Physics- Electrons
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
What is the photoelectric effect?
The photoelectric effect is the emission of electrons from the surface of a metal due to electromagnetic radiation of a suitable frequency.
How are electrons produced in an X-ray tube?
A hot cathode (low voltage filament) produces electrons by thermionic emission.
How are electrons accelerated in a cathode ray tube?
There is a large voltage (e.g. 2000 V) called the anode voltage between the cathode and anode. The electrons are accelerated from the cathode to the anode due to this voltage.
Give two ways of deflecting a beam of electrons.
By means of an electric field and a magnetic field
How are X-rays produced?
The electrons are accelerated towards an anode by a high voltage striking a tungsten target which is set into the anode and produce X-rays.
The physicist Robert Millikan is usually associated with what physical quantity?
The fundamental unit of electricity charge (the charge on electron = 1 ∙ 6 × 10−19 C)
What property of light controls the current in a photocell?
Intensity
Explain what is meant by the statement: “Zinc has a threshold frequency of 1.04 × 1015 Hz”.
Photons with a frequency below this value will not cause emission of electrons
Explain how electrons are (i) produced, (ii) accelerated in an X‐ray tube.
Electrons are produced by thermionic emission (at the cathode) and (towards the anode) accelerated by a high (anode) voltage
What particles move between the electrodes of the photocell?
Electrons
Explain with the aid of a labelled diagram how X-rays are produced.
A hot cathode produces electrons by thermionic emission. The electrons are accelerated towards an anode by a high voltage. A vacuum ensures that nothing slows down the electrons. The high-energy electrons hit a tungsten target which is set into the anode and produce X-rays. Lead shield with a small window ensures that X-rays are emitted in one direction; the shielding therefore protects the operator and patient from excess radiation. Only 1% of the electrons produce X-rays. About 99% of the electrons produce heat in the anode
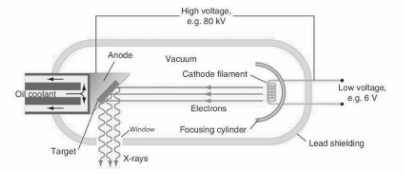
Justify the statement “X-ray production may be considered as the inverse of the photoelectric effect.”
In X-rays, light is emitted when sufficiently energetic electrons strike a metal target, whereas in the photoelectric effect, electrons are emitted when sufficiently energetic light electrons strike a metal target.
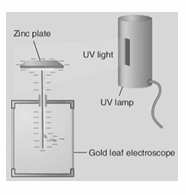
Describe an experiment to demonstrate the photoelectric effect.
Apparatus – UV light, visible light source, zinc plate, gold leaf electroscope. Procedure – • Place the zinc plate on the cap of the electroscope.• Charge the electroscope negatively (by induction). • Shine the UV light on the plate. Observation & Conclusion – The leaf in the GLE collapses
Give two applications of a photocell.
burglar alarms, smoke alarms, automatic doors, safety switches on cutting machinery, laboratory light meters, optical soundtrack in film and control sensors in central heating boilers.
Outline Einstein’s explanation of the photoelectric effect.
Light must be considered as a stream of ‘packets of energy’; each packet or bundle of energy is called a photon,
The energy 𝐸 of a photon is proportional to the frequency of the light associated with that photon, the constant of proportionality is Plank’s constant, giving the equation 𝑬 = 𝒉𝒇.
When light strikes the metal, each photon gives all its energy to one electron, each electron will only get struck by a maximum of one photon.
The energy needed to remove the loosest electrons from the surface of a metal is the work function of that metal, this corresponds to the threshold frequency of the photon.
If the energy of each photon is less that the work function of the metal, no electrons are emitted. If the energy of the photon is greater that the work function, electrons are emitted. The amount by which the energy of the photon exceeds the work function appears as kinetic energy of the electron. This is summarised in the equation ℎ𝑓 = Φ +1/2m𝑣max2.
List two properties of the electron.
It has a negative charge. Its mass is 1 1836 (6) amu, orbits nucleus, deflected by electric and magnetic fields
Name the Irishman who gave the electron its name in the nineteenth century.
G. J. Stoney
Give an expression for the force acting on a charge q moving at a velocity v at right angles to a magnetic field of flux density B
F=qvB
What happens to the energy of the electron when it hits the screen of the CRT?
