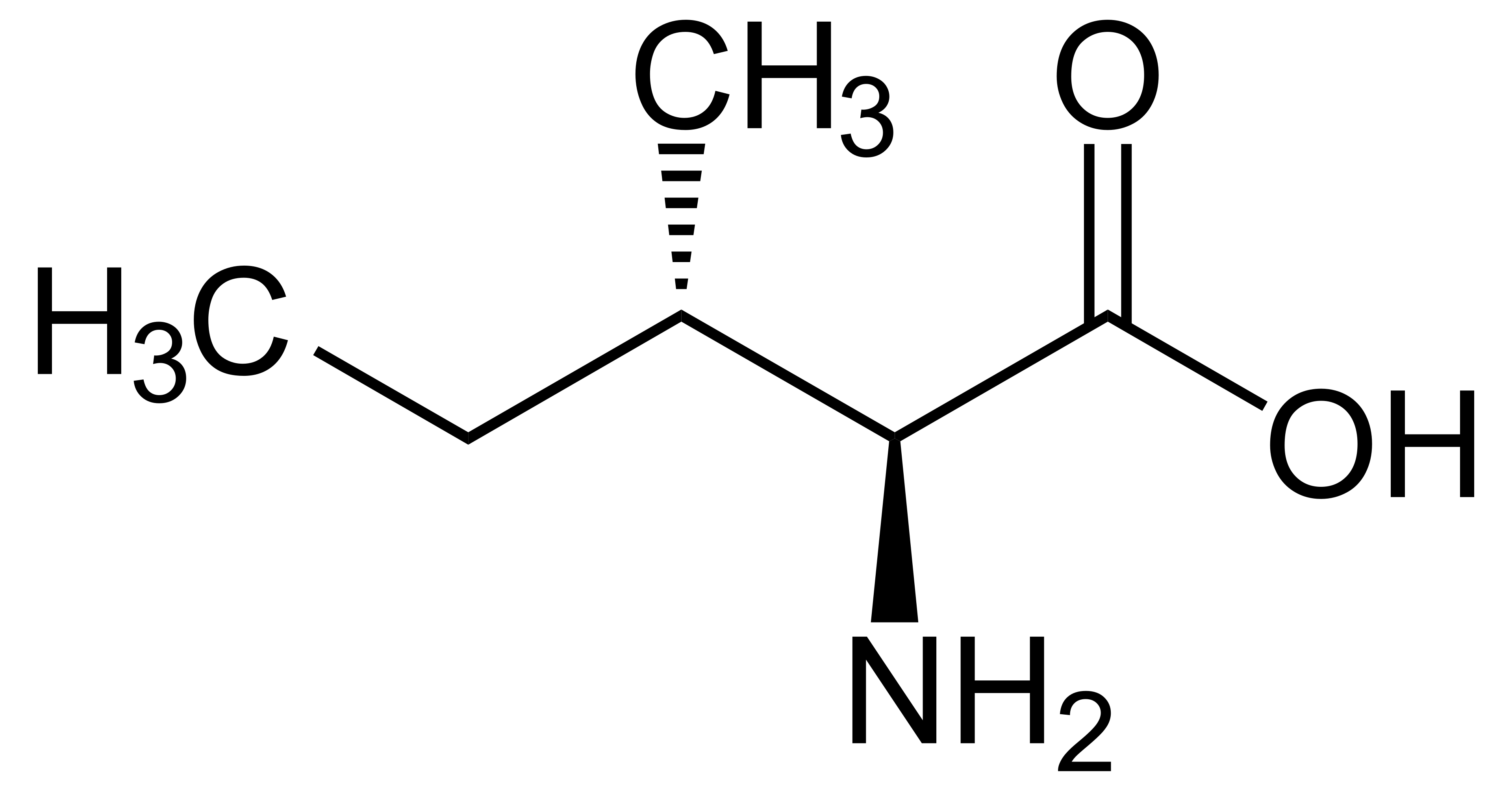
name this amino acid
isoleucine
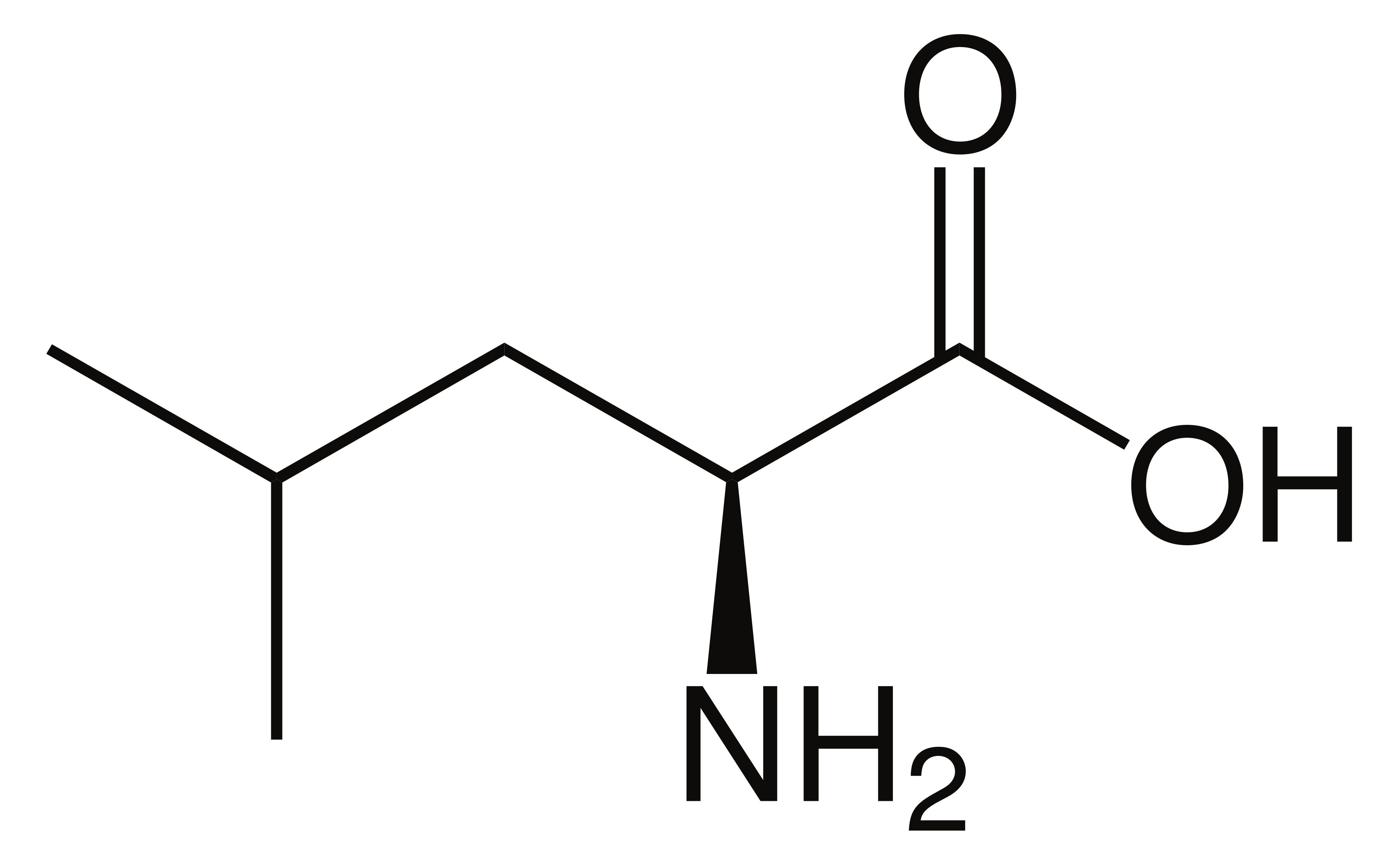
name this amino acid
leucine
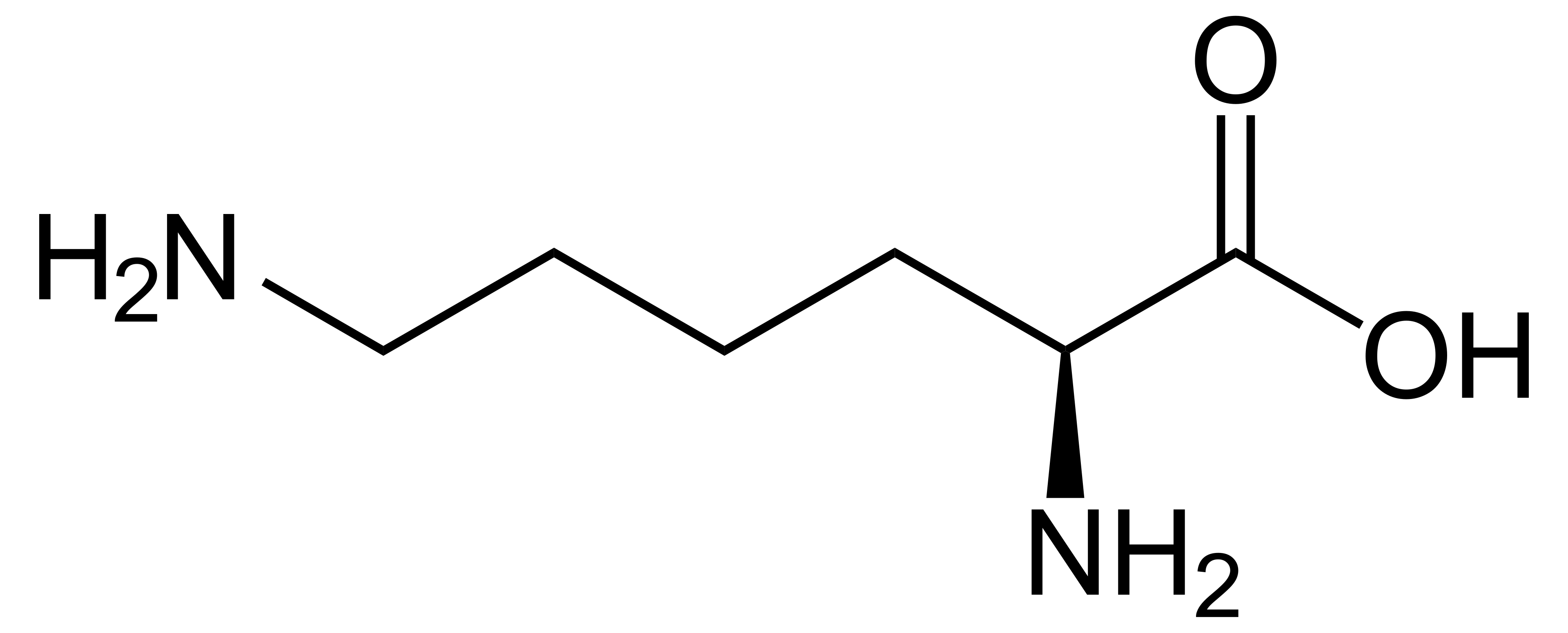
name this amino acid
lysine
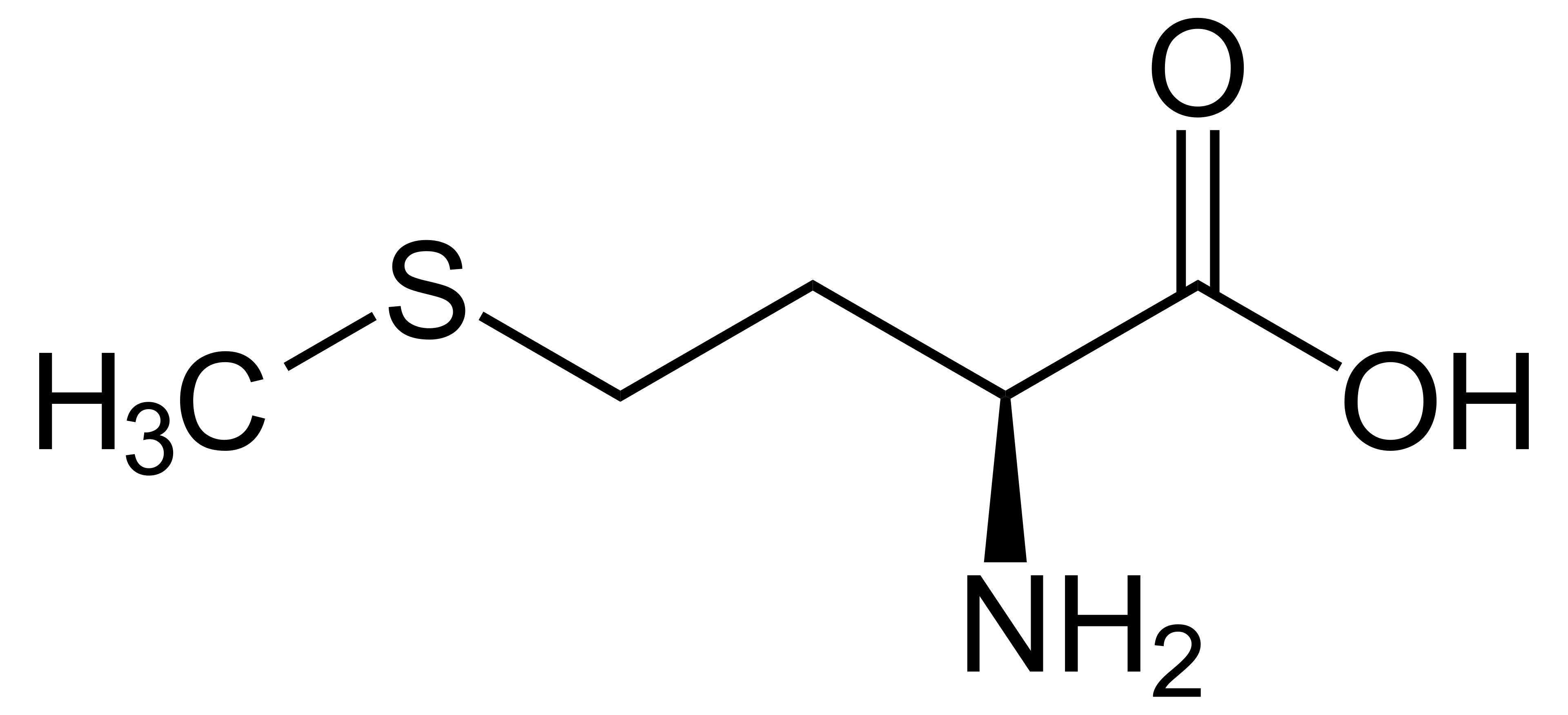
name this amino acid
methionine
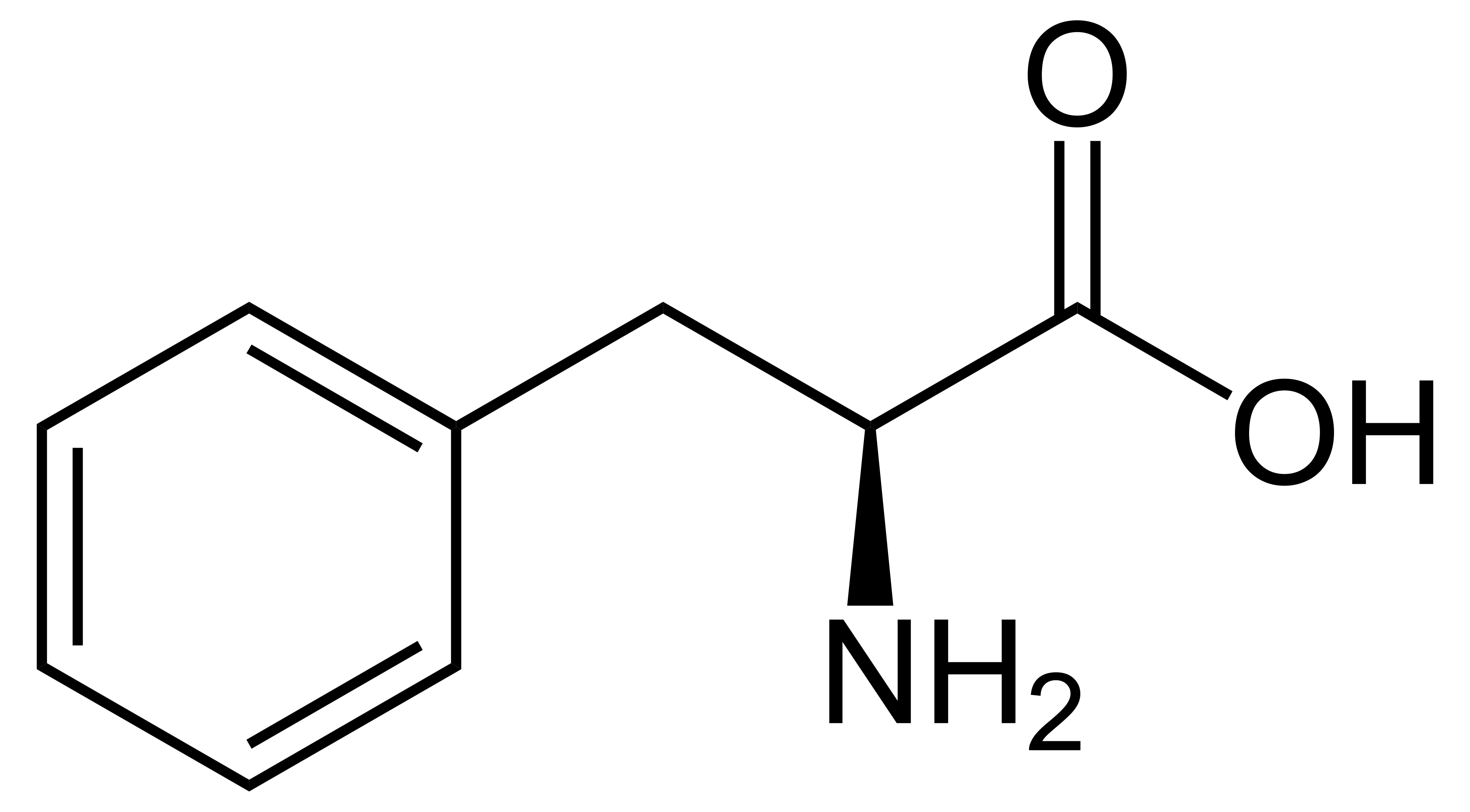
name this amino acid
phenylalanine
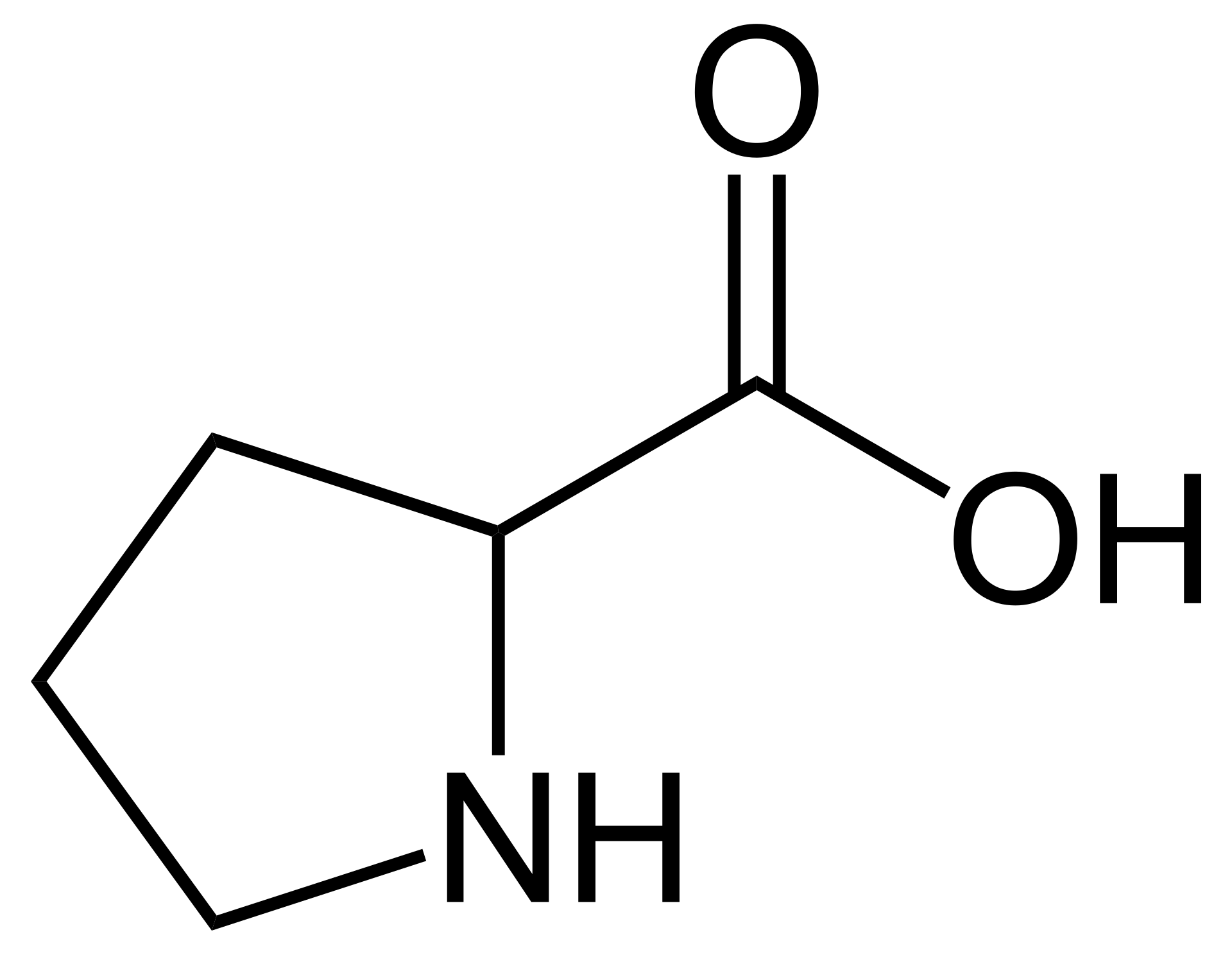
name this amino acid
proline
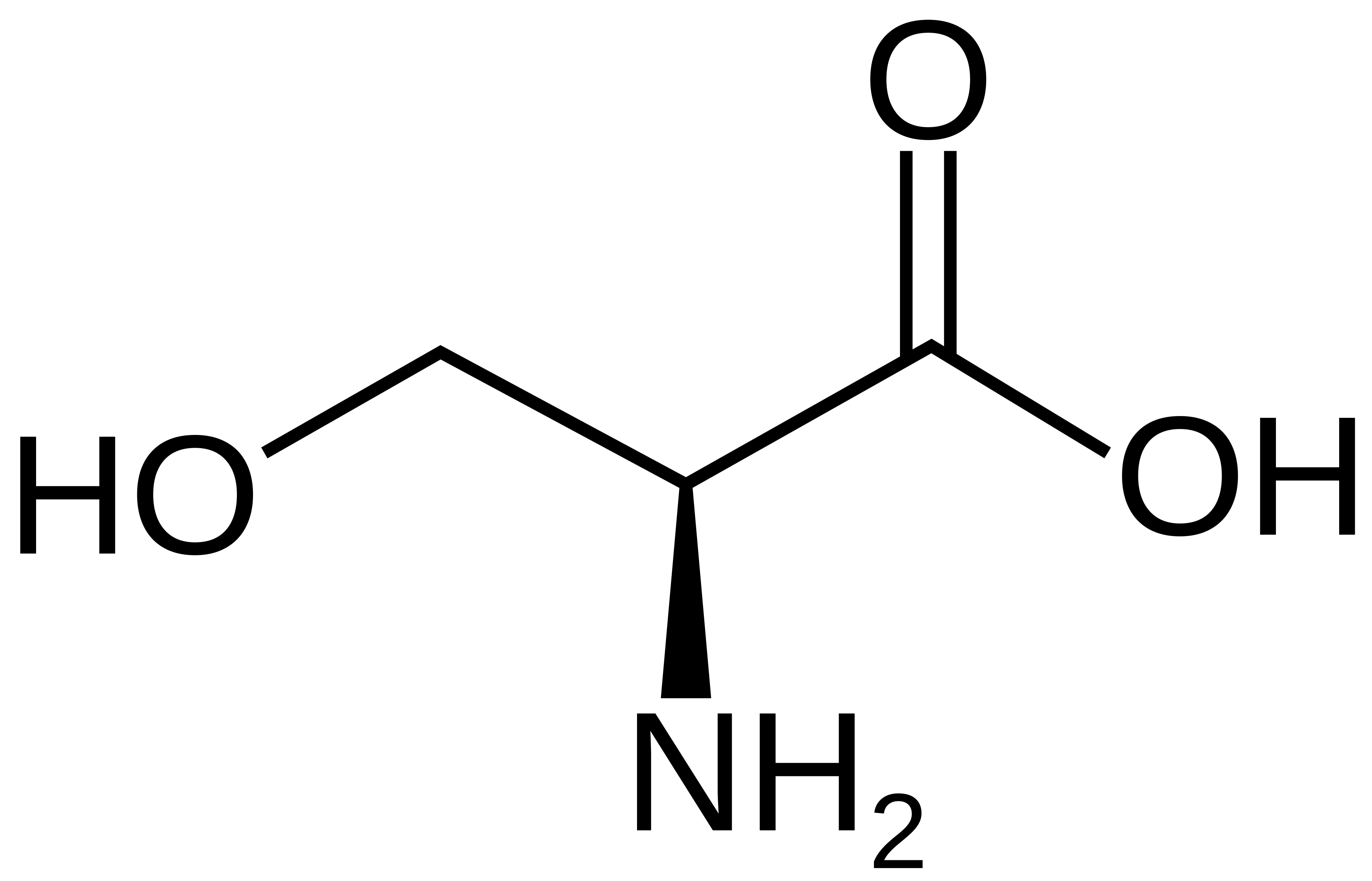
name this amino acid
serine
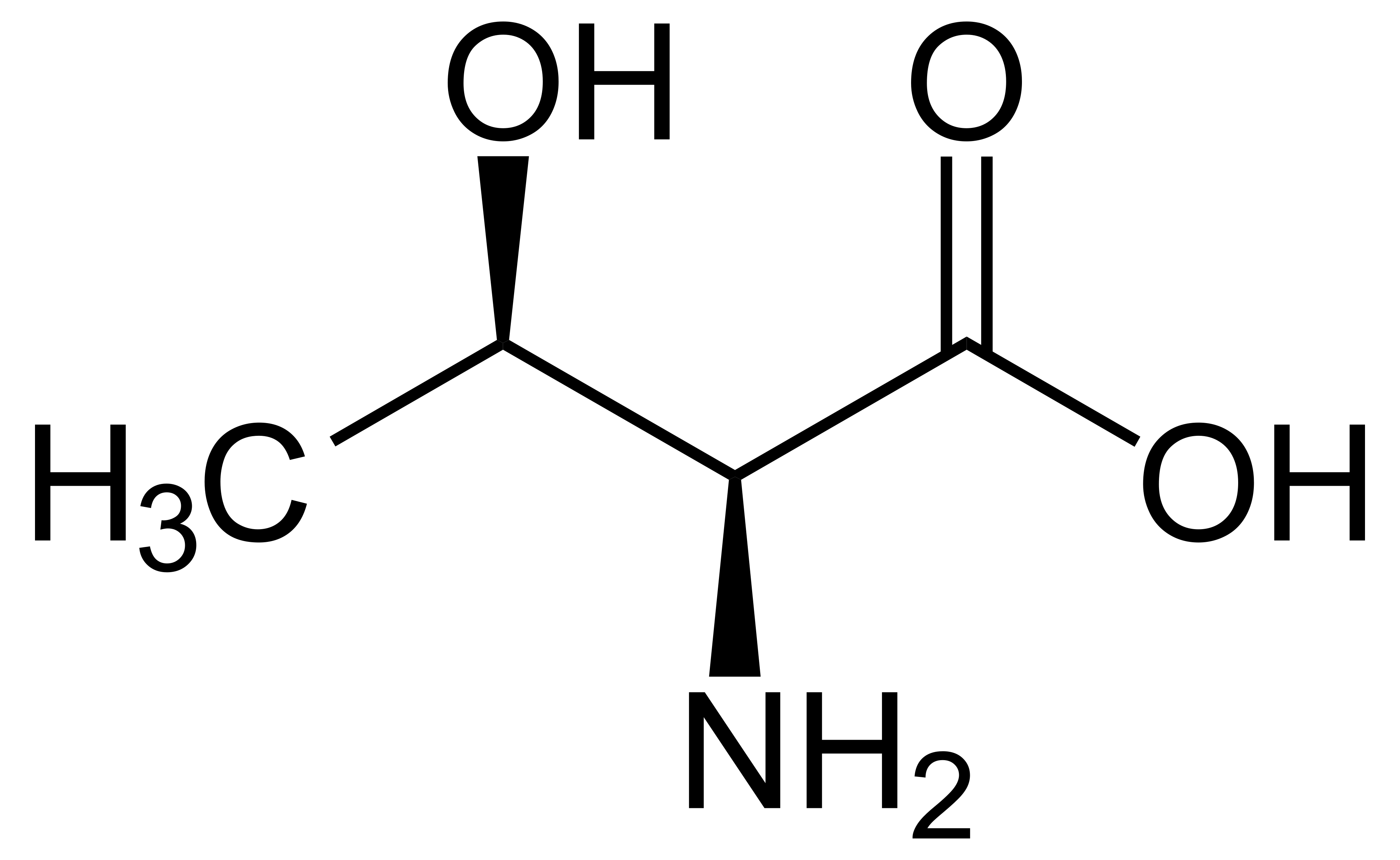
name this amino acid
threonine
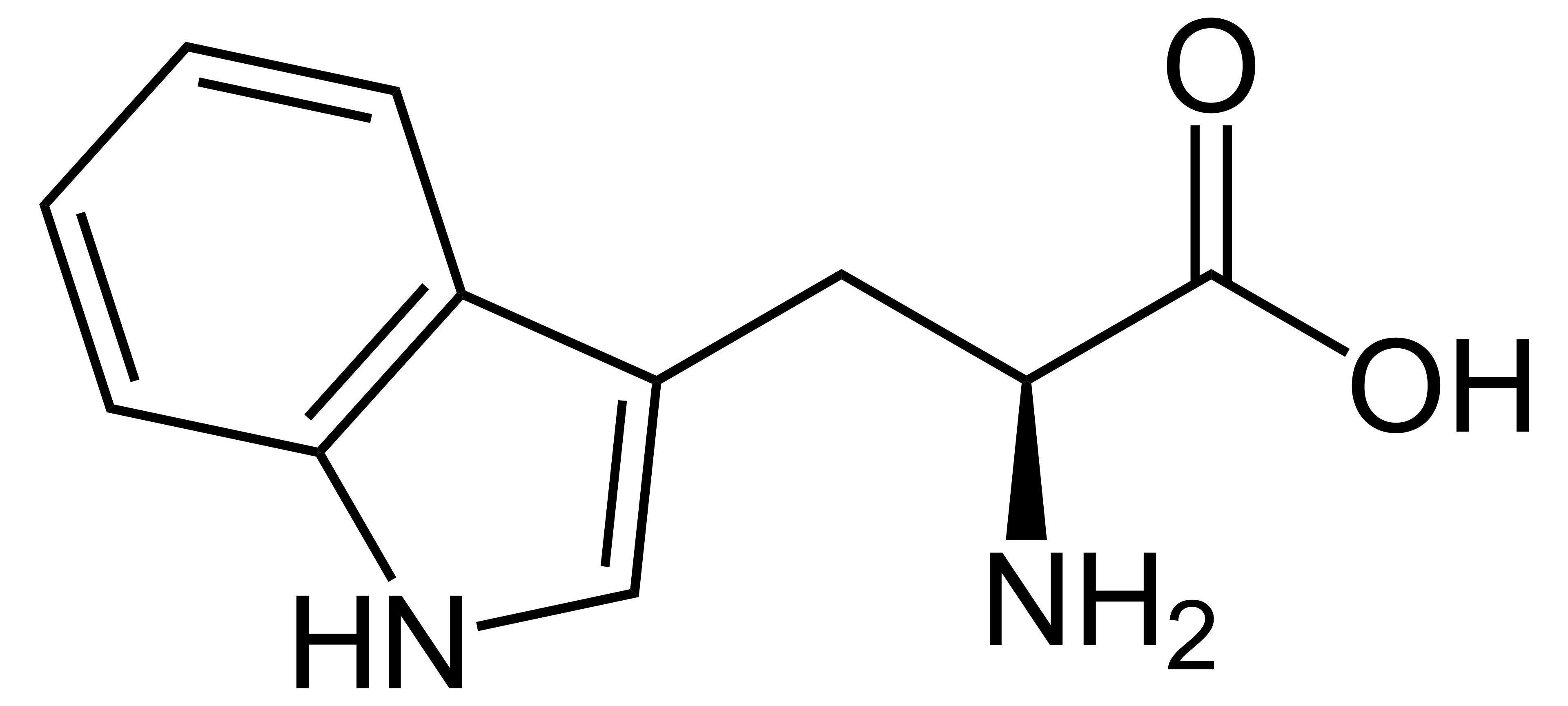
name this amino acid
tryptophan
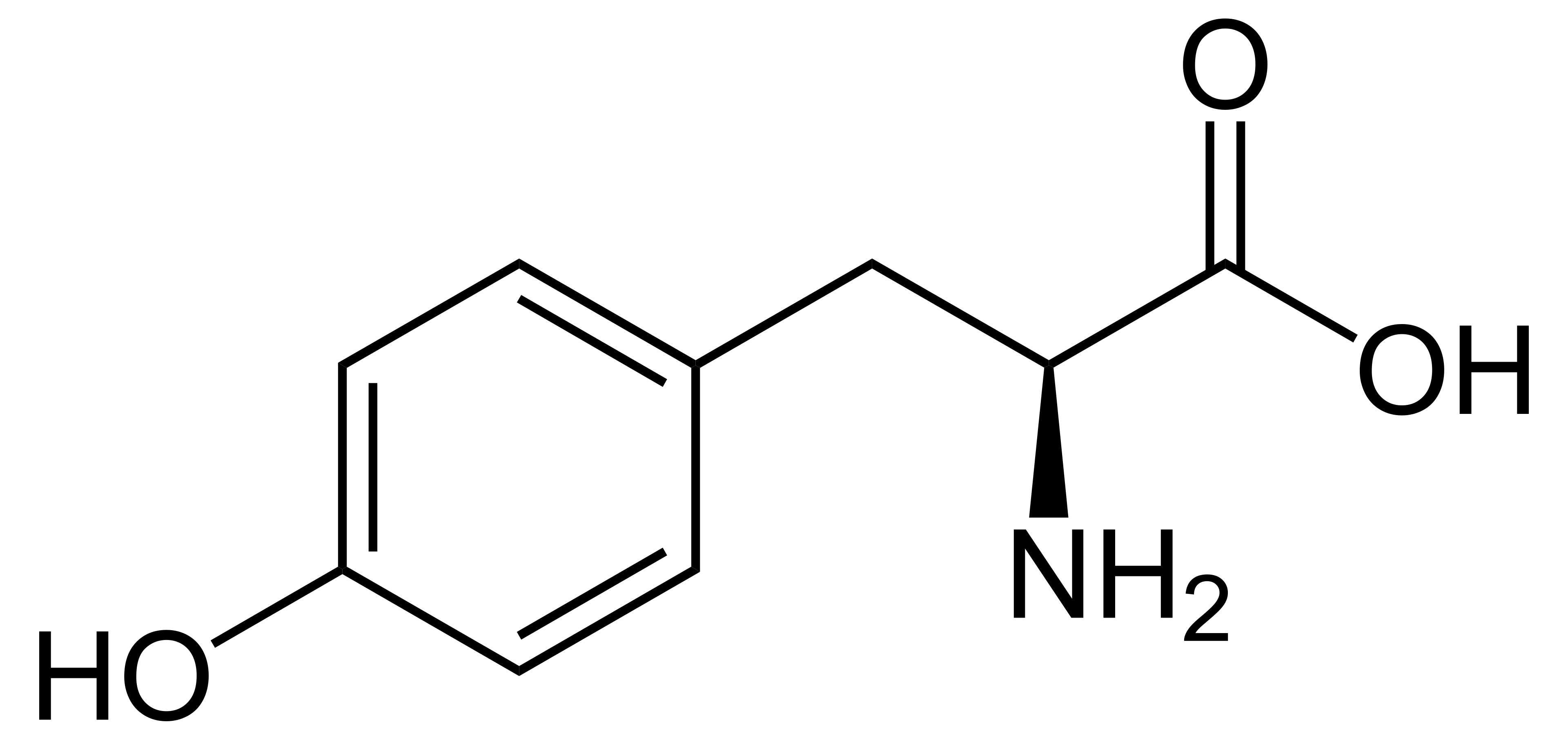
name this amino acid
tyrosine
