Chemistry RPs
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
r
RP 1a: making a standard solution
weigh a weighing boat on a mass balance
add the acid to mass balance and record mass
tip acid into beaker record mass
subtract initial from final
add 100cm3 distilled water to beaker mix until acid dissolves
transfer to volumetric flask with washings
add distilled water up to 250cm3 mark
invert multiple times to mix
RP 1b: acid-base titrations
fill burette with known concentration of acid/alkali with known volume
add fixed volume of acid/alkali (unknown conc.) into conical flask with volumetric pipette
add 3 drops of indicator and swirl white tile under
slowly add volume in burette, swirling between additions
add dropwise at first sight of colour change
stop at permanent colour change
record volume and repeat until titres are concordant calculate mean
RP 2: measuring an enthalpy change
measure acid using burette and pour into polystyrene cup
record solution temp. for at least 4 mins before addition with thermometer
weigh powdered carbonate on watch glass
reweigh watch glass afterward and calculate difference
add carbonate stir with thermometer and close lid
record temp. at 1 min intervals until trend is observed
plot graph temp (y)/time (x), extrapolate to point of addition
determine temp change at addition point
RP 3: effect of temp. on rate of reaction
place X underneath beaker in water bath
use water bath to keep temp. consistent at 25 degrees
add fixed volume of acid to beaker, then thiosulfate
start stopwatch immediately - record time taken for cross to disappear
monitor water bath temp. using a thermometer regularly
repeat at 30, 35, 40, 45, 50 degrees
calculate rate = 1/t
RP 5a: distillation as a separation technique
add 25cm3 reaction mixture to round bottom flask
add anti bumping granules (smaller bubbles)
use electric heater, clamp flask to Liebig condenser, end of condenser should drop into beaker in ice cold water bath
turn on tap, allow water to fill condenser
heat gently at boiling point of desired product tracked using thermometer (down at T junction)
keep temp constant at this point - compound will evaporate and condense in water bath
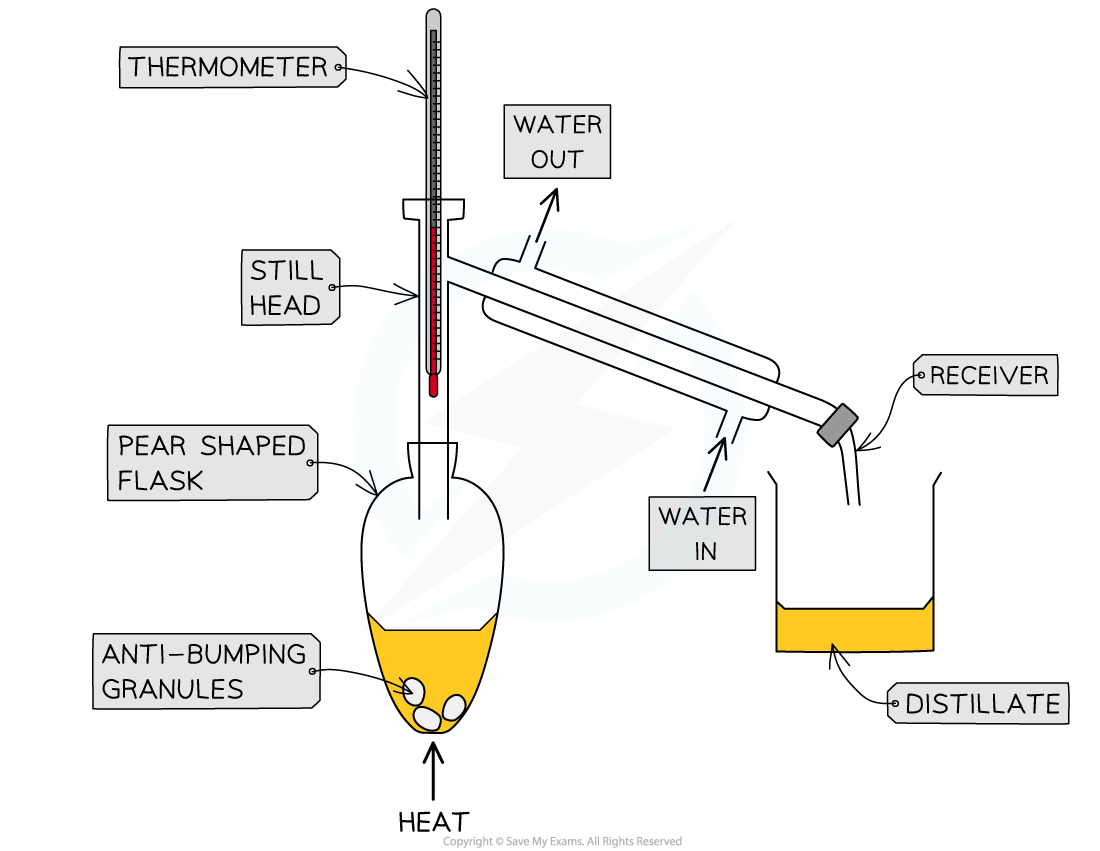
RP 6: organic tests
(gently heat wb) acidified K2Cr2O7 - 1o = orange → green aldehyde 2o = orange → green (ketone) 3o no change
aldehyde + tollen’s (heat gently wb) - colourless → silver mirror
aldehyde + fehling’s (heat gently wb) - blue → brick red ppt.
