ANS 220 Exam 4
1/95
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
96 Terms
Compare and Contrast male and female gametes
Female
1 oocyte
Largest cell in the human body (120 to 150 microns)
Male
Billions of Sperm
Smallest cell in the human body (2.5 to 3.5 microns)
Quantity over quality
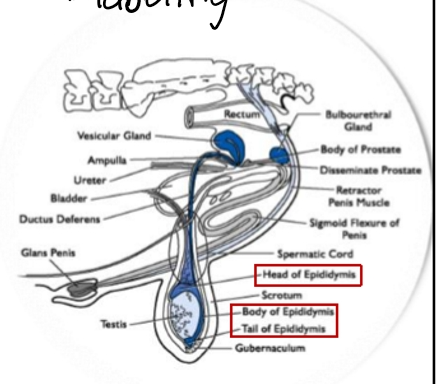
Male reproductive tract
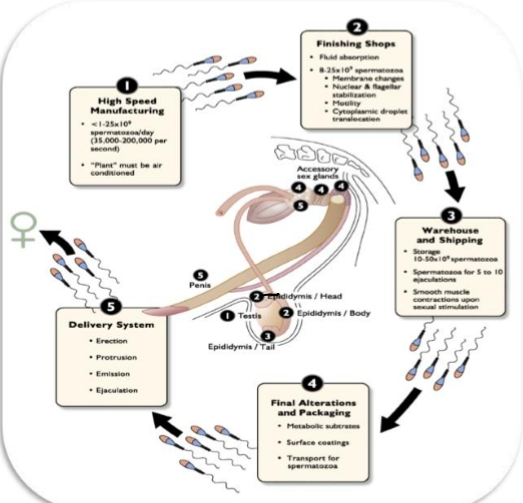
Overall Pathway of Sperm
Seminiferous tubules (testis)
Sperm are produced here
Temperature must be 4-6 degrees cooler than body temp
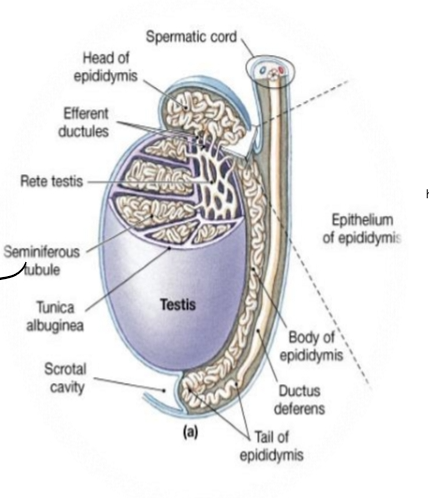
Rete testis → Efferent ducts → Epididymis
Epididymis= motility + fertility acquisition
Also, the storage warehouse for sperm
Vas deferens → Pelvic urethra
Here sperm encounter seminal plasma
Penile urethra → Glans penis
Delivery system for ejaculation
Seminal Plasma
Acts like bubble wrap for sperm
Provides:
Protection
Nutrients
Stabilization for travel through female tract
Testicle
Seminiferous tubule
Sertoli cells
“Nurse cells” supporting developing sperm.
Shape varies depending on surrounding germ cells
Leydig cells
outside the tubule
Produce testosterone
Basement membrane
Protective outer layer of each tubule
Capillaries
Provide nutrients and blood supply
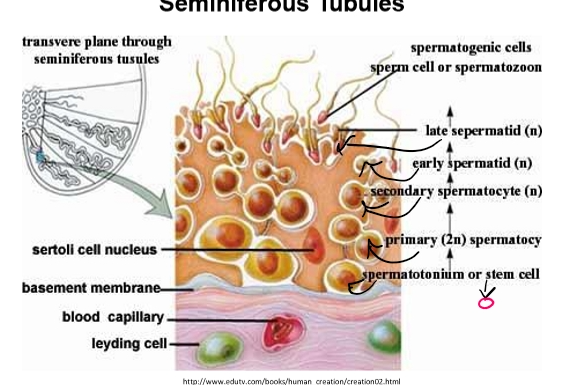
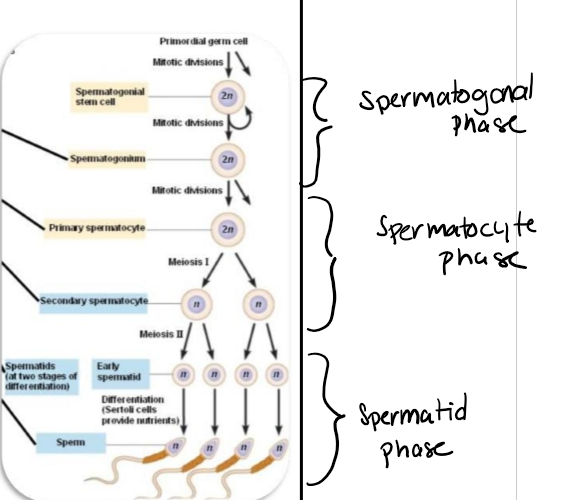
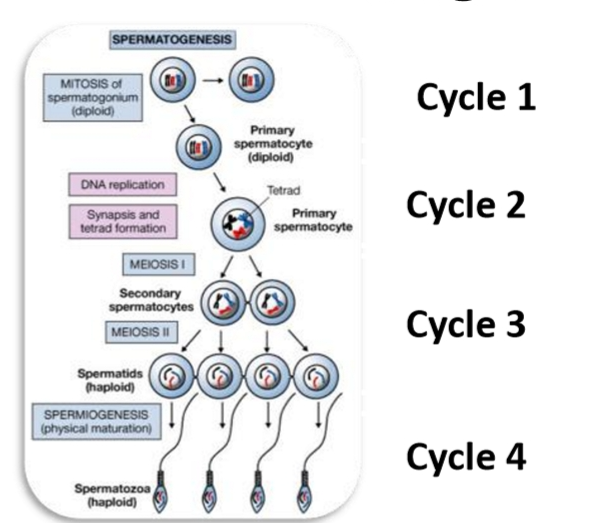
Spermatogenesis overview
Stem cells
Located along the basement membrane.
Called spermatogonial stem cells (SSC).
Two simplified types:
Type A = undifferentiated
Type B = differentiated
Progression of germ cells
Type A SSC → Type B SSC
Primary spermatocyte
Secondary spermatocyte
Spermatid (round)
Elongated spermatid → Spermatozoa
Movement
As cells mature, they migrate toward the lumen of the seminiferous tubule.
Cell division
Mitosis occurs early (stem cell replication).
Meiosis occurs during transition from primary → secondary spermatocytes → spermatids.
Function of Epididymis Functions
Gain motility
Gain fertility
Storage until ejaculation
Two important cells
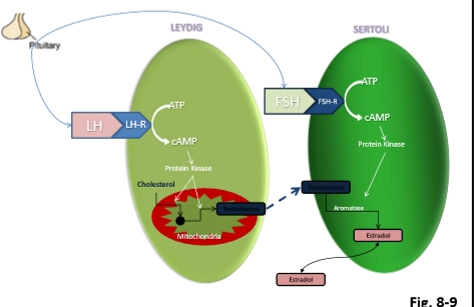
Intersitial cells (LEYDIG)
Produce testosterone
Location
Sit outside the seminiferous tubules, in the interstitial space.
Function
Produce testosterone — absolutely essential for spermatogenesis.
Equivalent to theca cells in the female.
Hormonal Regulation
Stimulated by LH from the anterior pituitary.
LH binds to receptors on Leydig cells → activates steroidogenesis.
Converts cholesterol → testosterone.
Why testosterone matters
Must be very high inside the seminiferous tubule for sperm production.
Drives:
Spermatogenesis
Sertoli cell function
Secondary sex characteristics
Negative feedback to hypothalamus/pituitary
Sertoli cells
Nurture germ cells through development
Produce numerous steroids
Location
Inside the seminiferous tubules, directly contacting developing germ cells.
Female Equivalent
Granulosa cells (closest to gametes, nurture them).
Functions
Sertoli cells produce:
Estrogen (via aromatization of testosterone)
Inhibin
Anti‑Müllerian Hormone (AMH / MIH)
Growth factors
Androgen Binding Protein (ABP)
They:
Support and nourish germ cells
Regulate the microenvironment
Control progression from spermatogonia → spermatozoa
Form the blood‑testis barrier
FSH Regulation
Sertoli cells express FSH receptors.
FSH stimulates:
Estrogen production
Inhibin production
ABP production
Growth factor secretion
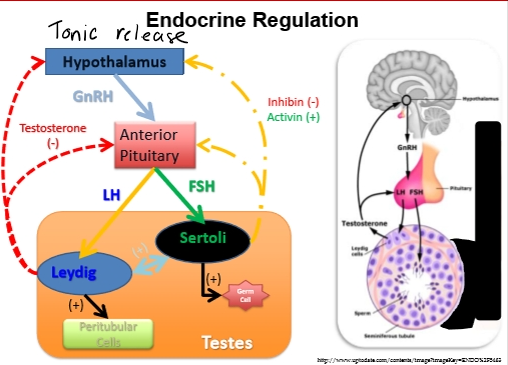
Endocrine Regulation
Male doesn’t have surge center only tonic pulses
Key difference from females
Males do not have a surge center.
