Design - Materials
1/166
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
167 Terms
Aesthetic Properties
Properties / aspects of a product that relate to appearance, smell, texture, taste
Alloy
A mixture that contains at least one metal: mixture of metals or mixture of metals and non-metals
Composite
A material comprised of two or more constituent materials that have different properties.
Compressive strength
The ability of a material to withstand being pushed or squashed.
Creep
The slow, permanent deformation of a solid material under the influence of a mechanical stress
Density
The mass per unit volume of a material.
measured in kg/m3
Ductility
The ability of a material to be drawn, pulled or extruded into wires / other extended shapes
Elasticity
The ability of a material to return to its original shape after being stretched, compressed, or deformed, once the force is removed.
Electrical insulator
Reduces transmission of electric charge
Electrical resistivity
The measure of a material's ability to conduct electricity. A material with low resistivity will conduct electricity well.
Electro-rheostatic
This smart property relates to a fluid that can undergo a dramatic change in its viscosity when exposed to an electric field.
Viscosity = how thick or thin a fluid is.
Glass
A hard, brittle and typically transparent amorphous solid made by rapidly cooling a fusion of sand, soda and lime.
Grain size (metals)
Metals are crystalline structures comprised of individual grains.
grain size depends on how quickly a metal is cooled.
Quick cooling = high number of small grains
slow cooling = low number of large grains.
as grain size increases, ductility increases, strength and toughness decreases.
Hardness
a material's ability to resist penetration, scratching, abrasion, indentation or cutting.
Hardness can be tested in a range of ways
Scratch hardness
Static & dynamic indentation hardness
Different materials have different hardness scales
original: Mohs hardness scale
Diamond = hardest mineral
talc = softest.
Plastics: Shore Hardness
Metals: Rockwell/Vickers/Brinell
Wood: Janka
Magneto-rheostatic
This smart property relates to a fluid that can undergo a dramatic change in its viscosity when exposed to a magnetic field.
Viscosity = how thick or thin a fluid is.
Mass
Relates to the amount of matter that is contained with a specific material. Mass is a constant whereas weight may vary depending upon where it is being measured.
Mechanical properties
The properties exhibited by a material when subjected to different forces, measurable through destructive testing:
hardness
stiffness
toughness
tensile strength
compressive strength
ductility
malleability
elasticity
plasticity
Young’s modulus
stress and strain
Oxidization resistance
A property of a metal that means that it does not readily react with oxygen and degrade.
Photochromicity
A material that changes colour in response to an increase in light. When the light source is removed, it returns to its original colour
Physical properties
Physical Properties are properties that can be tested without the damage or destruction of the material.
Mass
Weight
Volume
Density
Electrical resistivity
Thermal conductivity
Thermal expansion
Piezoelectricity
A property of certain smart materials that allows them to generate a small electrical charge when mechanically deformed, and to change shape (expand or contract) when an electric current is applied.
Plasticity
The ability of a material to be permanently deformed without breaking when a force is applied
Shape memory alloys
A smart material that can be plastically deformed and then return to its original, pre-set shape when exposed to a change in temperature or stress
Smart material
Materials that respond to external stimuli (eg. heat, light, pressure, or electricity) by changing one or more of their properties in a controlled, reversible, and repeatable way.
human-made
Stiffness
the ability of the material to resist being bent or deflected
can be found by calculating the gradient of the linear section of a stress-strain curve
steeper gradient = stiffer material
measured in Gigapascals (GPa)
Strain
The response of a material due to stress, defined as the change in length divided by the original length.
no units - is a ratio
Stress
A force on a material divided by the cross-sectional area of that material.
Measured in Pascals (Pa)
3 types of stress:
Tensile Stress: how much force a material can withstand when being stretched or pulled
Compressive Stress: how much force a material can withstand when being compressed
Shear Stress: a type of stress caused by forces parallel to the material plane
Super alloys
An alloy that exhibits
excellent mechanical strength,
resistance to thermal creep deformation,
good surface stability
resistance to corrosion.