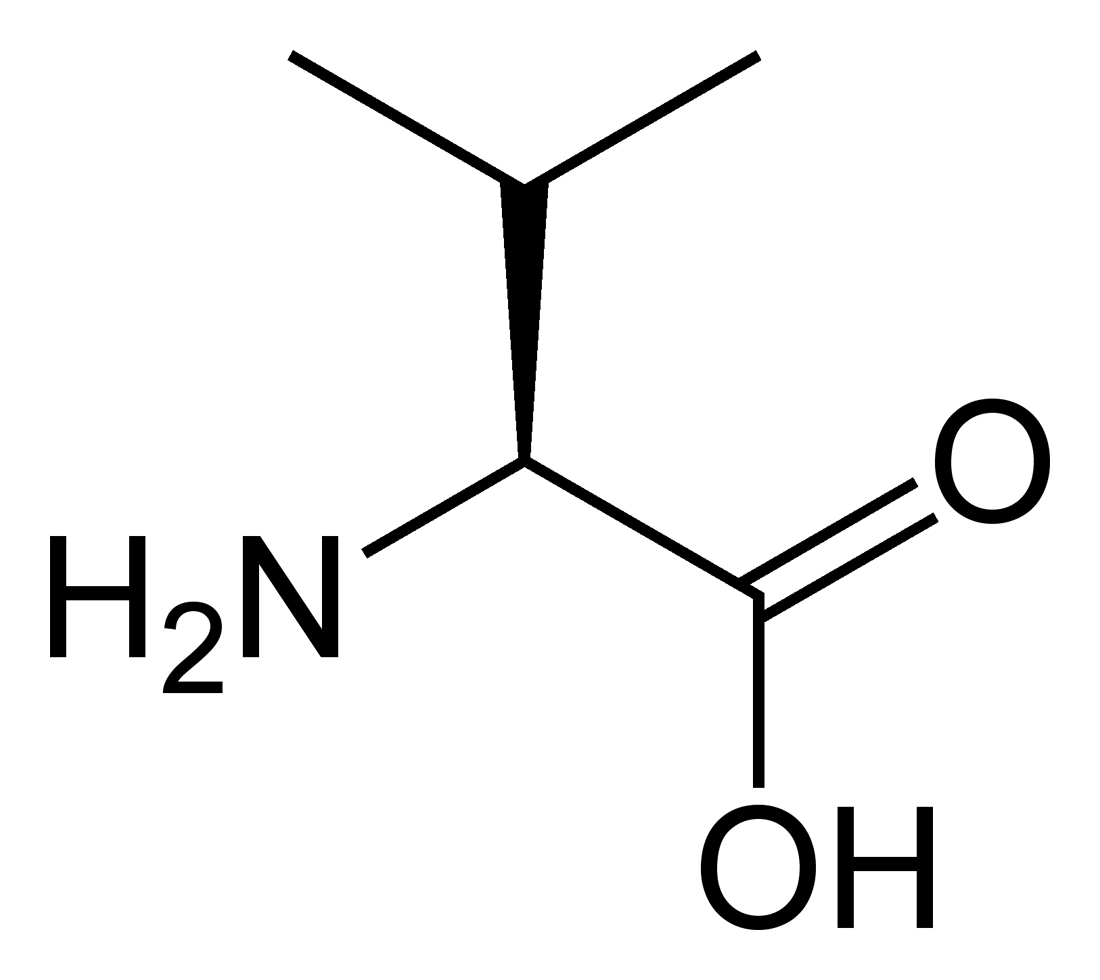
name this amino acid
valine
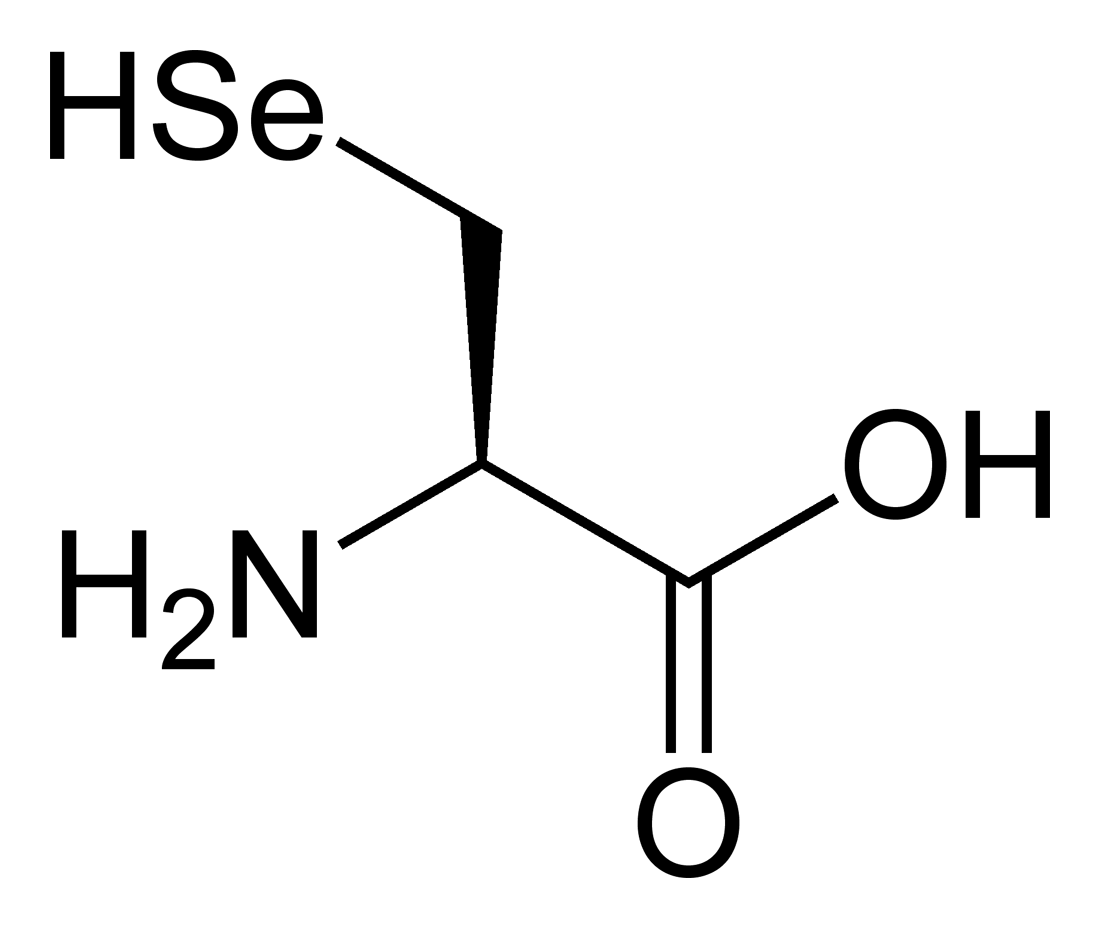
name this amino acid
selenocysteine
isomers that do not have the same connectivity are:
constitutional isomers
isomers that have the same connectivity are:
stereoisomers
stereoisomers that cannot be separated are:
confomers
stereoisomers that can be separated are:
configurational isomers
two configurational isomers that behave as image and mirror image are:
chiral
50:50 mixture of the two enantiomers is called:
racemate
unequal mixture of two enantiomers is:
enantioenriched
types of carbohydrates by the degree of polymerisation are:
monosaccharides, disaccharides, oligosaccharides, polysaccharides
if a carbohydrate’s ring has 6 atoms, it is called a:
pyranose
if a carbohydrate’s ring has 5 atoms, it is called a:
furanose
carbohydrates can be classified based on the location of the carbonyl group as ____ and ____
aldose, ketose
enantiomers have ___ chiral centers inverted
all
diastereomers have ___ chiral centers inverted
some, not all
diastereomers can be ___ or ___
syn, anti
diastereomers of ____ systems can be adressed as cis/trans
cyclic
L-arabinose has it’s lowest hydroxyl group on the ___ side in ___ projection
left, Fischer
diastereomers that differ in only one asymmetric center are:
epimers
saccharids have ___ and ___ anomers
alpha, beta
when cholesterol is transported from liver to a different organ, it is carried in _____ system
liver protein
the simplest amino acid is:
glycine
only non-chiral amino acid is:
glycine
only R-amino acid is:
cystein
proline can greatly influence structure of proteins due to its:
cyclic structure
carboxylic acid pKa is around:
5
cysteine stabilizes proteins by forming:
covalent bonds
peptide bonds are stiff due to similarity to:
alkenes
proline causes _____ in protein structure
bends
nucleoside becomes nucleotide when it is:
phosphorated
____ is a non-protein chemical compound or metallic ion that is required for an enzyme's role as a catalyst. They are usually not consumed.
cofactor
the ___ amino acids encoded by our genome are all ____ amino carboxylic acids, _-amino acids (except _____) and they differ in the substitution of the ___-carbon.