All hormone release is tonic (steady pulses).
Hormone flow
Hypothalamus → GnRH (tonic pulses)
Anterior pituitary →
LH (stimulates Leydig cells → testosterone)
FSH (stimulates Sertoli cells → ABP, estrogen, inhibin)
Testis →
Testosterone (negative feedback)
Estrogen (local mitosis)
Inhibin (FSH regulation)
Pulsatility
GnRH pulses → LH pulses → testosterone pulses.
Testosterone rises ~3 hours after each GnRH pulse.
Over a full day, testosterone looks “flat,” but actually oscillates.
Androgen Binding Protein
Produced by
Sertoli cells (in response to FSH).
Function
Binds testosterone inside the seminiferous tubule.
Keeps testosterone concentrations extremely high locally.
Prevents testosterone from diffusing out of the tubule.
Why this matters
Without ABP → testosterone would escape → spermatogenesis would fail.
ABP is like a “testosterone magnet” ensuring the tubule stays saturated.
4. Estrogen in the Testis — Not Just a Female Hormone
Produced by
Sertoli cells (aromatizing testosterone).
Function
Stimulates mitosis of germ cells.
Drives:
Type A → Type B spermatogonia
Type B → primary spermatocyte
Key point
Estrogen is required for the mitotic expansion phase of spermatogenesis.
Inhibin and Anti-Mullerian Hormone
Inhibin — FSH Regulator
Produced by
Sertoli cells.
Function
Negative feedback to anterior pituitary.
Decreases FSH secretion.
Helps fine‑tune Sertoli cell activity.
Anti‑Müllerian Hormone (AMH / MIH)
Produced by
Sertoli cells (even in adults).
Function
In development: regresses Müllerian ducts.
In adults: helps regulate differentiation of spermatogonial stem cells.
Pre-pubertal vs Post-pubertal Steroid Production
Before puberty
Primary steroid = androstenedione
Testis is not fully activated.
After puberty
Primary steroid = testosterone
Due to activation of Leydig cells by LH.
Why High Testosterone Is Required Inside the Tubule
Spermatogenesis cannot occur unless testosterone is:
High
Local
Bound by ABP
Testosterone drives:
Meiosis
Spermiogenesis
Sertoli cell function
Endocrine Functions; What do the hormones do?
Leydig cells
Respond to LH
Product androstenedione (prepubertal)
Testosterone required for spermatogenesis
Sertoli cells
Responds to FSH
Produces:
Estrogen
Inhibin
AMH
Various growth factors
Androgen Binding Protein
MIH differentiation
Inhibin regulates FSH
ABP maintains high levels of testosterone
Estrogen causes the changes of sperm and simulates Mitosis
Summary of Hormone Functions
Hormone | Produced By | Target | Function |
LH | Anterior pituitary | Leydig cells | Testosterone production |
FSH | Anterior pituitary | Sertoli cells | ABP, estrogen, inhibin, growth factors |
Testosterone | Leydig cells | Sertoli cells + germ cells | Spermatogenesis, negative feedback |
Estrogen | Sertoli cells | Germ cells | Stimulates mitosis |
Inhibin | Sertoli cells | Anterior pituitary | Decreases FSH |
AMH/MIH | Sertoli cells | Germ cells | Differentiation |
ABP | Sertoli cells | Seminiferous tubule | Concentrates testosterone |
Why the Seminiferous Tubule Produces So Many Sperm
Tubules are extremely long and tightly coiled.
Spermatogenesis occurs along the entire length.
Continuous tonic hormone pulses maintain constant production.
2-cell, 2- Gonadotropin Model
Leydig Cells = Theca Cells
Stimulated by LH
Produce testosterone
Located outside the seminiferous tubules
Sertoli Cells = Granulosa Cells
Stimulated by FSH
Directly contact germ cells
Produce:
Estrogen
Inhibin
AMH/MIH
Growth factors
Androgen Binding Protein (ABP)
Key point
Both LH and FSH are essential for spermatogenesis.
Without either → sperm production collapses
Spermatogenesis
Human spermatogenesis takes ~70 days
From spermatogonial stem cell → spermatozoa
Species differences:
Bull: ~60 days
Ram: ~48 days
Why this matters
If the testis is injured (heat stress, fever, trauma), you won’t see infertility immediately.
Instead:
Damage today → infertility 60–70 days later
Because the injury affects early-stage spermatogonia, not mature sperm already stored in the epididymis.
Clinical example
A bull spikes a fever for several days
His sperm output looks normal right now
But 60 days later, sperm count + quality drop sharply
This is why reproductive managers track:
Heat stress events
Illness
Environmental temperature
Scrotal insulation
3. Puberty and Onset of Spermatogenesis
Before puberty
No sperm production
Hypothalamus–pituitary–testis axis is not fully connected
Primary steroid = androstenedione
After puberty
GnRH pulses activate LH + FSH
Leydig cells begin producing testosterone
Sertoli cells begin supporting germ cells
Spermatogenesis begins and continues for life
4. Spermatogenesis Occurs in Waves
Your professor emphasized this pattern.
Waves
Along the length of a seminiferous tubule, different sections are at different stages of development.
This ensures continuous sperm output.
If you “straightened out” the tubule, you’d see:
Region A: releasing sperm
Region B: meiosis
Region C: spermatogonia dividing
Region D: spermiogenesis
Why waves matter
Prevents “batch production”
Ensures daily sperm release
Allows billions of sperm to be produced per day
Continuous production = billions per day
Spermatogenic Cycles
Definition
A “cycle” = the repeating pattern of cell associations within a segment of the seminiferous tubule.
Why cycles matter
They help identify:
Where damage occurred
What stage is disrupted
How long recovery will take
⭐ 6. The 3 Major Phases of Spermatogenesis
Your professor breaks spermatogenesis into three phases, based on cell type and type of division.
Spermatogenesis continued
Onset- puberty
Hormonal requirements
LH, FSH, testosterone regulation
Occurs in waves and cycles
Three phases
Spermatogonia phase
Proliferation, renewal and differentiation
mitosis
Spermatocyte phase
Meiosis
Spermatid phase
Final maturation steps
1 spermatogonium (stem cell)= 256 spermatozoa
Phases of Spermatogenesis
Phase 1: Spermatogonial Phase (Mitotic Phase)
Location
Along the basement membrane
What happens
Spermatogonial stem cells (SSCs) proliferate
Type A SSCs:
Self‑renew
Maintain the stem cell pool
Type B SSCs:
Differentiate
Commit to meiosis
Division type
Mitosis
Purpose
Expand the pool of germ cells
Prepare cells for meiosis
Phase 2: Spermatocyte Phase (Meiotic Phase)
Location
Middle region of the seminiferous tubule
What happens
Primary spermatocytes undergo meiosis I
Secondary spermatocytes undergo meiosis II
Haploid spermatids are formed
Division type
Meiosis
Purpose
Reduce chromosome number
Create genetic diversity
Phase 3: Spermatid Phase (Spermiogenesis)
Location
Near the lumen
What happens
Round spermatids → elongated spermatozoa
No mitosis or meiosis (already haploid)
Major structural changes
Nuclear condensation
Acrosome formation
Tail formation
Mitochondrial rearrangement
Cytoplasm reduction
Purpose
Final maturation
Create a functional sperm cell capable of fertilization
Massive Amplification: 1 Stem Cell → 256 Sperm
Your professor highlighted this as “impressive.”
Why so many?
Each spermatogonium undergoes:
Multiple mitotic divisions
Meiosis
Spermiogenesis
Result
1 spermatogonial stem cell → 256 spermatozoa
Multiply this across:
Millions of SSCs
Entire length of the seminiferous tubules
Continuous waves
→ Billions of sperm produced daily
Why Mammals Are the Focus
Spermatogenesis in mammals is highly organized into:
Waves
Cycles
Phases
Fish, reptiles, and other vertebrates have different patterns
This course focuses on mammalian reproduction
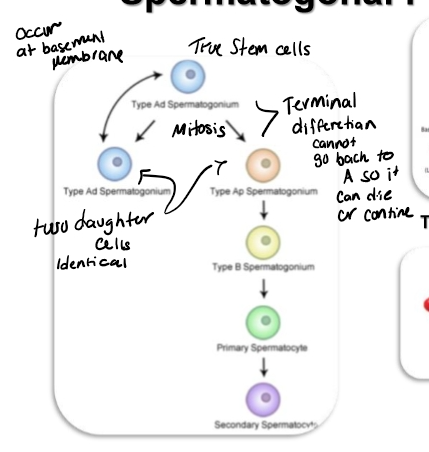
Spermatogonial Stage
ype A Spermatogonia = TRUE Stem Cells
Located along the basement membrane.
Capable of self‑renewal:
One Type A divides → two identical Type A daughters.
This maintains the stem cell pool for life.
This is why males do not undergo reproductive senescence like females.
Type A Can Also Differentiate
Type A → Type B spermatogonium
This is a terminal differentiation:
Once a cell becomes Type B, it cannot revert to Type A.
It is now committed to entering meiosis.