Tensile strength
The ability of a material to withstand pulling forces.
Thermal conductivity
A measure of how quickly heat passes through a material when there is a temperature difference across it.
measured in watts per metre per degree Celsius (W/m·°C).
Insulation: Materials with low thermal conductivity, like fiberglass, are used as insulators.
Heat Dissipation: High thermal conductivity materials, such as copper, are used in heat sinks to dissipate heat in electronics.
Metals track their electrical conductivity to match their thermal conductivity, but this does not hold true of other material families
Copper Pans are considered the best in cooking.
Copper is chosen for its thermal conductivity for the pan.
Cast iron is chosen for the handle due to lower thermal conductivity.
Tin is chosen as a lining for the inside as a sacrificial layer that is more resistant to corrosion
Thermal expansion
A measure of the increase in dimensions when a material is heated.
length, area or volume.
measured as the fractional increase in dimension per kelvin increase in temperature
Thermo-electricity
A smart material comprised of two dissimilar conductors that produce an electric current when heated. The temperature difference causes an electric current to form.
Thermoplastic
A type of plastic that can be heated and reshaped repeatedly.
Toughness
The ability of a material to resist the propagation of a crack under force
can be found by calculating the area under a stress-strain curve.
Transparency
Ability to let light be transmitted with minimal scattering allowing a clear view through material.
Young's Modulus
A measure of a material’s stiffness, defined as stress divided by strain.
high Young's modulus = material is very stiff (e.g., steel) and doesn't stretch easily
low Young's modulus = the material is flexible or elastic (e.g., rubber)
measured in Gigapascals (GPa)
Chemical Properties
Characteristics that describe how a material reacts with other substances, resulting in a change in its chemical composition
Corrosion Resistance
Reactivity (Food Safe)
Hygroscopy (Ability to retain moisture)
Flammability
Mass
the measure of the amount of matter
not affected by gravity - does not change until some matter is added or removed
measured in kg
Weight
The force on an object acted upon by gravity, which depends on the gravitational force, and the mass of the object
Measured in Newtons (N)
F = ma
Volume
the amount of space an object occupies
measured in m3
Electrical conductivity and resistivity
Electrical conductivity
a measure of how easily free electrons flow through a material.
Electrical resistivity
Measures how much a material resists the flow of electricity.
They are inversely related; meaning that when a material has high resistive properties, it is a poor conductor of electricity, and vice versa
Factors affecting conductivity: Resistivity is described in terms of the free flow of electrons, which can be caused by...
Collision with lattice imperfections in the material (could be caused by cold working)
Collisions with other atoms (e.g. impurities)
Collisions with thermally induced vibrations (temperature)
RWE: Gold plating is often chosen for sensitive electronic components due to its high conductivity/low resistance, and its low reactivity
Resistivity of Different Materials
Metals = good conductors
silicone and water = somewhere in between
plastics, ceramics and glass = good insulators.
Flammability
How quickly a material can be ignited, and how quickly it burns
Chemical Composition: Materials with high carbon content are often more flammable.
Surface Area: Finer particles or thinner materials ignite more easily.
Environmental Conditions: Oxygen levels and temperature affect flammability
Corrosion Resistance
The ability of a material to withstand damage caused by oxidization or other chemical reactions
Both steel and aluminum are used for vehicles, but steel needs a protective layer of paint to avoid corrosion.
Aluminium does from an oxide layer, but it protects it and doesn’t corrode further.
High humidity and salt increase corrosion rates
Reactivity (food safe)
The extent to which a material will react with another (including air) to become unsafe for use to come in contact with food.
Some plastics are food safe, others are not. You should not store water for long periods in a single-use bottle, as over time it will leach chemicals into the water
Copper is quite reactive, and will react with oxygen/acids/alkalis found in foods and cleaning fluids —> why copper pans are always lined with tin
Hygroscopy
The ability to absorb moisture from the surrounding environment.
Wood = absorbent material + needs protecting from wet rot, dry rot, and fungal/lice attack.