20, alpha, L, glycine, alpha
the only natural amino acid with _-configuration is ____ due to its ____ functional group
R, cystein, thiol
hydrophobic alpha-amino acids with no additional functions are:
alanine, valine, leucine, isoleucine, methionine
alpha-amino acids with polar charged groups are:
lysine, arginine, histidine, aspartic acid, glutamic acid
hydrophobic alpha-amino acids with an aromatic group are:
phenylalanine, tyrosine, tryptophane
alpha-amino acids with polar uncharged groups are:
serine, threonine, asparagine, glutamine
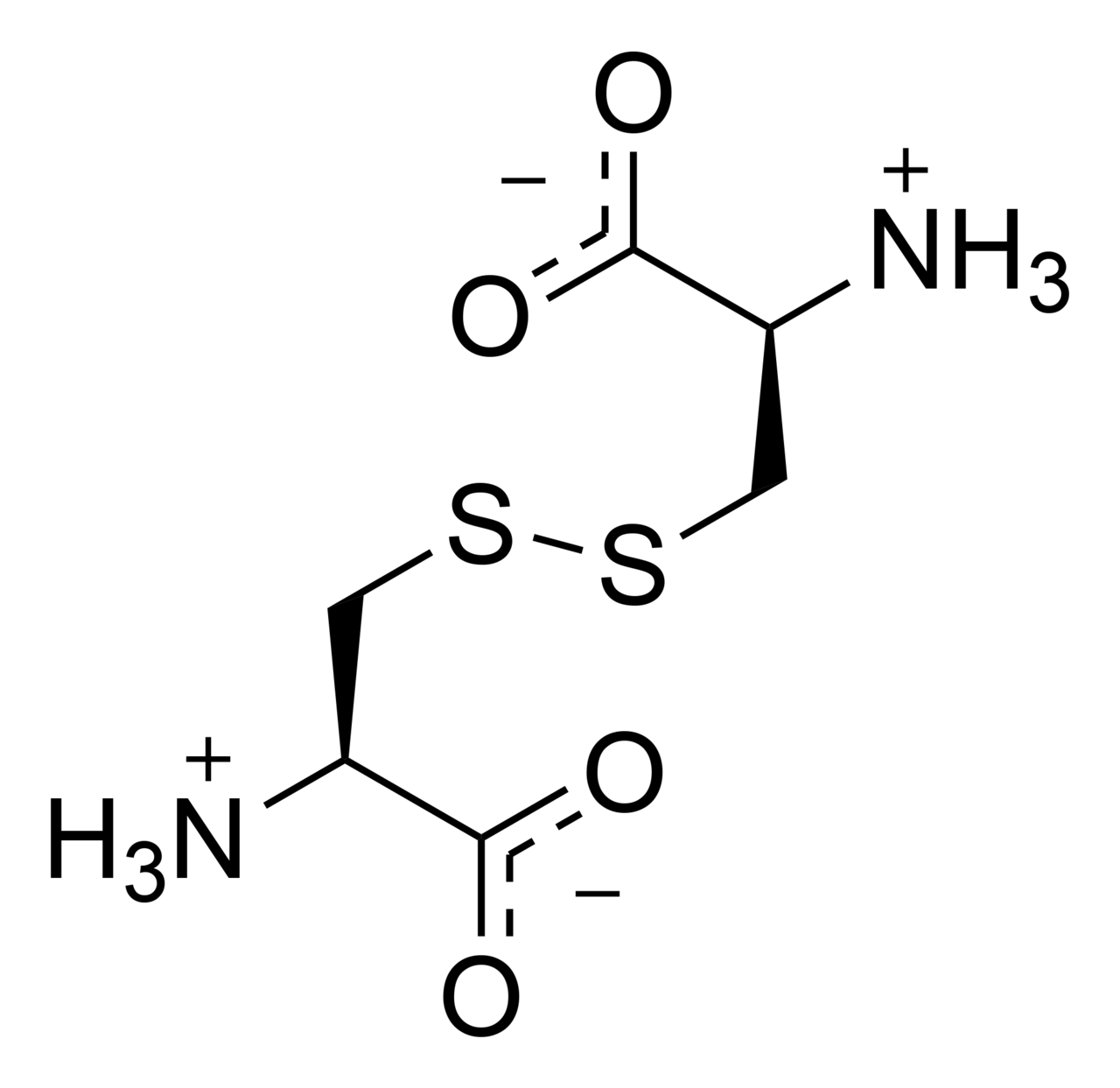
name this derivative of amino acid
cystine
two of amino acid _____ can oxidize and form _____
cysteine, cystine
amide bond between two amino acids is called:
peptide bond
metabolic pathway where complex molecules are broken down (usually under release of energy) is:
catabolism
metabolic pathway where simple molecules and energy are used to biosynthesize complex molecules is:
anabolism
ATP is a double _____
anhydride
during activation of carboxylic acid, following phosphate groups could be attacked:
any
