Vibrational Spectroscopy
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
What is vibrational spec
Vibrational spec is the study of how EM radiation interacts with a molecule to change its vibrational energy
Atoms in a molecule vibrate around their equilibrium positions, and the bonds act like springs
How do we observe vibrations of bonds
Apply classical mechanics to them, Hooke’s Law
Give Hooke’s law to describe bond vibrations, stating each coponant
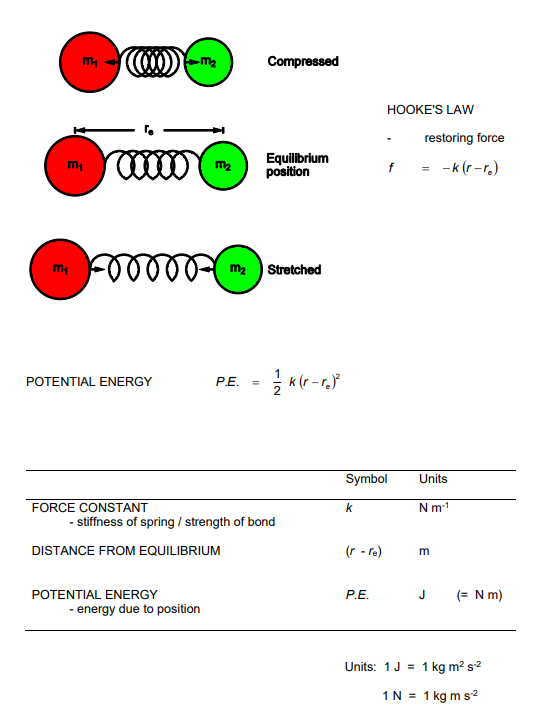
Draw a graph of energy against bond length for a simple harmonic oscillator, stating energies at equilibrium and turning points
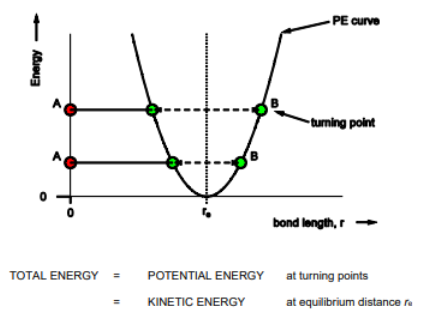
Draw graph of harmonic oscillators with big vs small k values
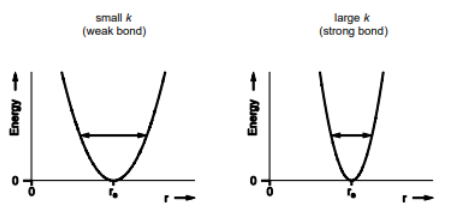
Give equation for Vibrational frequency
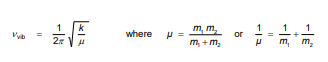
Give equation for vibrational wave number, giving units and definitions for each part

What does quantized and moderately spaced refer too
Molecule can only vibrate with discrete amounts of energy, meaning they are quantized
Molecules have moderate differences in energy between different vibrational levels. They are moderately spaced meaning we need to look at photons of higher energy looking at the infrared region rather than microwaves
What are diatomic harmonic oscillators
Refers to diatomic molecules that obey Hooke’s Law
Give equation for the energy of vibrational levels, describing all components and their units

Give the three different variations for the EV equation

What does quantum theory tell us of Vibrational energy levels
quantum theory tells us that vibrational energy levels are quantized
Vibrational Quantum Number defines the permitted energy levels Ev, in general, for all molecules
Vibrational Wave number determines the actual energy levels for a specific molecule
Draw a graph of energy against bond length displaying energy levels of a harmonic oscillator
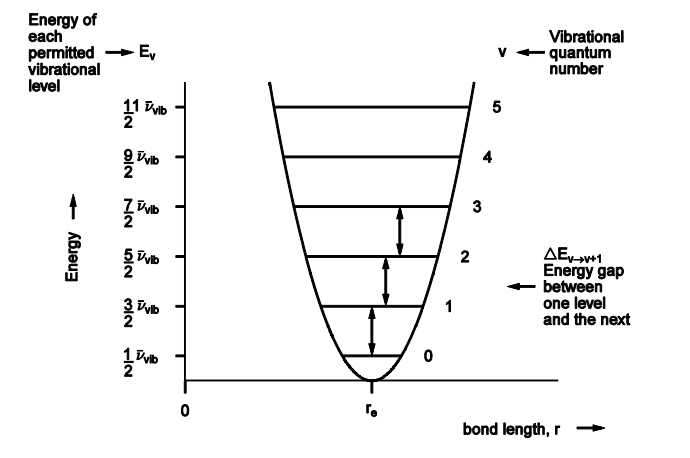
What is the lowest level for v and what does this mean for the molecule
Lowest level for v = 0, is E0 = ½ x Vibrational wave number,
This will still have a numerical value, as seen on the graph previously meaning we can see here that the atoms are always moving's, they are not permitted to have 0 vibrational energy
Give equation for Vibrational wavenumber describing each component and giving units, state what it is dependant on.
Constant for a specific molecules
Depends on the force constant and mass