Not all timber products absorb moisture at the same rate.
MDF for example, is very absorbent and needs sealing and coating or it expands and becomes ineffective
Natural textiles eg. cotton readily absorb moisture
Synthetic fibres like nylon = much less absorbent.
This important distinction helps designers choose appropriate fabrics based on their intended use.
Thermochromism
Thermochromic materials undergo a reversible chemical reaction in response to a temperature increase.
Strength
a material's resistance to stress or force without bending or breaking
3 types of strength:
Tensile strength: the ability of a material to withstand pulling forces without breaking or deforming
Compressive strength: the ability of a material to withstand forces that try to squash or compress it without breaking or deforming
Shear strength: the ability of a material to withstand forces parallel to the material plane
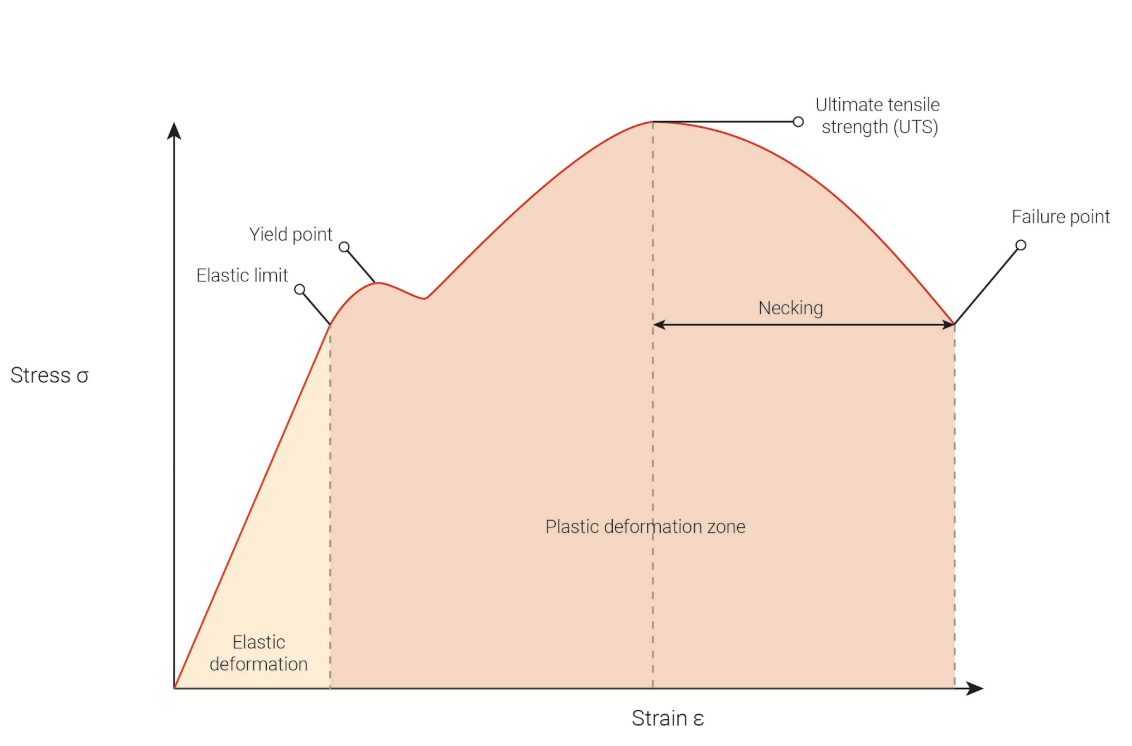
Young’s Modulus Graph
Stiffness
calculated by finding the gradient of the straight line at the start of the curve
stiffer material = steeper line
Elastic Limit
Any stress beyond this value will result in the material turning from elastic to plastic and will not return to its original shape
Ultimate tensile strength
the maximum force a material can endure
Necking
the localized thinning of a material before it breaks. Less force is required to continue to stretch the material
Failure point
the strain at which the material snaps
Stress
the force divided by the cross-sectional area. (Pascals)
Strain
a measure of how much the material stretches. It’s calculated by dividing the change in length by the original length. (no units)
Malleability
the ability of a material to be be plastically deformed under compressive forces
beaten, rolled or pressed into different shapes.