Type B Spermatogonia
Still diploid (2n).
Only two fates:
Become a primary spermatocyte, OR
Undergo apoptosis (if something goes wrong)
Stem Cell Niche
Type A spermatogonia survive only when attached to their niche along the basement membrane.
Removing them from this niche → they lose stem cell properties.
Research (like Dr. Oakley’s lab) shows:
The niche provides signals that maintain “stemness.”
Without it, they differentiate or die.
Intercellular Bridges Keeping cells in sync
Unique Feature of Spermatogenesis
Developing germ cells remain connected by intercellular bridges (called cytoplasmic bridges or syncytia).
These bridges:
Keep all linked cells at the same developmental stage.
Ensure synchronized mitosis and meiosis.
Allow sharing of nutrients, mRNA, and regulatory molecules.
If a cell loses its bridge
It becomes “out of sync.”
The body triggers apoptosis.
This is why one stem cell can produce 256 sperm — but often produces fewer.
Mitosis → Meiosis
Mitosis (Spermatogonial Stage)
Type A → Type A (renewal)
Type A → Type B (differentiation)
Type B → Primary spermatocyte
Primary Spermatocytes
Large cells.
Enter Meiosis I.
⭐ 4. Spermatocyte Stage — Meiosis Begins
Primary Spermatocytes → Secondary Spermatocytes
Meiosis I occurs.
Chromosomes recombine and segregate.
Cells become genetically unique.
Blood–Testis Barrier (BTB) Importance
Primary spermatocytes must cross the tight junctions between Sertoli cells.
BTB protects them because:
Once meiosis begins, they are no longer genetically identical to the rest of the body.
The immune system would otherwise attack them as “foreign.”
BTB prevents immune cells from entering the adluminal compartment.
Secondary Spermatocytes
Undergo Meiosis II quickly.
Meiosis II → Haploid spermatids (1n).
Numbers to Know
1 Type A stem cell →
2 Type A daughters →
4 Type B →
8 primary spermatocytes →
16 secondary spermatocytes →
256 spermatids
Spermatid Stage
Key Point
NO mitosis or meiosis here.
Spermatids are already haploid.
What happens
Round spermatid → elongated spermatozoon
Changes include:
Nuclear condensation
Acrosome formation
Tail (flagellum) formation
Mitochondrial migration to midpiece
Cytoplasm reduction
This stage = Spermiogenesis
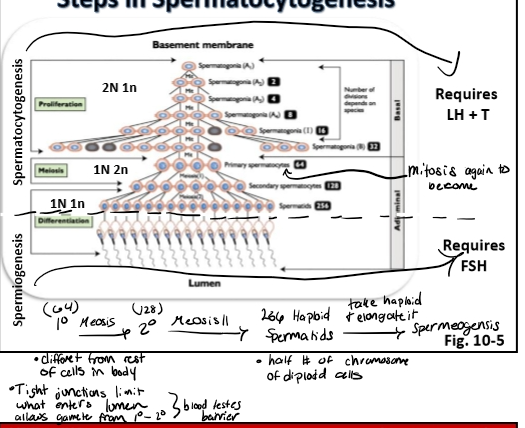
Spermatocytogenesis vs Spermiogenesis
Spermatocytogenesis
Includes:
Mitosis (Type A → Type B → primary spermatocyte)
Meiosis (primary → secondary → spermatid)
Requires:
LH → Testosterone
Blocking LH = blocking testosterone = no spermatocytogenesis
→ This is how chemical castration works.
Spermiogenesis
Round spermatid → elongated spermatozoa
Requires:
FSH → Sertoli cell activation
Blocking FSH = no spermiogenesis
→ Round spermatids accumulate but do not mature.
⭐ 7. Hormonal Control Summary
Process | Requires | Why |
Spermatocytogenesis (mitosis + meiosis) | LH + Testosterone | Drives proliferation + meiosis |
Spermiogenesis (final differentiation) | FSH | Sertoli cell support + remodeling |
⭐ 8. Key Cell Stages to Memorize
Type A spermatogonia (true stem cells)
Type B spermatogonia (committed)
Primary spermatocyte (Meiosis I)
Secondary spermatocyte (Meiosis II)
Spermatid (haploid, round)
Spermatozoa (elongated, mature)
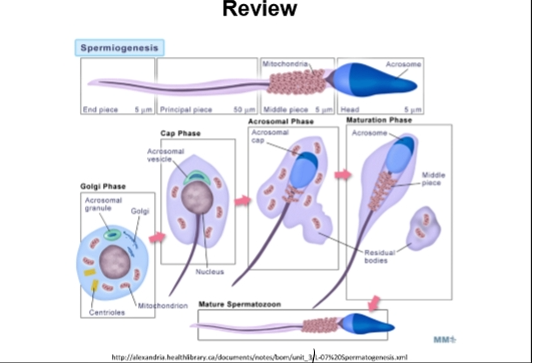
Spermiogenesis
Maturation and metamorphosis of a spermatid into a spermatozoan
Golgi phase
Cap phase
Acrosome phase
Maturation phase
Happens inside the seminiferous tubule, at the very tip of the Sertoli cell, right next to the lumen.
Sertoli cells act as nurse cells, physically supporting the spermatid as it elongates.
This is the final step before sperm are released into the lumen (spermiation).
⭐ 2. Hormonal Requirement
Spermiogenesis requires FSH
FSH stimulates Sertoli cells.
Sertoli cells provide:
Growth factors
Structural support
Enzymes
Nutrients
Blocking FSH → round spermatids cannot elongate → no functional sperm.
Contrast
Earlier stages (mitosis + meiosis) require LH + testosterone.
Final differentiation requires FSH.
⭐ 3. The Four Phases of Spermiogenesis
Your professor emphasized these must occur in order:
G → C → A → M
Golgi → Cap → Acrosomal → Maturation
Mnemonic from your professor: “Going Crazy And Mad.”
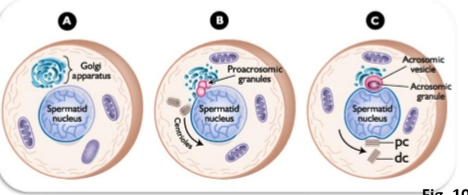
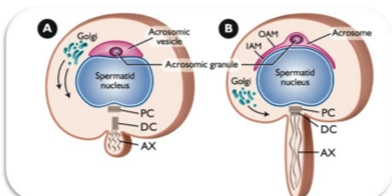
Golgi Phase
Acrosome vesicles forms
Golgi saccules come together and coalesce
Key Events
Round spermatid with:
Nucleus
Mitochondria
Organelles
Large Golgi apparatus
Main Transformation
Golgi forms the acrosome.
Acrosome = vesicle filled with digestive enzymes needed for fertilization.
What to memorize
Acrosome originates from the Golgi.
This is the defining event of the Golgi phase.
Cap Phase
Key Events
The acrosome spreads over the nucleus, forming a “cap.”
The nucleus begins to polarize (one side becomes the head region).
Tail (axoneme) begins forming from the centrioles.
What to memorize
Acrosome spreads over the nucleus → forms the acrosomal cap.
Key Events
The acrosomal vesicle (formed in the Golgi phase) now:
Moves to the top of the nucleus
Begins to spread over the nuclear surface
The round spermatid now looks like a mushroom (cap sitting on a round head).
Other events
Centrioles and other organelles begin to migrate and align.
These centrioles will later form:
The axoneme (core of the tail)
The base of the flagellum
What to memorize
Cap phase = acrosome sits on nucleus + begins spreading.
Tail precursors begin organizing.
Acrosome Phase
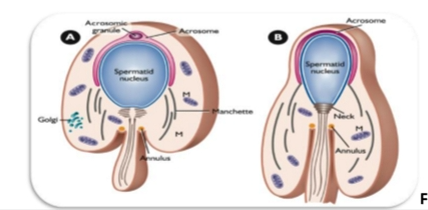
Nuclear & Cytoplasmic elongation
Manchette (centrioles) migrate to opposite end of nucleus
Acrosome wraps around nucleus
Key Events
Nucleus elongates and begins to condense.
Acrosome continues to enlarge and cover more of the nucleus.
Tail elongates further.
Spermatid rotates so the acrosome faces the basement membrane and the tail points toward the lumen.
What to memorize
Nuclear elongation + acrosome expansion + tail growth.
This is where the spermatid stops being round and starts becoming a sperm.
Key Events
Cell elongation begins
Both the nucleus and cytoplasm elongate.
This is the first major shape change.
Centrioles migrate
Move to the opposite end of the nucleus from the acrosome.
Begin forming the tail (flagellum).
Mitochondria begin migrating
Move toward the neck region where the midpiece will form.
Acrosome fully wraps around the nucleus
Covers ~⅔ of the nucleus.
This is essential for fertilization.
What to memorize
Acrosomal phase = elongation + tail formation + acrosome wrapping.
Maturation Phase
Final assembly & elongation
Acrosome completely wraps around nucleus
Mitochondria wrap around centriole in helical fashion
Key Events
Final shaping of the sperm head.
Excess cytoplasm is removed (forms the residual body).
Mitochondria migrate and wrap around the proximal tail → forming the midpiece.
Tail becomes fully functional.