during first stage of catabolism fats are turned into ____, carbohydrates are turned into ____ and proteins are turned into _____
fatty acids, monosaccharides, amino acids
although proteins can be digested for ATP, they are usually used for maintaining _____
muscle
acetyl-CoA is an:
activated acetic acid
to metabolize fatty acids they are activated with ____ molecule(s) of ____
one, ATP
short- to medium-chain oligomers of amino acids are:
peptides
____ is any neutral chemical compound with a positively charged cationic functional group that bears no hydrogen atom, such as a quaternary ammonium or phosphonium cation (generally: onium ions), and with a negatively charged functional group, such as a carboxylate group that may not be adjacent to the cationic site.
betaine
peptide bonds have a resonance structure due to lone pair of ____ pushing into ____ orbital of ____ bond
nitrogen, pi antibonding, C-O
resonance structure of a peptide bond includes a _____ between ______, which increases stiffness
double bond, nitrogen and carbon
in peptide bond amide oxygen acts a strong H-bond ____ and amide nitrogen acts as strong H-bond ____
acceptor, donor
peptides have structural flexibility in the bond between ____ and ____; and in bond between ____ and ____
nitrogen, alpha carbon, carbonyl carbon, alpha carbon
most peptide bonds have ____ configuration, but proline is a significant contributor to ___ configuration
trans, cis
the two organised local features of secondary protein structures are ____ and ____
alpha-helix, beta-pleated sheet
in alpha-helix each ___ bonds to ___, ___ amino acids away
N-H, C=O, four
____ does not fit into alpha-helix, and neither do ____ amino acids
proline, beta-branched
beta-pleated sheets prefer ____ amino acids
small
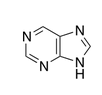
name this molecule
purine
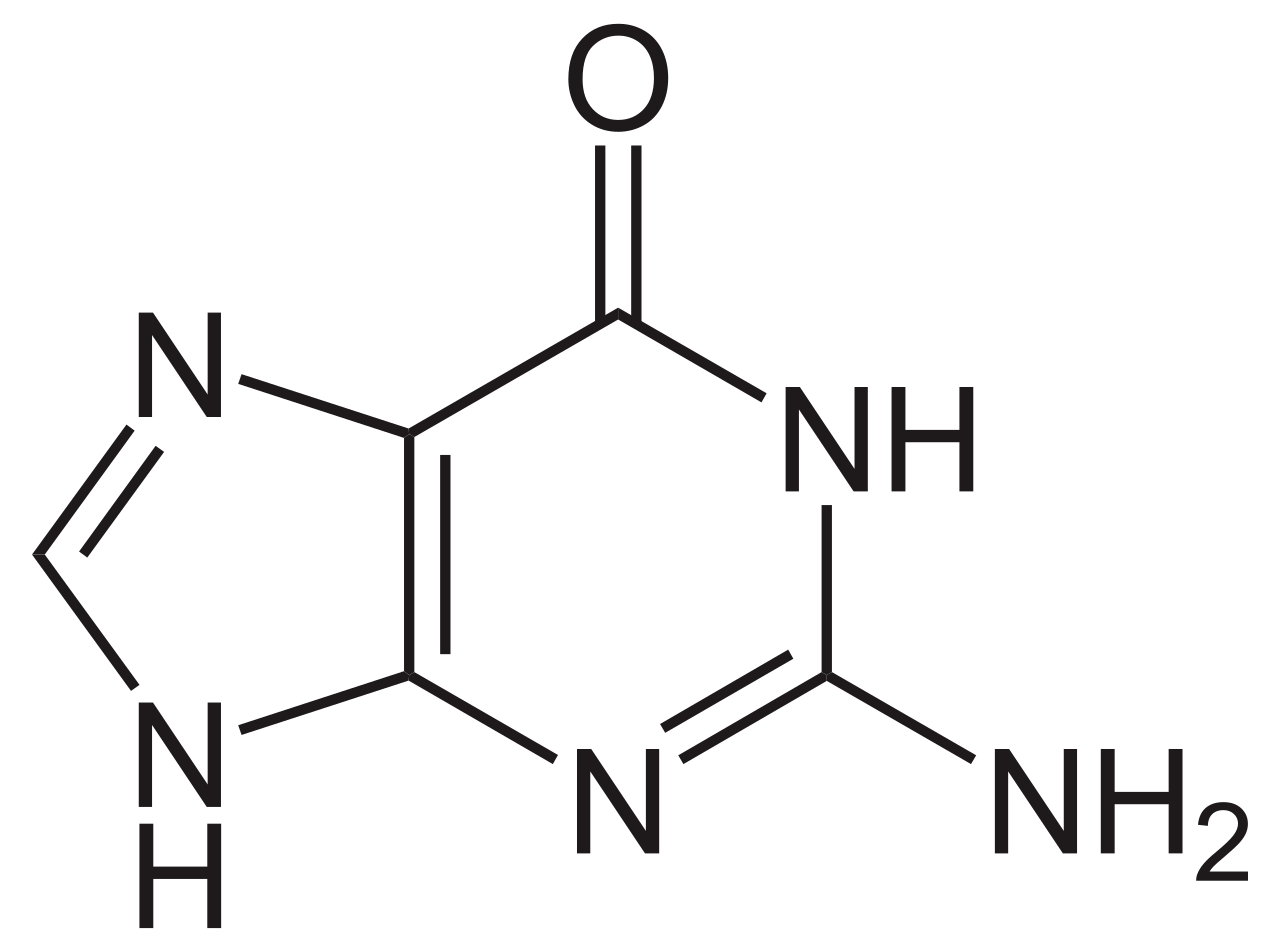
name this nucleotide base
guanine
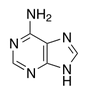
name this nucleotide base
adenine
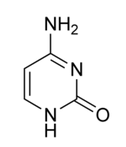
name this nucleotide base
cytosine
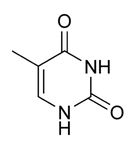
name this nucleotide base
thymine
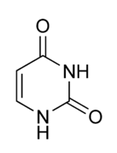
name this nucleotide base
uracil
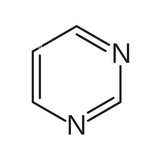
name this molecule
pyrimidine
nucleosides employ either ___ or ___ as sugars
ribose, 2-deoxyribose