the material must display good amounts of plasticity to be malleable
General Properties of Metals
typically hard and shiny
high electrical and thermal conductivity
Metals in pure element form are usually either too soft, brittle, or chemically reactive for practical use within design. Understanding how to manipulate the properties of these metals is key to their success
Categories of Metals
Ferrous Metals: Ones which contain iron
Low carbon steel, wrought iron, mild steel, cast iron
Non-ferrous metals: Ones which do not contain iron
Aluminium, Titanium, Copper, Brass
Alloys: A mixture of two or more metals
Stainless steel, Iron-nickel
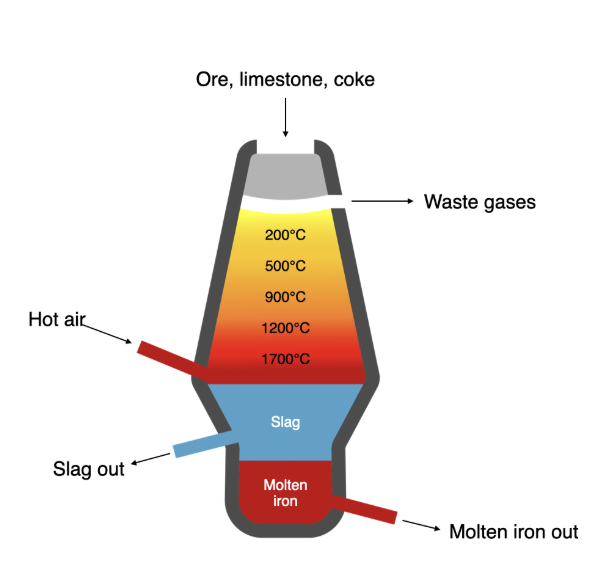
Smelting: Extracting Metal From Ore
The process of refining metal ores into usable metals
Metal is rarely found in its pure element form - it’s often chemically bonded with impurities of other elements, like oxygen.
To remove these other elements and extract the pure metal, a process like smelting is required
Smelting:
Iron ore is heated in a furnace with a reducing agent (often carbon).
The carbon reacts with the oxygen and you are left with a mix of iron and carbon.
The impurities bind with the carbon to produce waste called slag.
Iron Oxide (+impurities) + Carbon (reducing agent) → Steel (Iron + carbon) + Slag
Copper
excellent thermal and electrical conductivity.
Highly ductile and malleable
Used in:
electrical applications, cookware, pipes for plumbing
Aluminium
Aluminium & others (rarely used pure)
Highly formable
recyclable,
good corrosion resistance.
High strength to weight ratio + thermal conductivity
Used in:
electrical applications, cookware, pipes for plumbing
Stainless Steel
Iron, carbon, chromium & others
excellent corrosion resistance
similar mechanical properties to a range of low to high carbon steel
Used in:
general purpose metal work, medical utensils, cutlery
Wrought Iron
<0.08% Carbon
highly malleable,
soft,
lower strength and stiffness compared to other steels
Used in:
ironmongery, decorative metal work
Low-Carbon Steel (mild steel)
0.05 - 0.3% Carbon
highly malleable, ductile,
tough, stiff and strong
Used in:
general purpose metal work
High-Carbon Steel (tool steel)
>0.5% Carbon
very high strength, stiffness and hardness.
Lower ductility and toughness than mild steel
Used in:
cutting tools, vehicle chassis
Cast Iron
>2.5% Carbon
very stiffness and hardness.
Brittle, slightly higher thermal resistivity, and corrosion resistance
Used in:
cookware, manhole covers
Gold
aesthetic appeal,
high malleability,
excellent electrical conductivity,
high value
Used in:
electrical connections, jewellery, monetary purposes
Brass
copper, zinc & others
Highly formable, ductile, malleable.