What to memorize
Cytoplasm removed, mitochondria form midpiece, tail becomes motile.
Key Events
Final shaping of the sperm head
Nucleus becomes extremely condensed.
Acrosome finishes wrapping.
Mitochondria wrap around the midpiece
Form a tight spiral around the proximal tail.
Provide ATP for motility.
Tail becomes fully functional
Principal piece (flagellum) forms.
Sperm becomes hydrodynamic.
Excess cytoplasm is removed
Forms the cytoplasmic droplet.
What to memorize
Maturation phase = final head shaping + mitochondrial wrapping + cytoplasm removal.
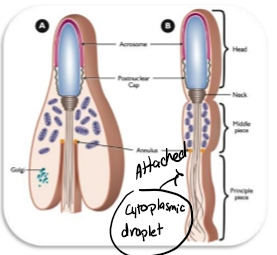
Final Sperm Structure
Head
Contains:
Nucleus (genetic material)
Acrosome (enzymes for penetrating the oocyte)
Midpiece
Packed with mitochondria → ATP production
Powers tail movement
Tail (Flagellum)
Propels sperm through the female reproductive tract
“Propeller” of the cell
Professor’s joke
A colleague calls sperm the “ultimate nuclear weapon.”
(Nucleus + power source + propeller)
Integration With Sertoli Cells
Sertoli cells cradle the developing spermatid.
Provide:
Structural support
Nutrients
Hormonal signals
Phagocytosis of excess cytoplasm
Release the mature sperm into the lumen (spermiation).
Cytoplasmic droplet
The cytoplasmic droplet is like a heavy backpack slowing you down.
What it is
A blob of leftover cytoplasm + organelles.
Attached to the sperm as it leaves the seminiferous tubule.
Where it is removed
NOT removed in the testis
NOT removed in the rete testis
NOT removed in the efferent ducts
NOT removed in the caput epididymis
It is removed in the corpus epididymis.
Why removal matters
Droplet removal = sperm gain motility.
Until then, sperm are immotile.
What to memorize
Cytoplasmic droplet removed in corpus epididymis → motility gained.
FINAL SPERM STRUCTURE
Head
Nucleus
Highly condensed DNA
Genetic variation due to crossing over during meiosis
Acrosome
Contains enzymes needed to penetrate the zona pellucida
Required for fertilization
Midpiece
Packed with mitochondria
Provides ATP for motility
Formed during maturation phase
Tail (Flagellum)
Also called:
Flagellum
Principal piece
Provides propulsion through the female reproductive tract
Professor’s quote
Sperm = “the ultimate nuclear weapon”
(Nucleus + power source + propeller
Sperm Components
Acrosome
Enzymes for penetration of the egg
Nucleus
Genes for fertilization/syngamy
Genetic variation
Mitochondria
Energy for motility
Flagellum- Principal piece
Mechanical basis for motility
Characteristics of Spermatogenesis
The duration of spermatogenesis at regular intervals
Every 16 days in man
Every 13.5 days in bull
Stem cells enter spermatogenesis in groups that are connected by intercellular bridges
Fixed and Constant Spermatogenesis
Fixed” = Spermatogenesis always occurs in the same physical place
Inside the seminiferous tubule, spermatogenesis is anchored:
Type A spermatogonia sit at the basement membrane
They differentiate upward toward the lumen
All development happens alongside Sertoli cells
Fixed means:
Type A → Type B → Primary spermatocyte → Secondary spermatocyte → Round spermatid → Elongated spermatozoon
All of this ALWAYS occurs in the same spatial order
Sertoli cells act as the “scaffolding” that holds the entire process in place
“Constant” = Spermatogenesis never stops
Once puberty begins:
Spermatogenesis runs continuously
New cohorts of cells begin development every few days
This ensures nonstop release of sperm from puberty → death
Species timing (important!):
Bull: new cycle every 13.5 days
Human: every 16 days
This is the number your professor wants you to remember:
➡ Bull = 13.5‑day cycle
Terminology
Cycle= progression through sequence of all stages
Cells changes
internal timeline
what the cells are doing
Stage= specific cellular associations
what you see at the top of the Sertoli cell at a single moment. Stages describe which cell types are present at the luminal edge at a given time.
Changes in an individual cell during successive cycles
Cycle 1: Freshman
Type A spermatogonia along basement membrane
Spermatogonia (mitosis)
Cycle 2: Sophomores
Intermediate and type B spermatogonia, and as a primary spermatocyte begins meiosis I.
1 layer closer to the lumen
Primary spermatocytes (meiosis I)
Cycle 3: Juniors
Developments into a secondary spermatocyte
About halfway to the lumen
Secondary spermatocytes (meiosis II)
Cycle 4: Seniors
Cell undergoes many morphological changes as it develops as a spermatid
near the lumen
Round spermatids
Cycle 4.5: Graduation
Undergoes final changes
Released into the lumen of the tubule as a spermatozoan
Spermiogenesis (Golgi → Cap → Acrosomal → Maturation)
Further maturation occurs as it travels through the male and female reproductive tracts.
Spermatogenic Waves (Stages)
Waves
Refers to sequentil ordering of stages which occur along the length of the seminiferous tubule
Stages = the “wave” in a football stadium
When the wave reaches your section, you stand up (release sperm)
When it passes, you sit back down (return to round spermatids)
The wave keeps moving around the stadium
The seminiferous tubule works the same way
Stage numbers:
Most mammals: Stages 1–8
Rodents: Stages 1–9 (not important for exam)
What each stage contains:
Stage 1: Round spermatids at the top
Stage 4: Spermatids in acrosomal/mid‑transformation
Stage 8: Fully mature spermatozoa ready for release
Key point:
Stages = snapshot in time
Stages = what is visible at the luminal edge
Stages = external pattern
How cycles + stages work together to produce billions of sperm
Inside the seminiferous tubule:
Each Sertoli cell supports multiple cohorts of developing sperm
Each cohort is at a different cycle
The luminal edge displays different stages
Every 13.5 days (bull), the stage shifts forward
Stage 8 releases sperm → resets to Stage 1
This repeats forever
Why this matters:
At any moment, every part of the tubule is producing sperm at a different step
This creates continuous output
This is why males can produce billions per day
. Putting it all together — the professor’s “stadium wave” analogy
Your professor wants you to visualize it like this:
The seminiferous tubule = a circular stadium
Each Sertoli cell = a section of seats
Each section has people (cells) at different points in the wave
When the wave reaches a section → sperm are released
Then that section resets and waits for the next wave
The wave keeps moving around the stadium forever
This ensures:
Continuous spermatogenesis
Continuous sperm release
No gaps in fertility (unless heat stress, injury, etc.)
⭐ 7. Why the 13.5‑day cycle matters clinically
Your professor emphasized this earlier, but it connects here:
If a bull has heat stress, fever, or testicular injury:
The damage affects spermatogonia first
You won’t see infertility immediately
You will see it ~60 days later (full spermatogenic timeline)
The 13.5‑day cycle helps predict when fertility will drop
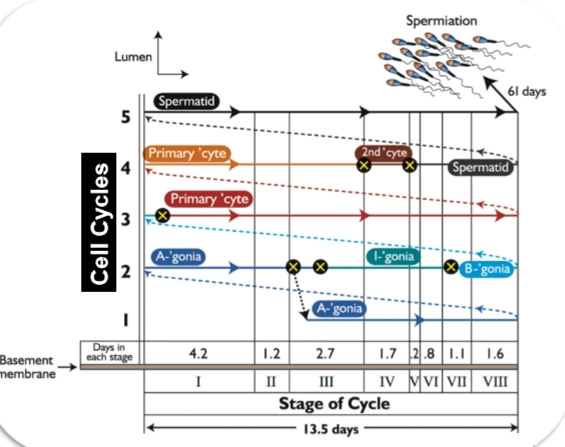
The diagram shows spermatogenesis as a repeating 13.5‑day cycle that occurs in waves along the seminiferous tubule.
Each vertical column represents one cohort of developing sperm cells moving from the basement membrane → lumen over time.
Multiple cohorts exist at once, so the tubule always contains cells at different stages.
🧱 Bottom Layer: Spermatogonia (Stem + Mitotic Phase)
Near the basement membrane, you see:
A₁, A₄, Intermediate (I), and B spermatogonia
These are the mitotic divisions that expand the germ cell population.Arrows show how each type transitions into the next.
These divisions occur in synchronized groups, not individually.
🔄 Middle Layer: Meiosis
As the cohorts move upward:
Primary spermatocytes (big cells) enter meiosis I
They become secondary spermatocytes (brief stage)
Then quickly transition into round spermatids
This is the meiotic phase, where chromosome number is halved.
🎯 Top Layer: Spermiogenesis (Spermatid Remodeling)
Near the lumen, the diagram shows:
Round spermatids → elongating spermatids → mature spermatozoa
This is where the acrosome forms, the tail develops, and the nucleus condenses.
At the very top right, you see spermiation — the release of mature sperm into the lumen.
The diagram notes that full development from spermatogonium → spermatozoon takes ~61 days.