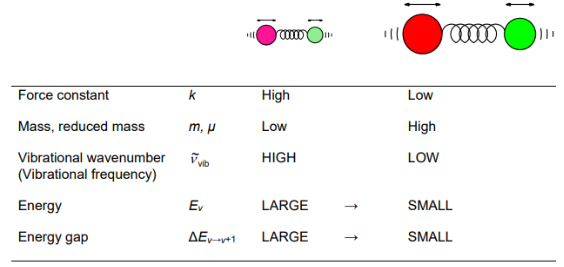
Consider relationship between the Force constant and mass of molecules, what this means for energy and wavenumber
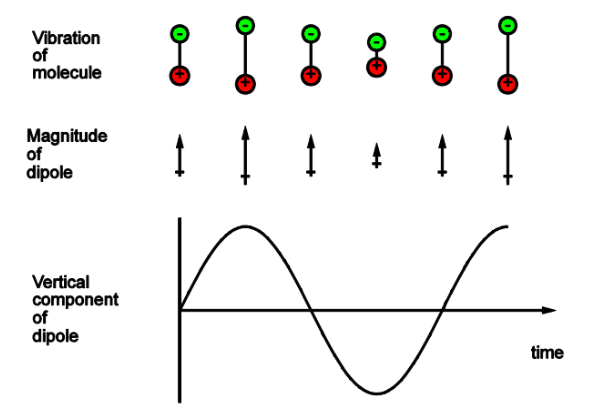
Describe the Gross selection rule, draw diagrams displaying this
For absorption or emission, there must be a change in dipole on vibration
The oscillating electric field needs to interact with the molecule and to let it do so its required to interact with the molecules oscillating dipole and it vibrates
Describe the specific selection rule
Delta V = + - 1
Transitions only allowed to next level.
Describe vibrational transitions
Vibrational energy levels are moderately spaced
most molecules are in the lowest level, v = 0, at room temperature
only ONE transition can occur, to v = 1
by absorbing a photon ONLY of energy equal to the gap ΔEv = 0→1
a vibrational absorption spectrum shows ONE band
What does an infrared spectrum show
That a molecule has a change in dipole on vibration
What is a diatomic vibrating rotor
A diatomic that is both vibrating and rotating
Describe how vibrational and rotational energies are observed together
Because the vibrational and rotational energies are very different from each other.
As such, the two types of motion can be considered to occur independently of each other, the total energy is the sum of the individual energies
Give the BORN-OPPENHEIMER approximation
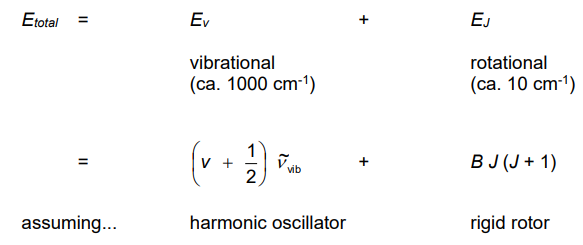
Give the general equation for EVJ
ΔEv,J = v~obs = v~vib ± 2 Bm
What is a diatomic anharmonic oscillator
Not a harmonic oscillator
At high vibrational energies, the bond lengthens more
What does quantum mechanics tell us about the anharmonic oscillators energy
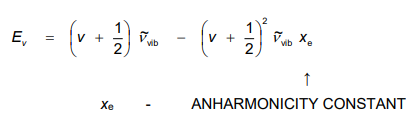
What is the selection rule for anharmonic oscillators
Selection rule relaxed to: Δv = ±1, ±2, ... but Δv = +1 still the strongest band
Describe how vibration differs between diatomic and polyatomic
For diatomics the bond can only stretch and compress. (One mode of vibration)
For polyatomics the modes of vibration differ and are calculated by 3N
As a molecule can contains N atoms, and there are 3 positions each atom can be specified by
It can move by translation (X,Y,Z)
It can move by rotation
It can move by vibration
How do you calculate number of vibrational modes in polyatomics
3N - (modes of rotation + modes of translation)
Name the two main types of vibrations
Stretching vibrations
Bending vibrations
Give the two types of vibration for when a molecule contains similar or identical groups
Vibrations do not occur independently, they couple to give
Symmetric vibrations (In-Phase)
Asymmetric vibrations (Out-Phase)
What do normal modes of vibration refer to, what does this mean for frequency and vibrations.
Normal modes of vibration refer to when :
All atoms oscillating synchronously
All atoms passing through their equilibrium position at the same time
They occur all the time in this state
Each of these vibrations are independent from each other
Each will also have its own specific frequency and as such each of the normal modes of vibration will generate its own specific band in the spectrum
What does a transition being IR active mean
The transition is allowed
There is a chance in dipole upon vibration giving an IR band
The large the change the stronger the band
Describe why we prefer either the condensed or the gas phase for rotational spectroscopy
Gas Phase
Preferred for more high resolution studies, this is due to its free rotation meaning there is rotational fine structure rather than just vibrational data
Condensed Phase
Much simpler spectra
Band positions may be shifted
Not free rotating
Hydrogen bonding causes a broad band due to forming many different structures and interactions
No fine structure
what are skeletal vibrations, what region do they occupy
Skeletal vibrations
involve SEVERAL or ALL atoms within a molecular skeleton
bands may occur in the FINGERPRINT region at ca. 700 - 1500 cm-1
characteristic pattern for "fingerprint" identification
What are group vibrations
Group vibrations
are LOCALISED on a specific bond or group
give bands at a SIMILAR wavenumber regardless of the molecule
Excellent for identifying specific groups