Good corrosion and microbial resistance.
Low coefficient of friction
Used in:
musical instruments, couplings, hinges, gears, bearings, ammunition
Bronze
copper, tin & others
excellent corrosion resistance,
highly formable,
ductile and malleable
Used in:
ship propellers, currency, bearings and bushings,
Titanium
very high stiffness, strength, toughness, strength to weight ratio.
High operating temperatures.
Difficult to form, expensive
Used in:
Turbines, prosthetics, sports equipment
Lead
soft, high density, toxicity to life.
Most uses of lead have now been illegal due to its toxicity and danger to living things
Used in:
radiation shielding, bullets, car batteries
Tin
low melting point,
high formability,
high thermal and electrical conductivity,
good corrosion resistance
Used in:
soldering and plumbing joints, tinning of copper cookware and steel cans
Effect of Rate of Cooling on Metals
When a metal solidifies, it forms into grains (crystals). The crystalline structure formed will impact the properties of the material.
Cooling Quickly:
material will form a higher number of smaller crystals.
results in a material that is harder, stiffer and stronger, but brittle
Cooling slowly:
material will form a lower number of larger crystals.
results in a material that is softer, more ductile, more capable of plasticity
Metal Grain Size - Relationships of Properties
As metal grain size increases
ductility increases
strength + toughness decreases
Metal: Normalising
Heating up to a critical temperature and then left to cool in air.
reduces the grain size,
produces a more uniform structure,
relieves some internal stress within the material.
reduces hardness within the material.
Metal: Annealing
Heating up to a critical temperature and then being allowed to cool very slowly.
makes the metal soft and ductile,
reducing hardness.
often done to allow further cold working without risk of material fracture
Metal: Hardening
Heating up to a critical temperature of 800°C and then being quenched rapidly in water or oil.
This converts the crystalline structure and then cools it quickly, leading to the storage of internal stresses within the material.
Hardened steel is very difficult to abrade or cut
great for the edges of knives, or the shackles on padlocks
Tempering
Heating up to a low temperature (sub 500°C) for several hours, and being left to cool in air.
This process is done after hardening, and it is performed to bring back some of the ductility of a material and increase its toughness.
A padlock or knife or sword = requires both hardness and toughness, so they are nearly always tempered.
Advantages of Alloying of Metals
Base metals are rarely used in their pure form, because there are great benefits of alloying them with another metallic element
Advantages of Alloying Metals
An increase in yield or tensile strength
Increased resistance to thermal shock
Increased strength at higher temperatures
Increased corrosion resistance
Increased creep resistance
Superalloys
This group of metallic alloys have unique properties:
Retain high stiffness and strength at up to 70% of their melting point
Extremely resistant to corrosion from heat, moisture and chemicals
Creep and fatigue resistance
Excellent resistance to thermal shock
Superalloys all exhibit an Face-centered cubic (FCC) structure and fall into three main groups:
Iron-nickel based alloys
Nickel based alloys
Cobalt based alloys
Applications:
aircraft jet engines
gas turbines for power plants
used in specialized medical prosthetics, dental equipment
Timber
Timber is wood that has been harvested from trees and processed for use in manufacturing, construction, and carpentry.
renewable building material that uses the sun’s energy to replenish itself in a continuous cycle
Softwoods (from evergreen/coniferous trees)
Hardwoods (from deciduous trees)
Man-made
Whilst most of its production emits significantly less CO2 than steel or concrete, consideration needs to be given to the consequences of mass-deforestation.
Wood VS Timber
Wood = raw form (tree trunk)
Timber = wood turned into useable formats that designers and builders can use effectively.
Subtractive and lamination processes, rather than moulding are used to create parts.
Advantages of Timber
Good strength-to-weight ratio.
Can be machined, sanded, and finished easily.
Naturally insulating (thermal and acoustic).
Disadvantages of Timber
cannot be easily moulded; you need a piece of timber that has been cut from a piece of wood big enough for the part you are making.