⏱ Bottom Timeline: The 8 Stages of the Seminiferous Epithelial Cycle
The colored bar at the bottom shows:
Stages I–VIII
Each stage lasts a specific number of days
(e.g., Stage I = 4.2 days, Stage VII = 1.1 days)Total cycle length = 13.5 days
A given cohort of cells moves through all 8 stages, then the cycle repeats.
🌊 Why the “Waves” Matter
The five vertical “columns” represent five overlapping cycles.
This explains why:
The tubule always contains all cell types at once
Sperm production is continuous, not episodic
Different regions of the tubule are in different stages at any moment
Pathway of Sperm
Seminiferous tubules – site of sperm production.
Rete testis → efferent ducts – transport.
Epididymis
Caput (head): immature sperm, cytoplasmic droplet still attached.
Corpus (body): droplet removed → sperm gain motility.
Cauda (tail): storage warehouse.
Vas deferens → pelvic urethra
Sperm meet seminal plasma → “bubble wrap” protection + nutrients.
Penile urethra → glans penis
Delivery system for ejaculation.
Temperature Requirment
Temperature Requirement
Testis must be 4–6°C cooler than body temperature.
Heat stress damages spermatogonial stem cells, causing infertility ~60 days later (bull) or ~70 days later (human).
Seminiferous Tubule Structure
Basement membrane – protective boundary.
Spermatogonia (stem cells) sit on the basement membrane.
Sertoli cells – “nurse cells” that support developing sperm.
Lumen – where mature sperm are released.
Leydig Cells
Leydig Cells (Interstitial Cells)
Located outside seminiferous tubules.
Produce testosterone in response to LH.
Testosterone is essential for:
Spermatogenesis
Secondary sex characteristics
Supporting Sertoli cell function
Male equivalent of female theca cells.
Equivalent to theca cells in females
Produce testosterone in response to LH
Testosterone:
Needed in high concentration inside seminiferous tubules
Supports mitosis + meiosis
Some enters Sertoli cells → converted to estrogen
Sertoli Cells
Inside seminiferous tubules, surrounding developing sperm.
Equivalent to female granulosa cells.
Functions:
Support & nourish germ cells
Form blood–testis barrier (tight junctions)
Convert testosterone → estrogen
Produce:
Inhibin → ↓ FSH
Anti‑Müllerian hormone (AMH)
Growth factors
Androgen-binding protein (ABP) → traps testosterone in tubule
Required for spermiogenesis (final differentiation).
Equivalent to granulosa cells in females
Functions:
Support and nourish developing sperm
Form blood–testis barrier (tight junctions)
Produce:
Estrogen (from testosterone)
Inhibin → ↓ FSH
Anti‑Müllerian hormone (AMH)
Growth factors
Androgen Binding Protein (ABP) → traps testosterone in tubules
Required hormone: FSH
Hormonal Regulation in males
Hypothalamus
Releases GnRH in tonic pulses.
Anterior Pituitary
LH → stimulates Leydig cells → testosterone
FSH → stimulates Sertoli cells → ABP, estrogen, inhibin, growth factors
Feedback Loops
Testosterone → negative feedback on LH.
Inhibin → negative feedback on FSH.
Important Distinction
Males do NOT have a surge center → no LH surge → constant tonic pulses.
Full Process of Spermatogenesis
Total Time
Human: ~70 days
Bull: 61 days (4.5 cycles × 13.5 days each)
Ram: ~48 days
Three Major Phases
1. Spermatogonial Phase (Mitotic)
Location: Basement membrane
Cells: Type A → Type B spermatogonia
Key points:
Type A = true stem cells
Can self‑renew (mitosis)
Or differentiate into Type B
Type B = committed; cannot revert
Must become primary spermatocytes or undergo apoptosis
Cells are connected by intercellular bridges (syncytium)
Keep divisions synchronized
If a cell disconnects → apoptosis
2. Spermatocyte Phase (Meiotic)
Location: Moving toward lumen
Events:
Primary spermatocyte undergoes Meiosis I → secondary spermatocyte
Secondary spermatocyte undergoes Meiosis II → round spermatids
Round spermatids are haploid.
Blood–Testis Barrier
Tight junctions between Sertoli cells protect meiotic cells from immune attack.
Primary spermatocytes must cross the barrier before meiosis.
3. Spermiogenesis (Differentiation Phase)
Round spermatid → elongated spermatozoon
Requires: FSH + Sertoli cell support
NO mitosis or meiosis here.
Four Phases (Going Crazy And Mad)
Golgi Phase
Golgi forms the acrosome vesicle.
Cap Phase
Acrosome spreads over nucleus like a cap.
Centrioles migrate to opposite pole → future tail.
Acrosomal Phase
Cell elongates.
Acrosome fully wraps nucleus.
Tail axoneme begins forming.
Mitochondria migrate toward midpiece.
Maturation Phase
Final assembly of head, midpiece, tail.
Mitochondria wrap midpiece.
Excess cytoplasm forms cytoplasmic droplet.
Cytoplasmic Droplet
Present in seminiferous tubules, rete testis, efferent ducts, and caput epididymis.
Removed in corpus epididymis → sperm gain motility.
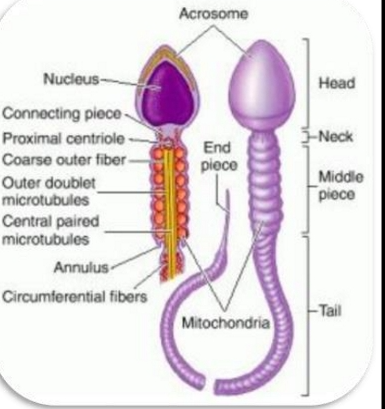
Sperm Structure
Head
Nucleus -tightly packed DNA
Acrosome- enzymes for penetrating zona pellucida
Midpiece
Mitochondrial sheath → ATP for motility
Tail
Axoneme (9+2 microtubule structure)
Propulsion through female tract
Sperm production Scale
Daily Production
Bull: 9–13 billion/day
Boar: 17–22 billion/day
Human: ~1000 sperm per heartbeat
Why so many?
Spermatogenesis is:
Fixed (always occurs in same spatial pattern)
Constant (continuous waves of development)
CYCLES & STAGES OF SPERMATOGENESIS
Cycles = Cellular progression
4.5 cycles in all mammals
Bull cycle length: 13.5 days
4.5 × 13.5 = 61 days total
Stages = What’s happening at the Sertoli cell apex
Stage 1: round spermatids
Stage 8: elongated spermatozoa ready for release
After release → stage resets to 1
Wave Analogy
Like a stadium wave:
Each Sertoli cell region “stands up” (releases sperm)
Then resets and waits for next cycle
NJURY, HEAT STRESS & INFERTILITY TIMING
Why infertility appears later
Damage hits spermatogonial stem cells first.
Mature sperm already in epididymis still function.
Infertility appears one full spermatogenic cycle later:
Bull: ~61 days
Ram: ~47–48 days
Human: ~70 days
Examples
Fever
Scrotal heating
Laptop on lap
Obesity (poor thermoregulation)
EPIDIDYMAL MATURATION
Caput
Immature sperm
Cytoplasmic droplet present
No motility
Corpus
Droplet removed
Motility gained
Cauda
Storage
Fertilization‑competent after ejaculation + capacitation in female tract
Semen Production
Functions
Fluid environment for transport
Provides energy source
Buffer
Maintains osmolality
Need to rely on environment
Composition
Fructose, inositol, citric acid, prostaglandins, growth factors, cholesterol, lipids
Contributions from testes & accessory glands\
Testes contributions
Sperm (immature)
Rete testis fluid
Epididymis contributions
Maturation of sperm
Loss of cytoplasmic droplet
Gain forward motility
Concertation
Storage- caput, corpus, cauda
Accessory gland contributions
adds the rest of the semen components (highly variable)
Epididymis
Caput= head
Absorption to concentrate sperm
Transport
Corpus= Body
Secretions “mature” sperm
Remove cytoplasmic droplet
Foward/progressive motility
Cauda= Tail
Storage for ejaculation
Sperm Transport
Seminiferous tubules
Passively moved by flow of fluids produced by Sertoli cells & flowing to rete testis
Efferent ductules
Flow of fluids into the ducts aided by the reabsorption of fluids within the ducts
Flow through the ducts aided by cilia
Epididymis
Spontaneous peristaltic contractions of smooth muscle lining the wall
Vas deferens
Flow into vas deferens due to steady flow through epididymis
Flow through vas deferens at ejaculation due to peristaltic contractions into uretha
Emptying of vas deferens leaves room for further flow from the epididymis
Pelvic & Penile urethra
Rhythmic contractions of bulbospongiosus & ischiocavernosus muscles at ejaculation
Simultaneous emptying of accessory glands to provide fluid vehicle
Help push seminal plasma into urethra to deposit in female
Sperm transport more
1. Seminiferous Tubules
Passive movement
Carried by fluid secreted by Sertoli cells
2. Rete Testis
Fluid movement continues
3. Efferent Ducts
Cilia + fluid flow pull sperm into epididymis
4. Epididymis
Caput: concentration
Corpus: maturation, droplet removed, motility gained
Cauda: storage
Movement: smooth muscle peristalsis
Hormone: oxytocin enhances contractions
5. Vas Deferens
Strong smooth muscle contractions move sperm to urethra
6. Pelvic & Penile Urethra
Skeletal muscles (ischiocavernosus, bulbospongiosus)
Forceful contractions → ejaculation
Important:
Sperm do NOT swim in the male tract.