Converted to light and heat
Distinguish between photoelectric emission and thermionic emission.
Thermionic emission is the emission of electrons from the surface a hot metal. The photoelectric emission is the emission of electrons from the surface of a metal due to electromagnetic radiation of a suitable frequency.
A freshly cleaned piece of zinc metal is placed on the cap of a negatively charged gold leaf electroscope and illuminated with ultraviolet radiation. Explain why the leaves of the electroscope collapse.
Photoelectric emission occurs. This causes the leaves become uncharged and collapse
A freshly cleaned piece of zinc metal is placed on the cap of a negatively charged gold leaf electroscope and illuminated with ultraviolet radiationExplain why the leaves do not collapse when;
the zinc is covered by a piece of ordinary glass;
the zinc is illuminated with green light;
the electroscope is charged positively.
Ordinary glass does not transmit UV light
The energy of green light is too small to overcome the work function of the metal.
Photoelectric emission does not occur because any electron emitted are attracted back to the positive electroscope.
What are X-rays and how do they differ from light rays?
X-rays are electromagnetic radiation of extremely high frequency
Give two uses of X-rays.
Analysis of bone structure (medical) / security luggage scanners (at airports) / detect structural problems and cracks in metals (industry)
When electrons hit the target in an X-ray tube, only a small percentage of their energy is converted into X-rays. What happens to the rest of their energy and how does this influence the type of target used?
Only 1% of the electrons produce X-rays. About 99% of the electrons produce heat in the anode. The heat produced is dissipated by cooling liquids circulating through the anode, which is made of tungsten which has a high melting point.
What is a photon?
A photon is a quantum of light energy
Explain why a current does not flow in the photocell when the frequency of the light is less than 5.2 × 1014 Hz .
The frequency is lower and drops below the threshold frequency.
Describe how a photocell conducts current.
When light falls on the cathode, electrons are emitted. These electrons accelerate across to the anode, thus a small current flow.
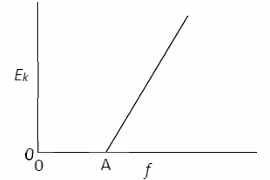
What physical quantity is represented by (i) point A, (ii) the slope of the graph?
(i) Threshold frequency
(ii) Planck constant
Where in the tube are the electrons produced?
Cathode
The kinetic energy of some electrons is converted into heat energy. State two design features of an X‐ray tube that take account of this.
The tungsten target has a high melting point. The cooling fluid brings heat away from the anode.
Write down an expression for Einstein’s photoelectric law.
hf= Φ +1/2mvmax2
Who discovered x-rays? when?
Wilhelm Röntgen 1895)
Describe an experiment to demonstrate how the current through a photocell can be increased.
Apparatus – Photocell, light source, milliammeter, power source, ruler. Procedure – • Set us apparatus as shown. • No current flows when no light is shining on the photocell. • Measure the distance (𝑑) from the light source to photocell. • Shine the light on the photocell. • Note the current (𝐼) using the milliammeter. (12) (12) • Change the distance and repeat process, so as to get six or seven readings. Observation– Current only flows when light is shining on the photocell. The current (𝐼) increases as the distance (𝑑) decreases. Conclusion – Photoelectric emission is taking place in the photocell when light shines on it.
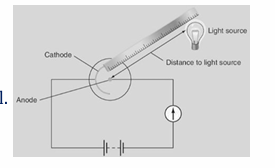
Draw a diagram to show the structure of a photocell.
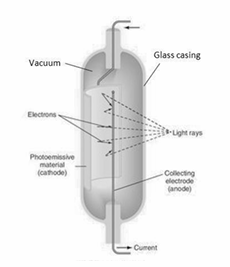
In the photoelectric effect electrons are emitted from the surface of a metal when the incoming light of intensity 𝐼 has a frequency 𝑓 that exceeds a certain value 𝑓0, the threshold frequency. Describe what happens when
(i) f >𝑓0 , 𝑓 is constant and 𝐼 is increasing,
(ii)𝑓 < 𝑓0 , 𝑓 is constant and 𝐼 is increasing.
(ii) f>𝑓0 , 𝑓 is increasing and 𝐼 is constant,
(i) more electrons emitted, with the same energy
(ii) no electrons emitted
(iii)same number of electrons emitted, but with greater energy