Can be affected by moisture, insects, or rot if untreated
Natural timber ranks significantly higher in strength over manufactured timber, so why is manufactured board used so commonly?
Manufactured board can be processed into much larger sheets, which makes it ideal for making things out of. \
It can also be made very cheaply out of recycled scraps
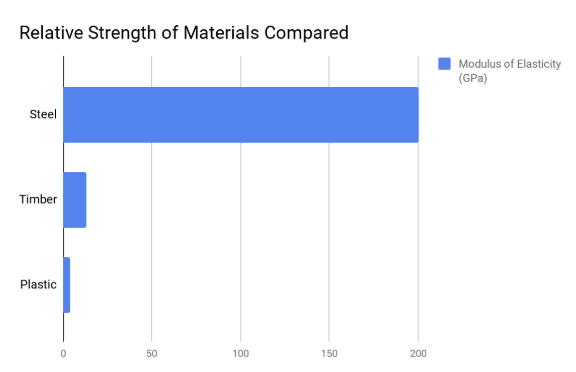
Strength of Steel VS Timber VS Plastic
The strength of most materials are dwarfed by that of steel, but due to its weight, expensive and energy intensive production process, we don’t see steel being used for everything.
Softwood
comes from coniferous trees
evergreen, needle-leaved and cone-bearing.
*the fibres of these trees are larger and allow for nutrient transport which allows for faster growth
Properties of Softwood
Rapid growth - reaching maturity in 30 years
grows in colder climates
Strength
Softwoods are not soft, and many have comparable tensile strength to hardwoods
Cheaper than hardwood:
Quicker to replenish, and can grow all year round in harsher climates means wood can be harvested and replaced quickly
Sustainability
The quicker growth rate allows for a more sustainable scale of production
Aesthetics
Softwoods have an attractive appearance and are often left bare to show off the natural grain of the wood
Not Moisture Resistant
All types of timber need to be protected from moisture, with softwood being no exception
Requires preparation
All natural wood needs to be seasoned; where the moisture content is reduced to below 20%. This provides many benefits, such as resistance to rot and fungi, increased strength, and better appearance.
Examples of Softwood
Douglas Fir
Siberian Larch
Scots Pine
Red Cedar
Hardwood
come from mainly deciduous trees or angiosperm trees which are seed or fruit bearing with broad leaves
*the presence of larger vessels that transport nutrients, with smaller fibres that do not
Properties of Hardwood
Hardwood trees take much longer to grow (up to 150 years to mature).
They can grow in the tropics, and in temperate climates, but not in very cold conditions
Strength
Hardwoods in general are stronger and denser than softwood, however exceptions like Balsa wood do exist
Cost
slow growth rate - significantly more expensive than softwood
Sustainability
Slow growth rate means that demand can easily outstrip the supply, making hardwood less renewable
Aesthetics
Hardwoods are a premium cost also due to their aesthetic appeal
Moisture Resistance
Hardwood can be more moisture resistant than softwood, but still need to be protected
Preparation
All natural wood needs to be seasoned; where the moisture content is reduced to below 20%. This provides many benefits, such as resistance to rot and fungi, increased strength, and better appearance.
Examples of Hardwood
Oak
Walnut
Cherry
Balsa
Man-Made Timber
manufactured by a series of industrial processes, where fibres, chips or layers are glued together
General Properties
cheaper than natural timber
larger sheet sizes than natural timbers
Susceptible to water damage unless protected
Easily machined from CAD
Very strong in both directions due to the lamination being perpendicular
Man-made timber: Plywood
Construction:
Layers called veneers laminated with the grain perpendicular to one another
Strength:
Very strong in both directions due to the lamination being perpendicular
Moisture resistance
Good- especially if using marine ply, but still needs to be treated and protected
Cost
Most expensive
Appearance
Comes in different grades, but generally looks appealing
Uses
Boats, furniture, flooring.