Movement is entirely due to fluids + muscle contractions.
Seminal Plasma & Accessory sex glands
Seminal Vesicles
Fructose
Energy substrates
Majority of seminal plasma volume
Prostate
Buffers
Enzymes
Helps activate sperm
Bulbourethral (Cowper’s) Gland
Mucus
Lubrication
Neutralizes urethra
Functions of seminal plasma
Protect sperm
Provide nutrients
Package sperm for transport (“bubble wrap”)
Facilitate movement through female tract
Semen evaluation & characteristics
Volume
Concentration
Sperm cells per volume
Sperm motility
percentage of motile sperm
Sperm morphology
percentage of normal sperm
Volume increases the concentration decreases. Volume decreases the concentration increases.
Volume & pH
sperm cells are extremely sensitive anything can kill sperm
Concentration
Use hemocytometer
Has 1mm X 1 mm grid
Calculate number
Spectrophotometer
Decrease density more light lower concentration
Increase density less light higher concentration
Morphology
Collect Smaple
Fix and stain sperm cells
Look abnormalites
Head- pear shape, slender, double head, micro or macro, cephalic
Midpiece- kinked, double, swollen
Tail- coiled, cytoplasmic droplet, absent, double
Spermatozoan
Head
plasma membrane
apical ridge
acrosome
nucleus
Tail
Midpiece
Proximal centriole
Mitochondrial sheath
Distal centriole
Principal piece
Motility
Droplet test
Place fresh sample on slide
Estimate percentage (7/10)
Ranking system
High
Medium
Low
Time lapsed photography
Narrower down by precent is 100% moving No is 50% moving yes so look for percent in between
Mate selection
Rely on:
odors
Visual cues
Vocalization
Pre-copulatory
Searching for females
Flehmen response → detects pheromones
Courtship: nudging, vocalizations, mounting attempts
Copulatory
Erection (hypothalamus-mediated)
Mounting
Intromission
Ejaculation
Post-copulatory
Dismount
Refractory period
Memory-based learning → older males more efficient
0. Mating Behavior & Copulation Sequence
Pre-copulatory
Searching for females
Flehmen response → detects pheromones
Courtship: nudging, vocalizations, mounting attempts
Copulatory
Erection (hypothalamus-mediated)
Mounting
Intromission
Ejaculation
Post-copulatory
Dismount
Refractory period
Memory-based learning → older males more efficient
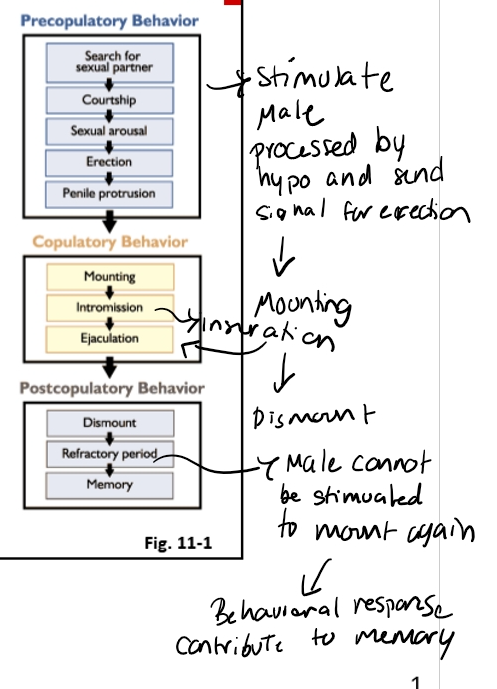
Cus function on hypothalamus
Step 1
Erotogenic stimuli cause sensory nerves to fire
Step 2
Sensory nerves activate
“Reproductive Behavior Center” in hypothalamus
Step 3
Stimulation of parasympathetic nerves that innervate penile arterioles
Step 4
Parasympathetic nerve terminals release nitric oxide
Step 5
Nitric oxide initiates biochemical cascade that causes erection
What starts everything?
Sensory cues → Hypothalamus → Erection
The male detects:
Sight of females in estrus
Smell (pheromones via Flehmen response → vomeronasal organ)
Sound / behavior (mounting invitations, restlessness)
These cues are processed by the hypothalamus, specifically the reproductive behavior center, which then activates:
Parasympathetic nerves → Nitric oxide (NO) release
Parasympathetic fibers innervate the penile arterioles.
They release nitric oxide (NO).
NO causes vasodilation + trapping of blood in the corpora cavernosa and corpus spongiosum.
This is the physiological basis of erection in both:
Vascular penis species (stallion, human)
Fibroelastic penis species (bull, ram, boar — with sigmoid flexure)
2. Erection vs Ejaculation: Two Different Control Systems
Erection = Parasympathetic + Hypothalamus
Requires sensory input + hypothalamic processing.
Uses NO to trap blood.
Ejaculation = Spinal Reflex (NO brain involvement)
Once the glans penis receives the correct mechanical stimulus, ejaculation is triggered by a simple spinal reflex:
Sensory nerves in glans penis detect pressure/temperature.
Signal travels to spinal cord.
Motor neurons fire to:
Ischiocavernosus muscle
Bulbospongiosus muscle
These skeletal muscles contract rhythmically → expel semen.
Species differences in the stimulus:
Boar: corkscrew penis locks into cervix → pressure triggers ejaculation
Ram/Bull: vaginal pressure + temperature
Stallion: must press glans firmly against cervix
Copulation Physiology
A. Sexual Arousal
Female cues (sight, smell, sound) → hypothalamus
Hypothalamus → parasympathetic nerves → nitric oxide release
Nitric oxide traps blood in erectile tissue → erection
B. Intromission
Glans penis enters female
Species-specific stimulation:
Boar: pressure + corkscrew lock in cervix
Stallion: pressure against cervix
Ruminants: vaginal pressure + temperature
C. Ejaculation
Does NOT involve the brain
Simple spinal reflex:
Sensory nerves in glans penis → spinal cord → motor neurons
Motor neurons → ischiocavernosus & bulbospongiosus muscles
Rhythmic contractions → semen expelled
D. Refractory Period
Male temporarily unresponsive
Longer in young males
Shorter in experienced males
🐂 12. Optimizing Ejaculate Output (False Mount Technique)
Used in AI studs (e.g., Select Sires).
One false mount → 55% increase in sperm per ejaculate.
Two false mounts → no additional benefit.
Works because:
Extra stimulation → more oxytocin → stronger epididymal/vas deferens contractions
More sperm moved into pelvic urethra before ejaculation
Site of Semen Deposition
Ruminants (bull, ram, buck)
Anterior vagina
Goal: deposit as close to cervix as possible
Cervical mucus becomes less viscous under estrogen → easier passage
Boar
Intra‑cervical
Corkscrew penis locks into cervix
Very large volume ejaculate
Ends with a gel plug to prevent backflow
Stallion
Intrauterine + cervical
High volume
Cervical folds guide semen into uterus
Sperm transport in the Female
Two phases:
A. Rapid Phase (minutes)
Triggered by oxytocin release during copulation
Oxytocin → uterine peristaltic contractions
Moves sperm quickly toward the uterus/oviduct
B. Slow Phase (hours)
Combination of:
Sperm’s own motility
Ongoing uterine contractions
Takes ~8 hours in ruminants to reach the AIJ (ampulla–isthmus junction)
6. Cervical Phase (Ruminants)
The cervix acts as:
A filter
A barrier
A selector for normal sperm
During estrus:
Estrogen dilates the cervix
Mucus becomes watery → easier passage
Abnormal sperm get trapped in cervical folds
Two types of Penes:
Vascular
No Sigmond flexure
Penis fills with blood
Increase blood pressure
Humans and Stallions
Fibroelastic
Rigid in non-erect state
“S” shape due to Sigmond flexure
Increase blood pressure= straightens
Bulls, Rams, Boars
Phases of Ejaculation
What the image is about
It’s a diagram of the ejaculation reflex — a spinal reflex that coordinates sensory input from the penis with rapid, rhythmic muscle contractions that push semen out of the urethra.
Think of it as:
Stimulus → Spinal cord reflex → Powerful pelvic muscle contractions → Semen expulsion
🔍 Step‑by‑step explanation1. Intromission
This just means penetration.
Once the penis is inside the reproductive tract, stimulation begins.
2. Sensory stimulation of the glans penis
The glans (tip of the penis) has dense sensory receptors for:
Pressure
Temperature
Touch
These sensory signals travel through afferent neurons to the lumbosacral spinal cord.
This is important:
Ejaculation is controlled by a spinal reflex center — not the brain.
3. Spinal reflex activates pelvic muscles
The spinal cord sends motor signals back to specific muscles:
Urethralis muscle
Squeezes the urethra like a pump.Bulbospongiosus muscle
Provides the strongest contractions that actually propel semen.Ischiocavernosus muscle
Stabilizes the penis and increases pressure inside erectile tissue.