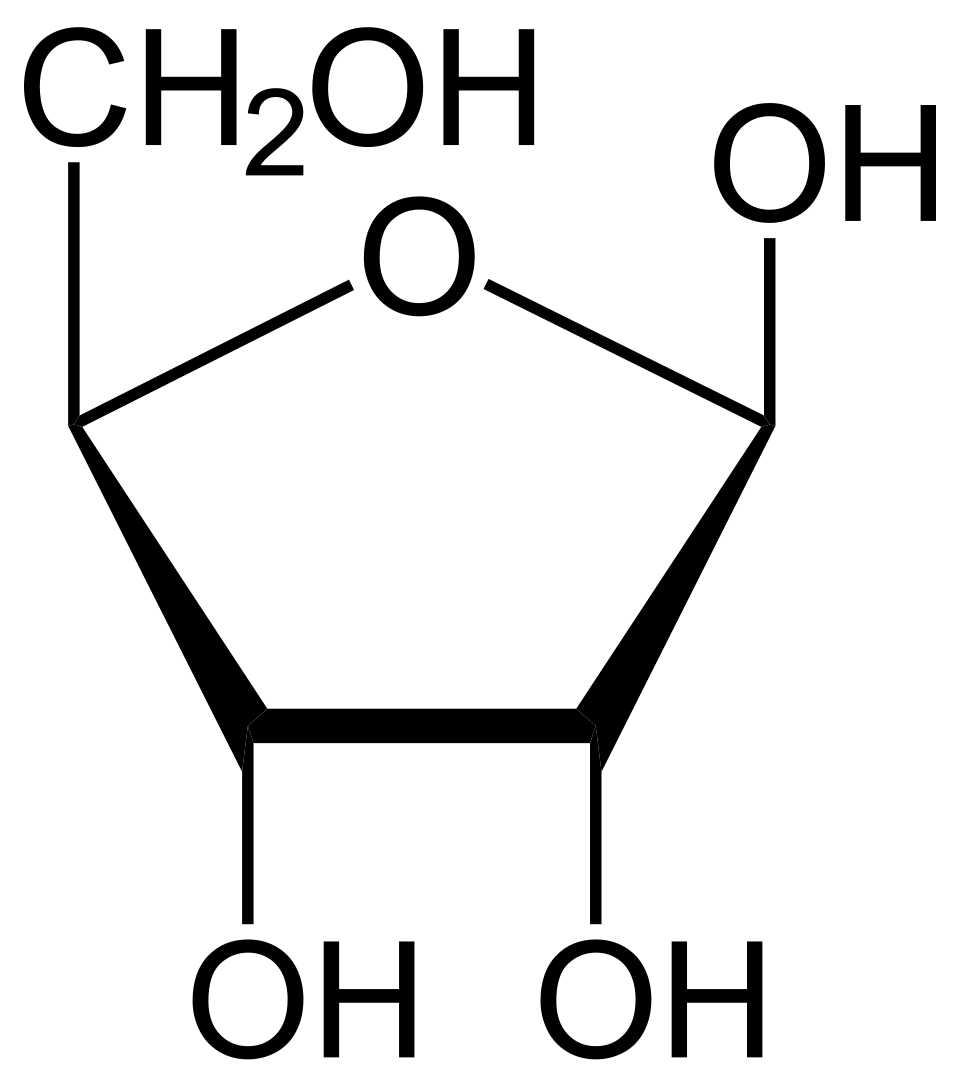
name this saccharide
beta-D-ribofuranose
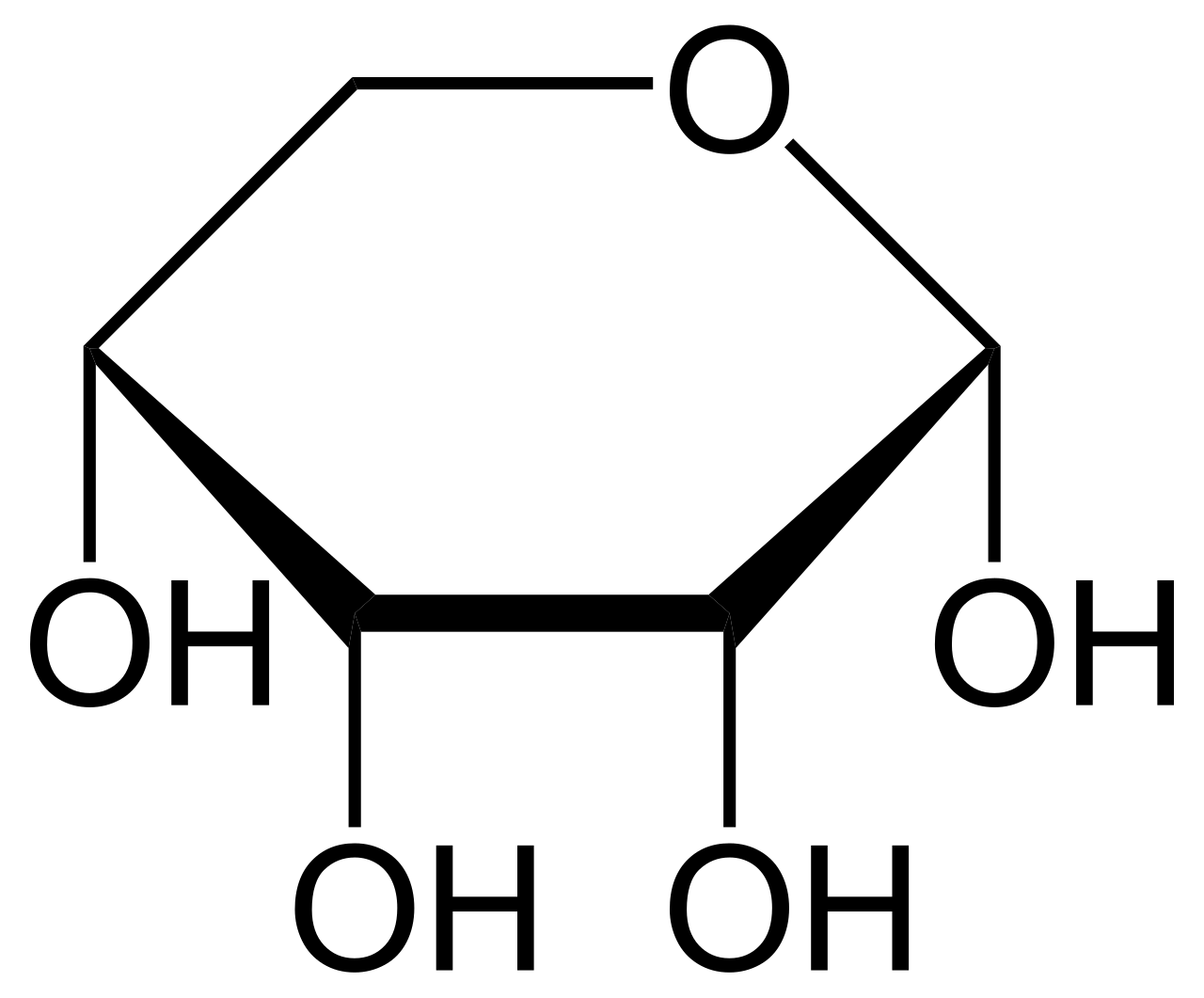
name this saccharide
alpha-D-ribopyranose
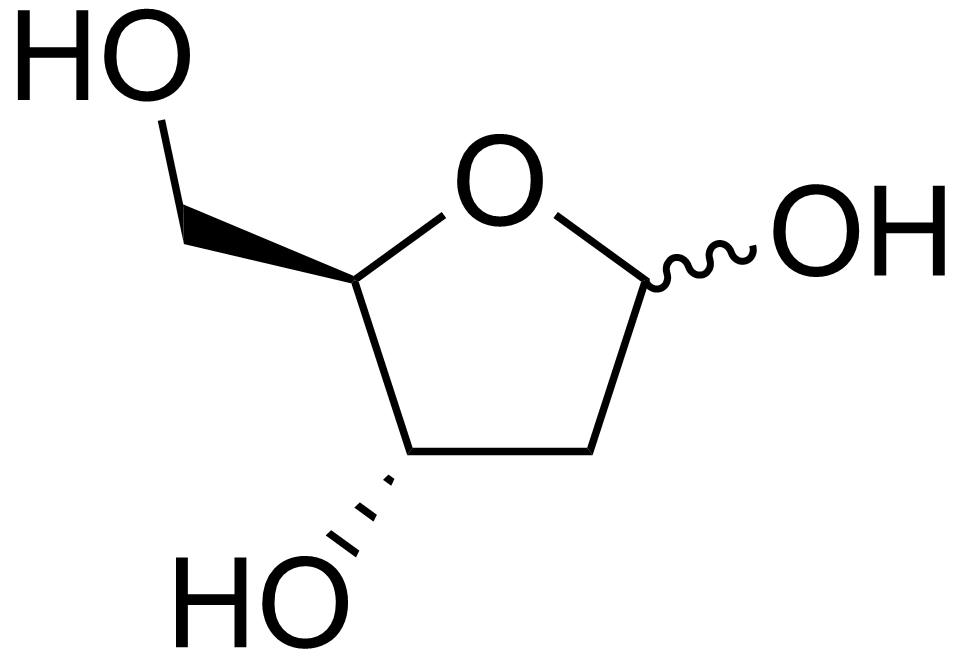
name this saccharide
2-desoxyribose
in nucleosides, nucleobases are linked to the furanose via its ____ center
anomeric
the carbon where a sugar’s ring closure occurs is the ___ carbon
anomeric
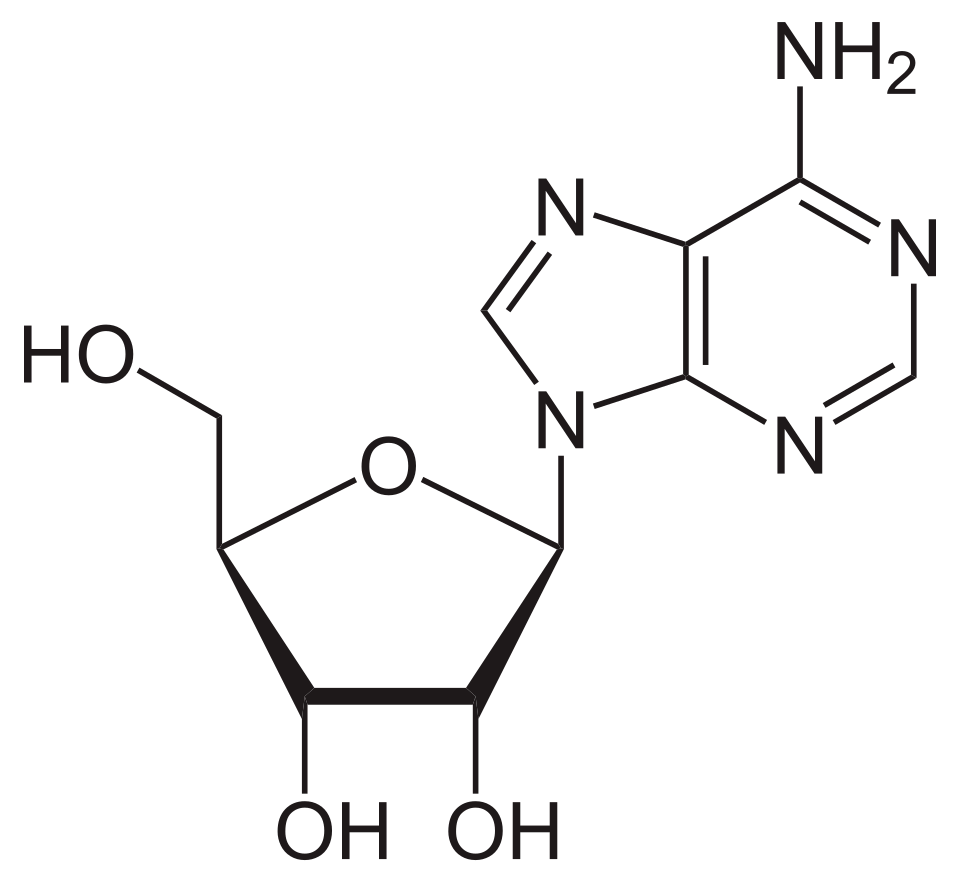
name this nucleoside
adenosine
in a nucleoside, the anomeric carbon is linked through a glycosidic bond to the N_ of a purine or the N_ of a pyrimidine
9, 1
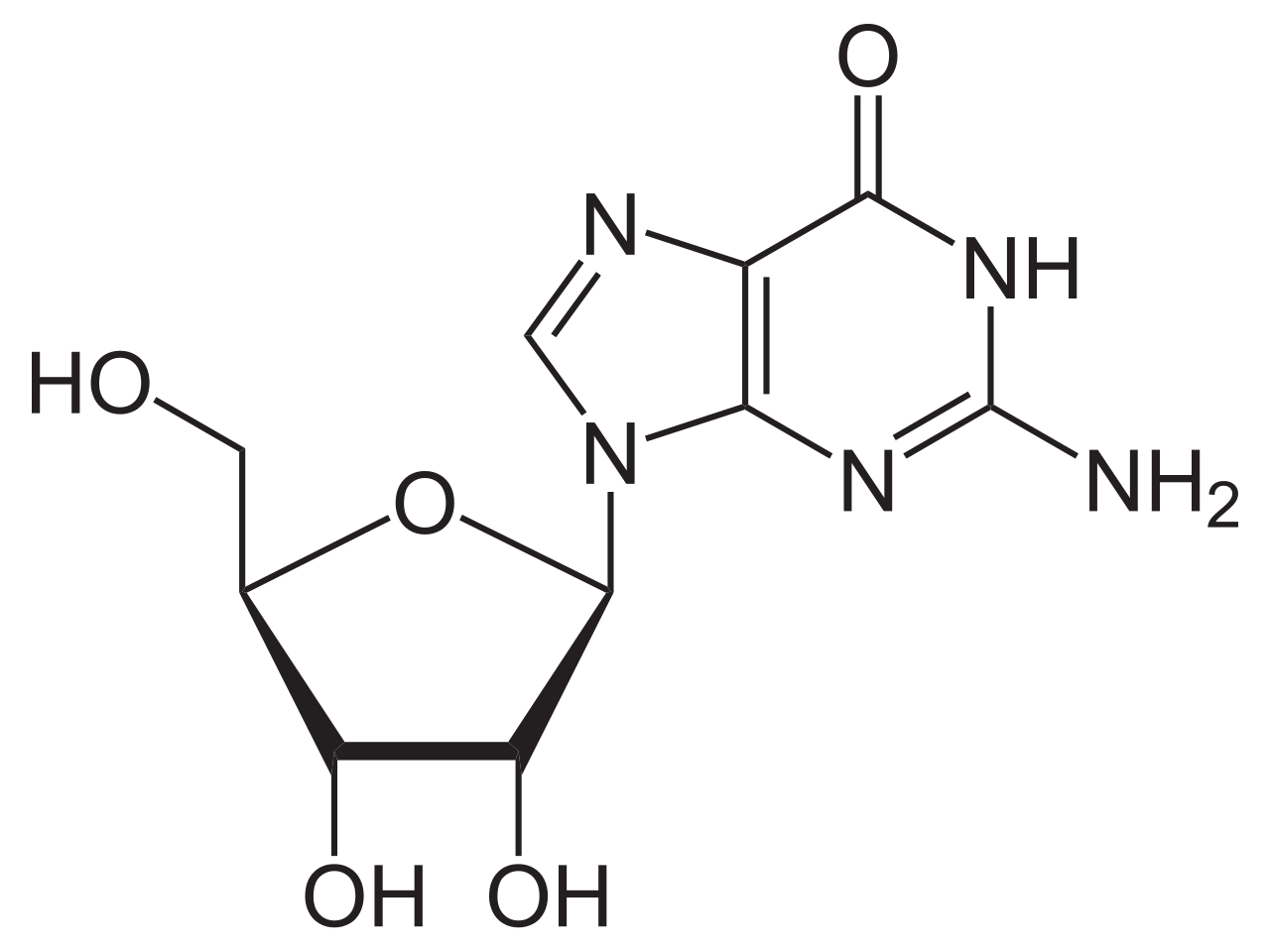
name this nucleoside
guanosine
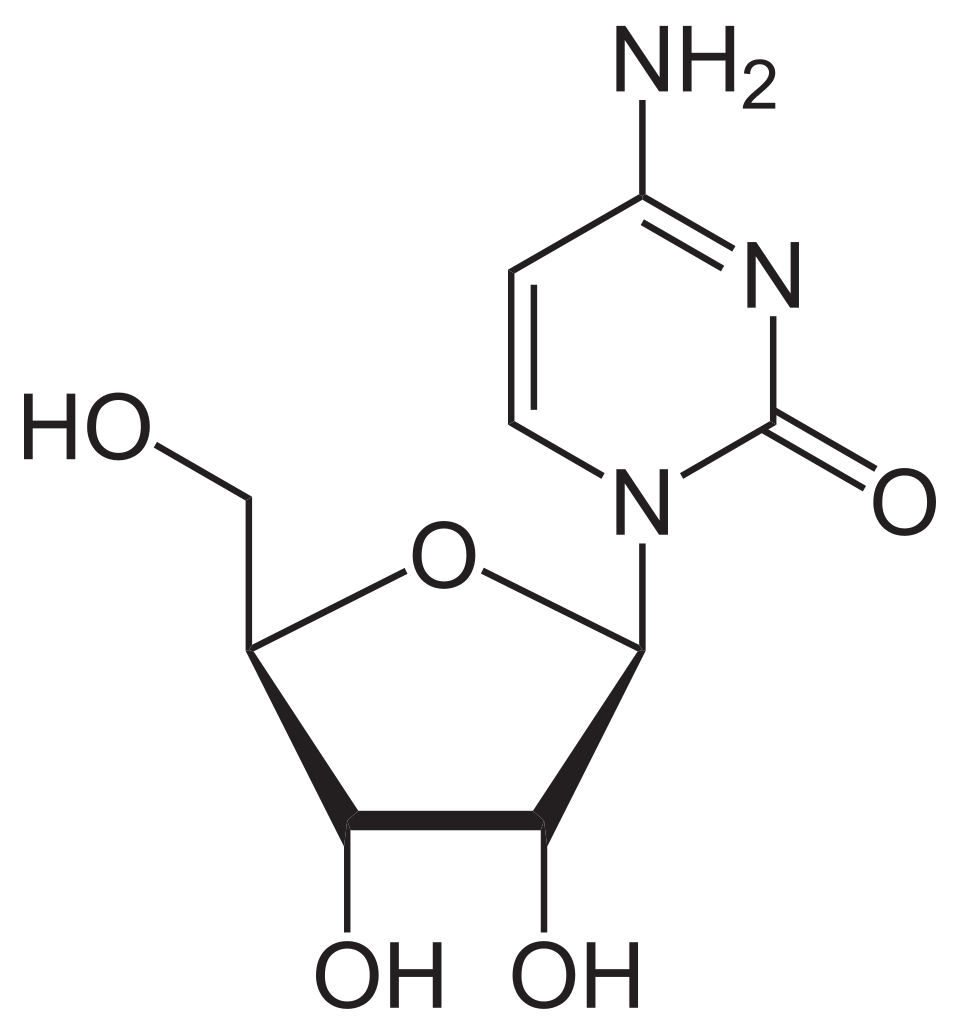
name this nucleoside
cytidine
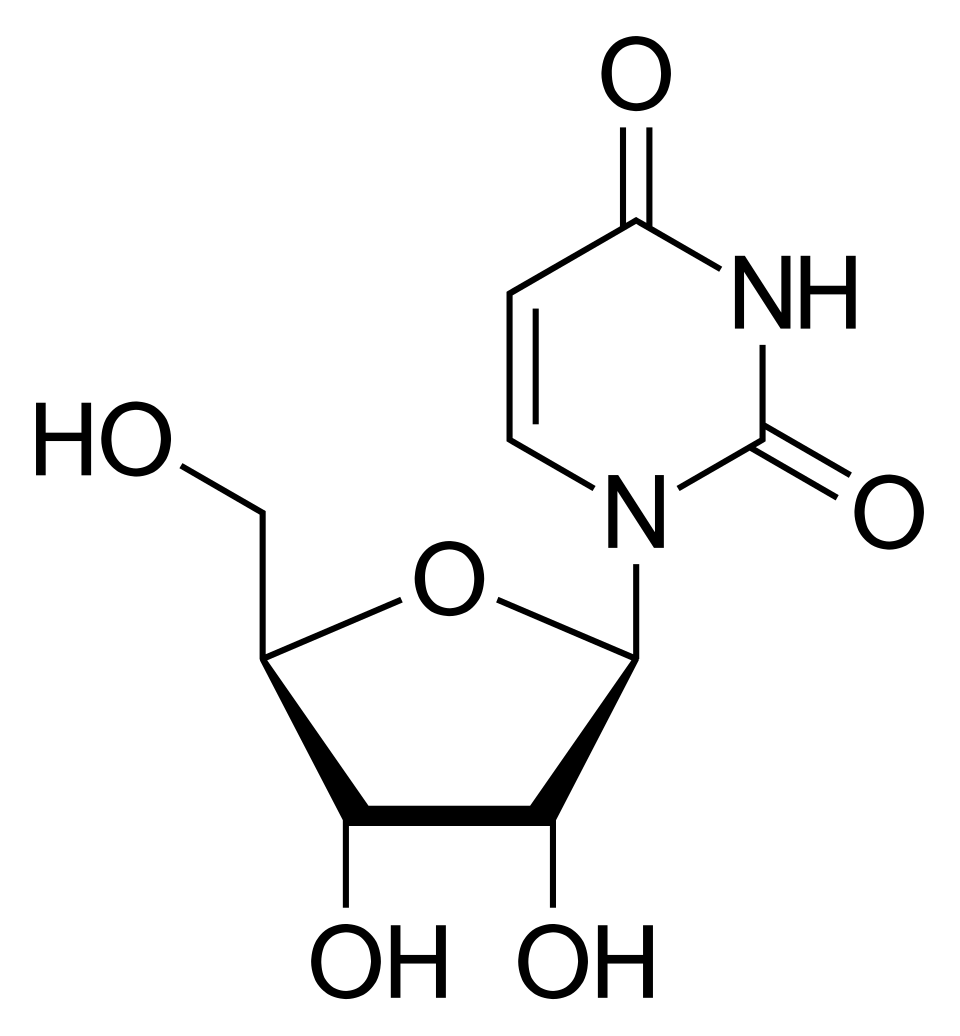
name this nucleoside
uridine
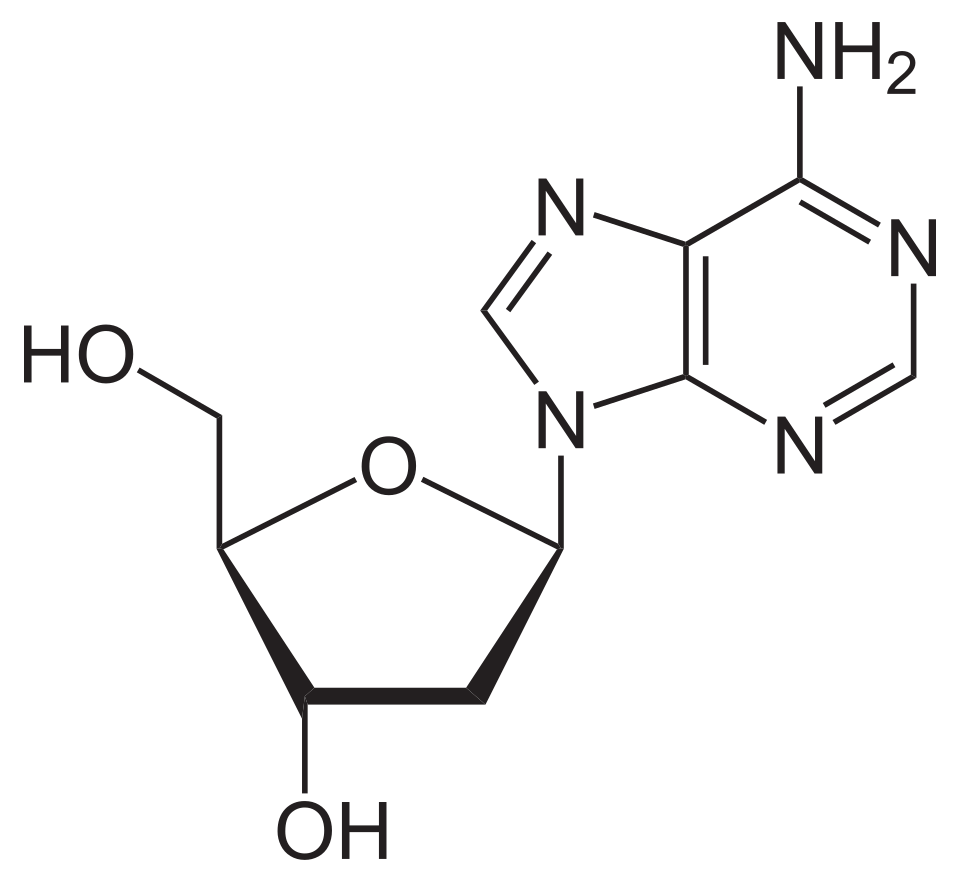
name this nucleoside
2’-deoxyadenosine
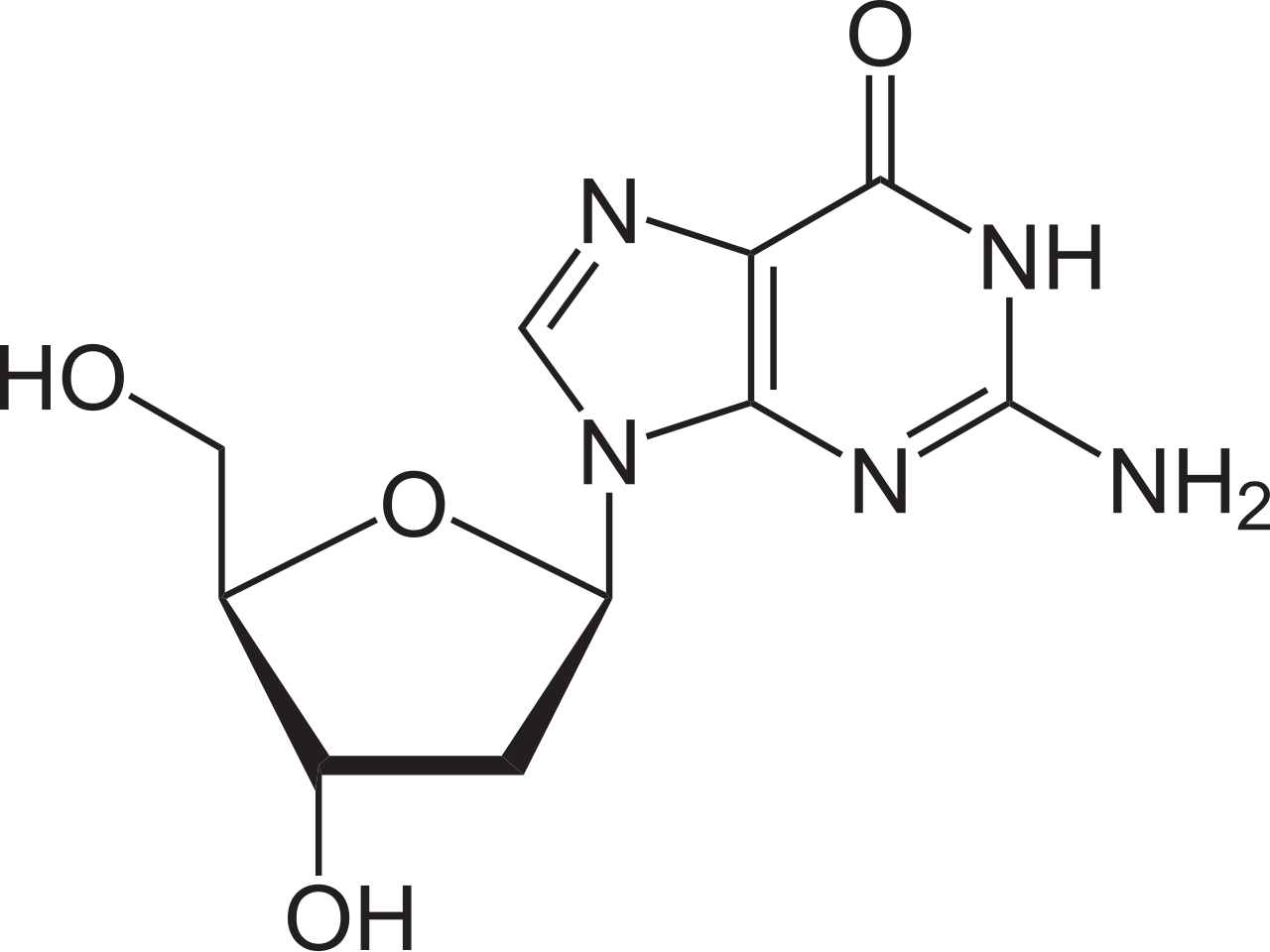
name this nucleoside
2’-deoxyguanosine
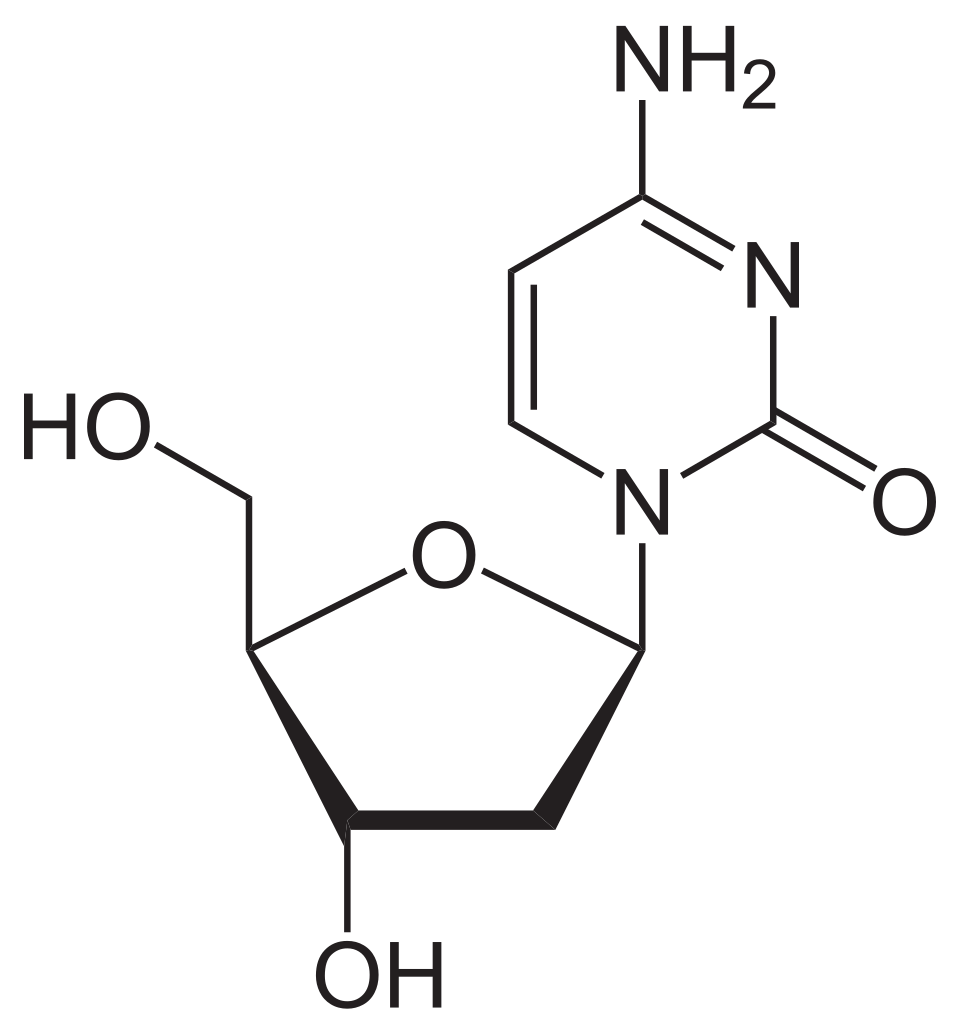
name this nucleoside
2’-deoxycytidine
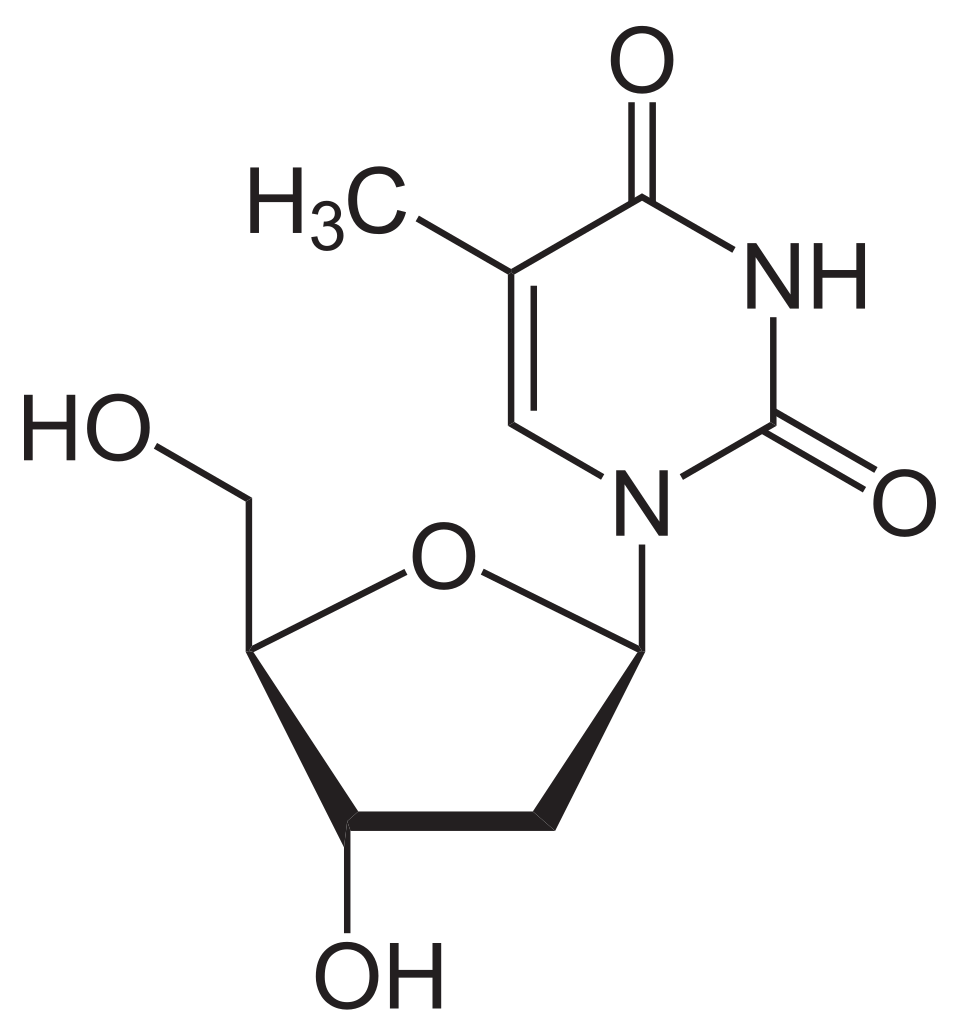
name this nucleoside
thymidine (deoxythymidine)