chloroalkane + acidified AgNO3 (heat gently wb) - white AgCl ppt forms slowly
acyl chloride + acidified AgNO3 - white steamy HCl fumes, immediate white AgCl ppt
RP 7a: initial rate of reaction
→ iodide ions + H2O2 → I2 reacts with S2O32- the excess I2 reacts with starch - blue black
measure required volume of KI, Na2S2O3, starch and water into conical flask
measure H2O2 separately, immediately start timer after addition
stop when solution is blue-black
repeat with diff. concs/ of KI
→ can measure the amount of time taken for a fixed amount of X to be produced
RP 7b: continuous monitoring of reaction
measure 50cm3 HCl in conical flask using burette
attach gas syringe to conical flask with delivery tube
weigh 0.2g Mg and add to conical flask
place bung firmly and start stopwatch
record vol of H2 gas every 30 secs for 3 mins
→ can also measure change in mass, remove small samples (quench reaction) then titrate to find conc., colorimetry
RP 8: EMF of a cell
clean pieces of copper and zinc using emery paper
degrease them using cotton wool + propanone
place copper into beaker with 50com3 CuSO4 solution
place zinc into beaker with 50cm3 ZnSO4 solution
soak filter paper in KNO3 for salt bridge & place between beakers each end in solution
use crocodile clips connect each half cell to voltmeter, record voltage
repeat with different metals
→ if metal is non-conductive us Pt electrode
→ salt bridge should be unreactive unlike KCl
→ wire not use for bridge because it would set up its own electrode system
RP 9: pH changes
calibrate pH probe using known buffers & plot calibration curve
transfer 25cm3 acid to conical flask with burette
measure initial pH using pH probe record results
add 2cm3 alkali, stir, measure pH and record it
repeat and when approaching endpoint add smaller volumes of alkali
add alkali until in excess
use pH calibration curve to adjust pH results from experiment
plot corrected pH values against alkali volume
RP 10a: making aspirin
20cm3 benzoic acid + 4cm3 ethanoic anhydride
add 5 drops H2SO4 & mix
heat beaker in water bath over Bunsen burner, leave to cool to room temp
mix with 10cm3 distilled water
pour mixture through Buchner funnel & filter - crystals will form
RP 10b: obtaining pure crystals
dissolve impure compound in min vol of hot solvent (ethanol) 70oC
hot simple filtration
leave to cool in ice bath
filter under reduced pressure with Buchner flask
pour a small volume of cold water through crystals to wash away soluble impurities
dry crystals with pieces of filter paper
→ loss of yield: crystals lost whilst washing, some product stayed in solution after recrystallisation
RP 10c: preparing and purifying an ester
50cm3 propanol + 50cm3 ethanoic acid round bottom flask with anti bumping granules
add 10 drops H2SO4 & fit reflux condenser onto flask and heat under reflux for 20 mins
cool mixture and transfer to a separating funnel
add 20cm3 of aqueous Na2CO3 and shake
shake gently then open cork, close and repeat to remove CO2 gas
allow to separate into aqueous & organic layer & add 5g anhydrous MgSO4
distil organic layer with anti bumping granules & heat with water bath just below b.p. of the ester
collect ester in conical flask and weigh ester
→ anti bumping granules prevent large bubbles explode
→ Na2CO3 reacts with any unreacted acid
→ H2SO4 is a catalyst
RP 12: TLC
draw pencil line 1cm above bottom of TLC plate
use capillary tube to add tiny drop of each solution on pencil line - allow dry
pour solver in beaker, place TLC plate with pencil line just above solvent
place beaker in fume cupboard with lid
remove plate 1cm before solvent reaches top, mark solvent front
leave to dry in fume cupboard
calculate Rf value from centre of spot