These muscles contract suddenly and powerfully, in rhythmic bursts.
4. Expulsion of semen
Those coordinated contractions force semen through the urethra and out of the body.
This is the ejaculatory phase, following emission (movement of semen into the urethra).
🧩 Why this matters physiologically
Ejaculation is not voluntary once the reflex is triggered.
It depends on sensory input, spinal integration, and striated muscle contractions.
The muscles involved are the same ones used in pelvic floor function.
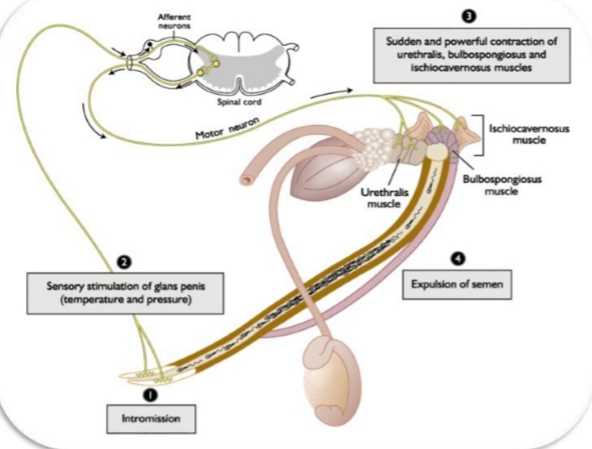
Erection & Ejaculation
Sensory stimuli and Psychic stimuli cause Reflex, sympathetic & parasympathetic nerves
Reactions:
Increased vascular supply causes erection
Smooth muscle contractions of accessory glands cause emission
Contraction of muscles causes ejaculation
Post Copulatory Phase
Characterized by period of Refractory
Time in which additional stimuli will not stimulate male to copulate again
Results in satiation & unwillingness
Differs from exhaustion
Occurs when over copulated
Unable to copulate even with appropriate stimuli
Refractory Period
A temporary period where the male cannot be stimulated to mate again.
Length depends on:
Age
Libido
Temperature
Number of females
Young bulls: rule of thumb = 1 female per month of age
(12‑month bull → ~12 females)
Exhaustion (different from refractory)
Occurs when:
Many females come into estrus at once (e.g., synchronization)
Male attempts to breed too many in a short window
Results in:
Depletion of sperm reserves
Inability to copulate even after refractory period ends
Sperm Transport
Rapid Phase
Occurs within minutes
Peristaltic contractions induced by copulation
Oxytocin
Slow phase
Fertilizing sperm to AI junction
8 hrs.
Motility + Contractions
Sperm Transport: Cervical Phase
Billions Phase
Roles of Cervix
Receptive at estrus (mucus)
Reservior cervical crypts (minor)
Protection from vagina (phagocytosis)
Energy (from mucus)
Filtration of dead + defective sperm
Influenced by estrogen
Motility required to traverse mucus
Where it happens: Cervix
When: Immediately after semen is deposited in the anterior vagina (ruminants)
What estrogen does during estrus:
Opens (dilates) the cervix
Thins the cervical mucus → easier for sperm to swim through
What the cervix does:
Acts as a filter
Removes:
Dead sperm
Dying sperm
Abnormal sperm
These get trapped in cervical crypts (little pockets in the cervix)
Immune system role:
Estrogen activates immune cells
Neutrophils perform phagocytosis of sperm that don’t make it through
After ovulation:
Progesterone rises → cervix closes
Mucus becomes thick → forms a cervical plug (pregnancy protection)
Key takeaway:
Billions enter → only thousands make it past the cervix.
Sperm Transport: Uterine Phase
Thousands survive
Movement
Primarily contractions
Contraction stimulators
PGF in Semen
Oxytocin form Posterior Pituitary
Sperm Capacitation
Hypermotility
Acrosome Reaction
Where: Uterine body → uterine horns
How long: ~8 hours in cattle
What moves sperm forward:
Oxytocin pulses (from posterior pituitary)
Prostaglandin F2α (from seminal plasma)
Both stimulate smooth muscle contractions → peristalsis
What sperm must do here:
Continue swimming
Survive immune attack
Begin capacitation
Capacitation — Final Maturation Step
Where: Uterus
When: During the slow phase of transport
Why: Sperm MUST complete capacitation to fertilize the oocyte
What capacitation actually is:
Removal of seminal plasma proteins (specifically glycocalyx glycoproteins / glycosaminoglycans) from the sperm head.
What capacitation accomplishes:
Hyper‑motility
Tail beats become faster + more forceful
Needed to penetrate the zona pellucida
Exposes the acrosome
Acrosomal membrane becomes accessible
Prepares sperm for the acrosome reaction
Timing:
Sheep: ~1.5 hours
Pigs: 3–6 hours
Cattle: ~7 hours
Cool fact:
Capacitation is reversible.
If you put capacitated sperm back into seminal plasma → proteins re‑coat the head → motility decreases.
The cervix removes:
Dead sperm
Abnormal sperm
Low-motility sperm
Why?
Cervical crypts trap defective sperm.
Estrogen (estrus):
Dilates cervix
Thins mucus → easier passage
Activates immune cells → phagocytosis of dead sperm
After ovulation:
Progesterone rises
Cervix closes
Mucus becomes thick → forms a cervical plug
Only thousands of sperm make it past the cervix.
4⃣ Uterine Phase — Longest Part of the Journey
Sperm move by:
Uterine contractions (oxytocin + PGF2α)
Their own motility (now beginning to matter)
Key event: Capacitation
Occurs in the uterus.
Capacitation =
Removal of seminal plasma proteins (glycocalyx/glycoconjugates)
→ Exposes the acrosome
→ Enables hyperactivated motility
→ Prepares sperm for acrosome reaction
⏱ Species timing:
Sheep: ~1.5 hr
Pig: 3–6 hr
Cattle: ~7 hr
💡 Capacitation is reversible
If you put capacitated sperm back into seminal plasma, the coating reforms
What is Capacitation?
Biochemical change
Sperm outer plasma membrane
Alters glycosaminoglycans
Requires 1-7 hours
Sheep 1.5
Pigs 3-6
Allows acrosome reaction to occur
Reversible process
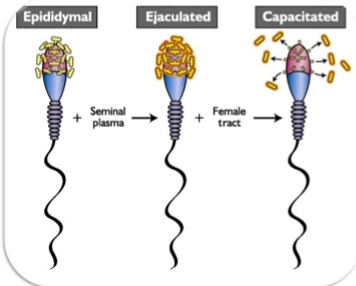
Deposition → Rapid Phase transport
Happens within minutes of copulation.
What drives it?
Oxytocin release during mating
Prostaglandin F2α from seminal plasma
→ Both stimulate uterine peristaltic contractions
Purpose:
Push a portion of sperm rapidly toward the uterus and oviducts.
⚠ These sperm are NOT the ones that fertilize.
They arrive too early and are not yet capacitated.
2⃣ Retrograde Loss
A huge portion of sperm is immediately lost:
Flowing out of the vulva
Trapped in vaginal mucus
Destroyed by immune cells
This is why billions must be deposited
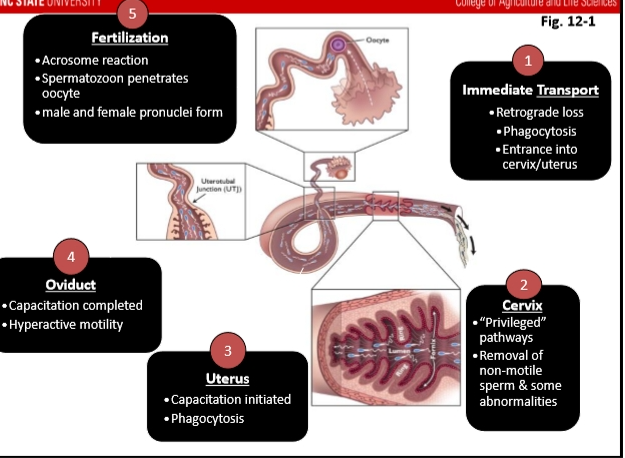
Sperm Transport: Utero-tubal Junction
Limits number of sperm reaching oviduct
Acts as a 2nd filter
Sperm reservoir
Affected by estrogen: Progesterone
Analogy: I‑40 going from 4 lanes → 2 lanes.
Functions:
Limits sperm entry into the oviduct
Ensures only high‑quality, capacitated sperm pass
Prevents polyspermy by controlling sperm numbers
Special case:
Some species (e.g., bats) use the UTJ as a sperm reservoir for months.
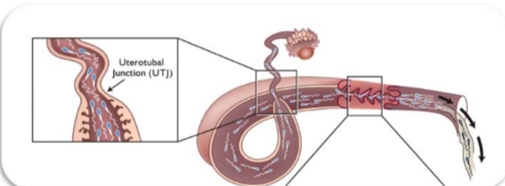
Sperm Transport: Oviduct Phase
Only hundreds to thousands survive
Transport
By contractions
By fluid “currents” caused by cilia
Sperm pool (reservoir) in isthmus
Fertilization occurs at Ampulla-Isthmus junction (AIJ)
Only hundreds to a few thousand sperm reach this point.
