ANTH-3153: Arch Sci II final exam
1/202
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
203 Terms
What chemical elements are in organic matter?
carbon, nitrogen, oxygen, hydrogen, sulfur
What are the 4 macromolecules?
proteins, lipids, nucleic acids, carbohydrates
Which conditions preserve organics?
extremely dry, extremely cold, anoxic, wet, alkali pH
Which conditions do organics not preserve in?
alternation between wet and dry, acidic pH, active soil processes
When was raw material sourcing introduced?
1950s
When did aDNA become popular?
2010s
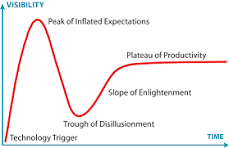
What is this?
Hype cycle
What are the 4 major components in bone?
mineral, protein, lipid, water
What percentage of bone is mineral?
70%
what makes up the mineral component in bone?
calcium (hydroxyapatite)
what makes up the protein component in bone?
mostly type I collagen
what percentage of bone is protein?
20%
what percentage of bone is lipid?
3%
what percentage of bone is water?
5%
what are the characteristics of bioapatite?
can decide strength of bone, very small crystal size, directly interacts with collagen fibrils
what makes up bioapatite?
Ca10(PO4)6(OH)2
What does carbonate commonly substitute for?
phosphate
how much of protein does type I collagen make up?
>90%
What is the structure of collagen?
a triple helix
What holds collagen chains together?
hydrogen bonds
how many amino acids in each helix of collagen?
about 1000
What is interesting about collagen’s amino acid composition?
every third residue is glycine
where is the only place you can find hydroxyproline?
in collagen
what is an isotope?
atoms of the same element that have a different mass from different number of neutrons
what do protons determine in an element?
the chemical properties
what do neutrons determine in an element?
the physical properties
what are isotones
elements with the same number of neutrons
what are isobars?
elements with the same mass
What is the symmetry rule for stability of isotopes?
for all elements lighter than Ca N=Z or N/Z is >1
why does the symmetry rule work?
protons repel each other and need more neutrons to stabilize
what is the oddo-harkins rule? (even vs. odd)
the greatest stability in isotopes is when the Z# and N# are even
According to the oddo-harkins rule, what combination would be the least stable?
odd # of Z and N
How many isotopes do lighter elements tend to have?
2
how do isotopes differ from each other?
strength of bonds with other elements will be stronger or weaker depending on isotopes involved
what isotopes react more quickly?
lighter isotopes
which isotopes have stronger bonds?
heavy isotopes
what is fractionation?
partitioning of isotopes between two substances or phases of the same substance with different isotope ratios
what are the types of fractionation?
equilibrium and kinetic
what is kinetic fractionation?
when heavy isotopes want to be in the more stable phase
will light isotopes go into a more or less stable phase?
less stable
do heavier elements have more or less fractionation?
less fractionation
what kind of measurement is the delta value? (δ)
a relative measurement
what is δ?
represents the relative difference in the ratio of stable isotopes compared to an international standard
how is δ expressed?
per mil ‰
what kind of δ13C values are in organic material?
negative values
what kinds of δ15N values are in organic material?
positive values
what are the stable isotopes of hydrogen?
1H and 2H
what percentage of all hydrogen does 1H make up?
99.9%
what percentage of all hydrogen does 2H make up?
0.156%
what are the stable isotopes of oxygen?
16O, 17O, and 18O
what percentage of all oxygen does 16O make up?
99.6%
what percentage of all oxygen does 17O make up?
0.03%
what percentage of all oxygen does 18O make up?
0.19%
which oxygen isotope would most likely go into liquid phase when ice melts?
O16
which oxygen isotope would prefer to stay in the ice phase when in water?
18O
when rain falls, would 18O rather be in the rain or vapor from the cloud?
rain
are δ18O values of precipitation higher or lower in the winter?
lower
where are ice cores derived from?
glacial ice formed over 1000s of years
what is an isotope paleothermometer?
a way to get the known relationship between temperature and isotope values of material
what is meteoric water?
water that has fallen from the sky
where does most precipitation happen?
over oceans
what are the mechanisms that control the δ18O and δ2H of meteoric waters?
latitude, cloud temp, altitude, proximity to coast, amount effect
When a cloud rains, what isotope do we lose more?
heavy isotope
when latitude increases, what does the δ18O precipitation do?
decrease
when is ΔT (source-precip) lower?
the summer
when altitude increases, what does δ18O precipitation do?
decreases
where does the amount effect apply?
the tropics
when precipitation amounts increase, what happens to δ18O?
it decreases
if there are gentle tropic rains, what happens to the δ18O?
the values get higher
if there are heavy tropic rains, what happens to the δ18O?
the values get lower
what is phenylalanine?
an essential amino acid that does not come from water
where does the oxygen come from in phosphate and carbonate?
the water we drink
what does carbonate often substitute in for?
phosphate
what is the best source of carbonate to analyze?
tooth enamel
why is tooth enamel good for carbonate?
not porous, little organic matter to degrade, the minerals are large
what is the best source of phosphate to analyze?
tooth enamel
why is tooth enamel good for phosphate?
very hard to alter, lasts millions of years, very stable
what is a not good source of carbonate to analyze?
bone
why is bone not good for carbonate?
porous, lots of organic material, crystals small, prone to alteration/contamination
why is tooth enamel valuable for oxygen values?
O values are locked in from the time or formation
what are the most analyzed proteins in archaeological contexts?
collagen and keratin
where are CNS from the body derived?
CNS in foods we consume
when is contamination a problem?
when the contaminant isotope is really different from the target compound
what is degradation?
loss of target compound
which kind of amino acid is subject to fractionation during synthesis?
non essential amino acids
what are trophic discrimination factors?
offsets that represent the change in stable isotope ratios between diet and tissues
what do we need for diet reconstruction?
good tools, no alteration in burial, need to know the relationship between analyzed tissue and food isotopic compositions, turnover rate, routing, and isotope values of food consumed
what is routing?
the preferential incorporation of elements from certain macromolecules into certain tissues
what element is routing a problem for?
carbon
how does routing work?
the 3 macros all have carbon in them and the carbon gets directed towards particular tissues. This makes it nearly impossible to know which macro the carbon came from
what is the best source of baseline isotopic composition values?
materials from ancient contexts
What happens if two foods have the same isotopic composition?
you cannot tell which is more important in the diet
what are example of C3 plants
trees, moss, ferns
what are examples of C4 plants?
tropical grasses, sugar cane, millet
do marine animals have higher or lower δ13C and δ15N values than terrestrial animals?
higher
how many trophic levels in marine life?
5
how many trophic levels in terrestrial life?
3
which chemical element has the most stable isotopes?
tin
how many stable isotopes does tin have?
10
which one of tin’s stable isotopes are the most abundant?
Sn 120