Its high strength + ability to be produced in large sheets = a good choice for many applications
Man-made timber: Particle Board (MDF/HDF)
Construction:
Fibres of recycled or scrap wood, pressed and glued together under immense pressure and heat
Strength:
Low stiffness and strength compared to plywood
Moisture resistance
Poor resistance to moisture, needs to be treated as it acts like a sponge
Cost
Fairly inexpensive
Appearance
Poor; never used in its natural state. Often veneered with PVC or hardwood
Uses
Used primarily for cheap mass produced furniture due to its ability to be machined easily and low cost
Man-made timber: Chipboard
Construction:
Fibres of recycled or scrap wood, pressed and glued together under immense pressure and heat
Strength:
High stiffness, low weight but not as strong as plywood
Moisture resistance
Poor resistance to moisture, can be protected but typically never used outside
Cost
Fairly inexpensive
Appearance
Poor, never used in its natural state. Often veneered with PVC or melamine
Uses
Similar to MDF, and used in kitchen counters, IKEA furniture and packaging blocks
Timber Treatment: Seasoning
reducing the moisture content of timber to make usable lumber
a drying process where the moisture content is lowered to below 20%.
prevents wood from rotting, shrinking, twisting or warpingover time.
can take place in an oven, or in a naturally dry environment.
Timber Treatments: General
Timber treatments are designed to protect the wood from insects or fungi which would attack and decompose the wood.
They penetrate the wood fibres and usually stain the wood a darker colour.
Fungal Decay - Wood Rot

Timber Finishes
applied to the surface of a wood and mostly sit on or near the surface rather than penetrate the wood fibres.
form a barrier which protects from moisture + are often used to improve the aesthetic quality of the wood
Types of Timber Finishes
Varnish
Forms a tough, protective barrier which often gives the wood a shiny and smooth appearance.
Several layers, with sanding in between are required for full effect.
Often used on boats to protect timber from the harsh environment of salt water
Wax
Often beeswax is used, dissolved into a solvent, and wiped on with a cloth.
Offers only low levels of protection as a barrier to moisture as there is no thickness compared to varnish.
Brings out the aesthetic grain of natural timber
Oil
Applied with a cloth, and forms a thin protective barrier to moisture.
The range and scope of different oils used for different purposes is wide
Mineral Oil (derived from petroleum)
Linseed Oil
Sunflower Oil
Tung Oil
Walnut Oil
Glass
A hard, brittle and typically transparent amorphous solid made by rapidly
cooling a fusion of sand, soda and lime
Glass General Properties
Hard
Brittle
High compressive strength
Low tensile strength
Transparent
Low thermal and electrical conductivity
Advantages of Glass
Non-porous and low reactivity
Glass doesn’t absorb liquids or react with most chemicals
can be wiped clean easily and, when manufactured under circumstances is resistant to changes in heat
chemical + culinary: used in chemical-resistant lab glassware (like beakers) and airtight jars for storing food.
Insulating
often has a layer of air between multiple layers which prevents the heat from escaping via conduction
Structural
widely used building material.
great aesthetic qualities, bringing light and warmth to a space,
its insulating properties keep heat trapped inside,
can support large forces and can be molded into precise and complex forms
Recyclable
Because glass isn’t easily fused to other materials + it melts, it can be easily separated and recycled.
It won’t decompose if dumped in a landfill, but also won’t leech chemicals.
Up to 95% of recycled glass can be used when making new glass, if no specialist properties are required
Flexible
With the right composition + specialist tooling, glass can be drawn out into tiny long fibres that are flexible.
used either for textiles to create fibreglass, or in a hollow form called optical fibre, which transmits the majority of digital data across nations in the form of light
Types of Glass
Annealed glass
Toughened glass
Borosilicate glass - copes well with thermal expansion and temperature change
Laminated glass
Annealed Glass
The most common and untreated type of glass.
Made by
heating silica (the primary component of sand) to a high temperature where it melts,
mixing with various compounds to alter its properties
slowly cooling the glass to relieve internal stress.
Weak —> breaks into large, sharp shards, which can be dangerous.
Used in:
Picture frames, regular drinking glasses, standard windows (where safety isn’t a big concern).