Movement aided by:
Ciliary currents
Smooth muscle contractions
Sperm hyperactivation
Hormonal control:
High estrogen → flow toward AI junction (helps sperm reach oocyte)
High progesterone → flow toward uterus (helps embryo move back)
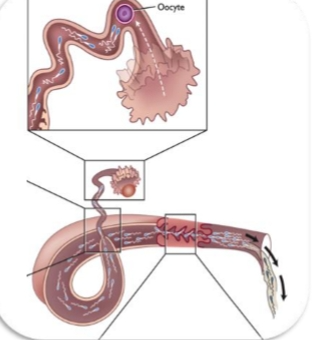
Sperm transport
Occurs at the ampulla–isthmus junction.
Steps:
Hyperactivated sperm reach the oocyte
Acrosome reaction occurs
Sperm penetrates zona pellucida
Sperm enters oocyte cytoplasm
Male + female pronuclei fuse → zygote
Fertilization: What Happens When Sperm Reach the Oocyte
The oocyte has several layers the sperm must get through:
Cumulus oophorus
Cloud of granulosa cells surrounding the oocyte
Held together by hyaluronic acid
Sperm use hyaluronidase (from the acrosome) to wiggle through
Zona pellucida (ZP)
Thick glycoprotein shell
Contains ZP1, ZP2, ZP3
ZP3 is the key sperm-binding molecule
Binding to ZP3 triggers the acrosome reaction
Vitelline membrane / oolemma
Actual plasma membrane of the oocyte
Only one sperm is allowed to fuse
Acrosome Reaction
Triggered when a capacitated sperm binds ZP3.
What happens:
The acrosomal membrane fuses with the sperm plasma membrane
Enzymes (acrosin, hyaluronidase) are released
These digest a path through the zona pellucida
The sperm becomes “drilled down” to its inner acrosomal membrane
This exposes the equatorial segment → the part that actually fuses with the oocyte
Fusion and Blocks to Polyspermy
Fusion
Once a sperm reaches the oolemma:
The sperm’s equatorial segment binds the oocyte membrane
Membranes fuse
The sperm nucleus enters the cytoplasm
The oocyte completes meiosis II
Second polar body is expelled
Male and female pronuclei form
Pronuclei merge → syngamy
A diploid zygote is created
Blocks to Polyspermy
To prevent multiple sperm from entering:
Fast block (electrical, in some species—not strong in mammals)
Cortical reaction (main mammalian block)
Cortical granules release enzymes
Zona pellucida hardens
ZP3 is modified so no more sperm can bind
This is called the zona block
Endogenous Factors affecting Sperm Motility
Age (sperm and donor)
Time between and after ejaculation
Sperm Maturation
Morphology
Energy Stores (ATP)
Flagellar movement
Cell surface
Membrane integrity
Exogenous Factors affecting Sperm Motility
Biophysical & Physiological Factors
pH, temperature, viscosity
Suspending fluids
Male & Female tracts
Stimulation/Inhibition
Hormones
Environmental pollutants
Inorganic ions
Caffeine
Luminal Fluids in contact with Spermatozoa
Fate of Unsuccessful Sperm in the Cow reproductive tract
1 billion sperm inseminated
73% recovered
Mucus discharge- 61%
Urine - 1%
Vagina & Cervix- 4%
Uterus 7 Oviducts (retained)- 6.5%
Undergo phagocytosis
Abdominal cavity- 0.5%
Ovulation & Egg Transport
Oocyte and cumulus cells captured by infundibulum
Transport of Oocyte (ovum)
Mechanism
Cilia- infundibulum and ampulla
Fluid currents
Rhythmic segmented peristaltic contractions
Gamete longevity (hours)
Fertilization
Layers of the Oocyte
From outside → inside:
Cumulus granulosa cells
Sticky cloud of follicular cells surrounding the oocyte.
Sperm must push through these first.
Zona pellucida (ZP)
Glycoprotein shell around the oocyte.
Contains ZP3 and ZP2 (critical for sperm binding + acrosome reaction).
Perivitelline space
Space between zona pellucida and oocyte membrane.
Vitelline membrane (oocyte plasma membrane)
The membrane the sperm must fuse with.
2⃣ Four Major Steps of Fertilization
Fertilization = Acrosome Reaction → Zona Reaction → Vitelline Block → Syngamy
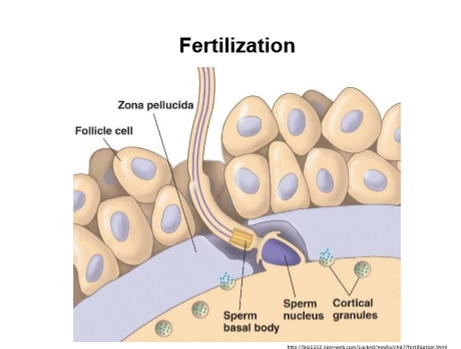
Steps in Fertilization
Acrosome Reaction
Zona reaction
Vitelline Block
Pronuclear development & syngamy
tep 1: Acrosome Reaction
Triggered when sperm contacts the zona pellucida.
Key proteins:
• ZP3 = docking protein
○ First contact.
○ Binds the sperm at the apical region (between acrosome + nucleus).
○ Think: “ZP3 = Velcro → holds sperm in place.”
• ZP2 = detonator protein
○ Triggers the acrosome reaction.
○ Causes the sperm to release enzymes.
What happens during the acrosome reaction?
• Outer acrosomal membrane fuses with sperm plasma membrane.
• Releases enzymes:
○ Acrosin (main one) → digests zona pellucida.
○ Hyaluronidase → helps break apart cumulus cells.
• Hyperactivated motility pushes sperm through the hole it creates.
4⃣ Step 2: Zona Reaction (First Block to Polyspermy)
Triggered when the FIRST sperm enters the perivitelline space.
Cortical granules (just under the vitelline membrane):
• Release calcium into the perivitelline space.
Calcium causes:
1. Hardening of the zona pellucida
○ Zona becomes impenetrable.
2. Down‑regulation of ZP3
○ No more sperm can bind.
Purpose:
✔ Prevents additional sperm from binding to the zona pellucida.
5⃣ Step 3: Vitelline Block (Second Block to Polyspermy)
Triggered when the sperm nucleus actually touches the vitelline membrane.
What happens:
• The vitelline membrane engulfs the sperm head.
• A membrane‑level signal spreads across the oocyte.
• Prevents any additional sperm nuclei from fusing with the oocyte cytoplasm.
Purpose:
✔ Prevents multiple sperm nuclei from entering the oocyte.
6⃣ Step 4: Syngamy
Final step = formation of the zygote.
What happens:
• Sperm nucleus detaches from the midpiece.
• Sperm DNA decondenses → forms the male pronucleus.
• Female pronucleus + male pronucleus migrate toward each other.
• They fuse → zygote (2N).
7⃣ Why Polyspermy Is Fatal
• Humans: 23 chromosomes from sperm + 23 from oocyte = 46 total.
• If multiple sperm enter → too many chromosomes → embryo dies.
8⃣ Factors That Reduce Block Efficiency
• Aged oocyte (older egg = weaker blocks)
• Aged female
• Heat stress / high temperature
These increase the risk of polyspermy.
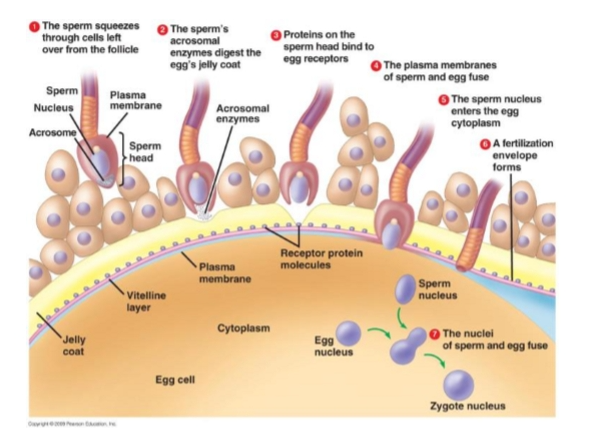
Acrosome Reaction
Penetration of cumulus granulosa cells and corona radiata
Hyaluronidase, corona penetrating enzyme
Fusion of plasma membrane and outer acrosomal membrane
Multiple vesicles appear on cell surface
Exposure of inner acrosomal membrane
Penetration of zona pellucida
Acrosin
Acrosome swells & is lost
ZP3: sperm receptor
ZP2: initiates acrosome reaction
Step 1 — Acrosome Reaction
Triggered when sperm binds zona pellucida.
Zona proteins:
ZP3 = docking protein
ZP2 = triggers acrosome reaction
Acrosome releases enzymes (acrosin) → digests zona → sperm pushes through.
Step 2 — Zona Reaction (Block #1 to polyspermy)
First sperm enters perivitelline space
Cortical granules release calcium
Calcium:
Hardens zona
Down‑regulates ZP3
Prevents more sperm from binding
Step 3 — Vitelline Block (Block #2)
Oocyte membrane engulfs sperm nucleus
Membrane changes prevent additional nuclei from entering
Step 4 — Syngamy
Sperm nucleus decondenses
Male + female pronuclei fuse
Zygote formed