KAAP 310 (electrolytes and balance)
1/98
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
99 Terms
total body water (TBW)
content of average young adults is
60% of the body weight in men
50% in women
fluid compartments
areas separated by selectively permeable membranes and differing from each other in chemical composition
major fluid compartments
65% intracellular fluid
35% extracellular fluid (divided into:)
25% tissue fluid
8% blood plasma and lymph
2% transcellular fluid
transcellular fluid
catch all category for cerebrospinal, synovial, peritoneal, pleural, and pericardial fluids; vitreous and aqueous humors of the eye; bile; and fluid in the digestive, urinary, and respiratory tracts
how is fluid exchanged
continually between compartments by way of capillary walls and plasma membranes
water movement
water moves by:
osmosis from digestive tract to bloodstream
capillary filtration from blood to tissue fluid
from tissue fluid it may be:
reabsorbed by capillaries
osmotically absorbed into cells
taken up by lymphoid system to return to bloodstream
why do osmotic gradients between ICF ad ECF never last long
osmosis restores balance within seconds
How is direction of osmosis determined?
determined by relative concentration of solutes in each compartment
most abundant solute particles
electrolytes - especially:
sodium salts in ECF
potassium salts in ICF
Electrolytes
play principal role in governing body’s water distribution and total water content
fluid balance
when daily gains and losses are equal and fluids are properly distributed in the body
typical loss and fain of ~2500 mL/day
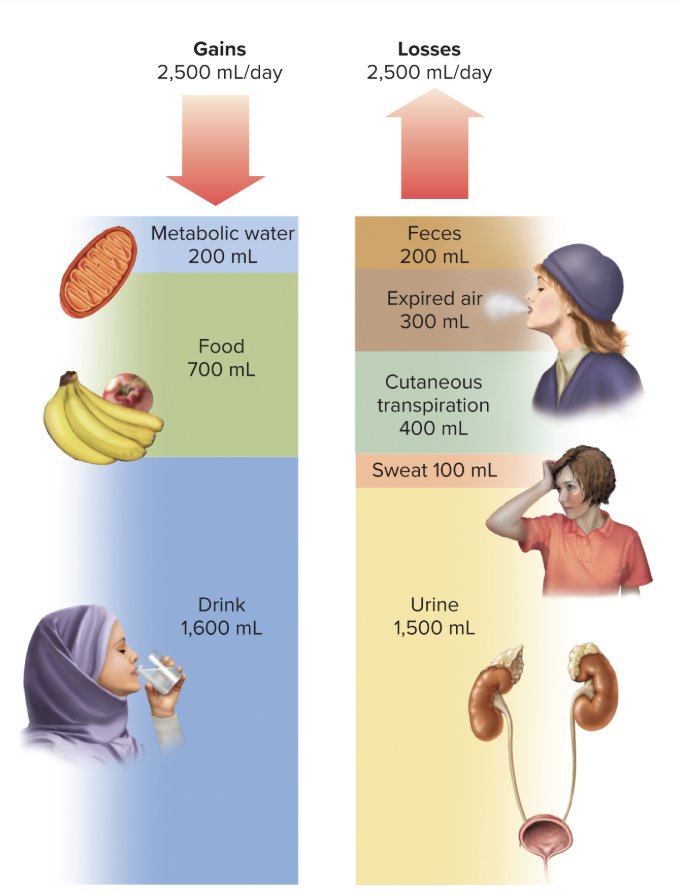
sources of fluid gain
metabolic water (200mL/day) - produced as by-product of dehydration synthesis reactions and aerobic respiration
preformed water (700mL/day) - ingested in food and drink
routes of water loss
urine
cutaneous transpiration - water that diffuses through the epidermis and evaporates
expired breath
feces
sweat
***listed in order of magnitude
factors affecting respiratory loss
respiratory loss increases in cold weather
hot, humid, weather slightly reduces respiratory loss, but increases perspiration
prolonged heavy work can raise respiratory loss although it significantly reduces urine output
insensible water loss
output through the breath and cutaneous transpiration
sensible water loss
noticeable output, particularly through the urine and in case of sufficient sweating to produce obvious wetness of the skin
obligatory water loss
output that is relatively unavoidable
expired air
cutaneous transpiration
sweat
fecal moisture
minimum urine output needed to prevent azotemia
**dehydrated individuals cant prevent these losses
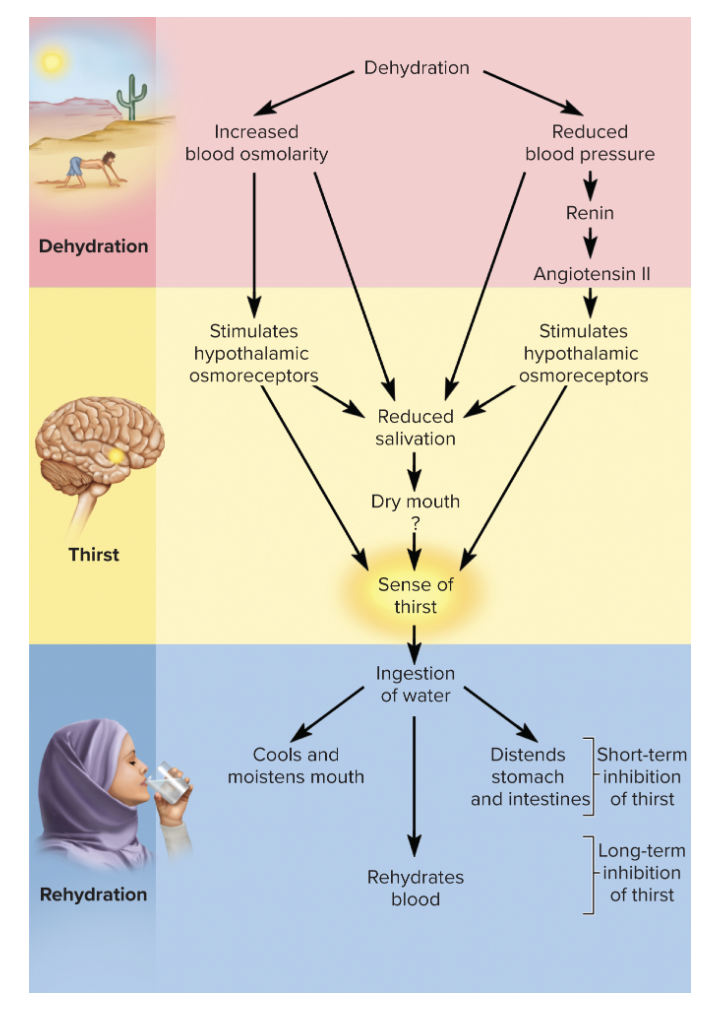
how is the regulation of intake governed?
mainly by thirst
dehydration effects on blood
reduces blood volume and pressure while raising osmolarity
osmoreceptors
respond to angiotensin II and rising osmolarity of ECF
signs that body has water deficit
osmoreceptors communicate with other hypothalamic neurons that produce antidiuretic hormone
promote water conservation
communicate with cerebral cortex to produce conscious sense of thirst
why do we salivate less when thirsty
osmoreceptor response leads to sympathetic output from hypothalamus that inhibits salivary glands
saliva is produced primarily by capillary filtration
in a dehydrated person, it is opposed by the lower capillary blood pressure and higher osmolarity of the blood
what does long term satiation of thirst depend on?
absorbing water from small intestine and lowering blood osmolarity
reduced osmolarity stops the osmoreceptor response, promotes capillary filtration, and makes saliva more abundant and watery
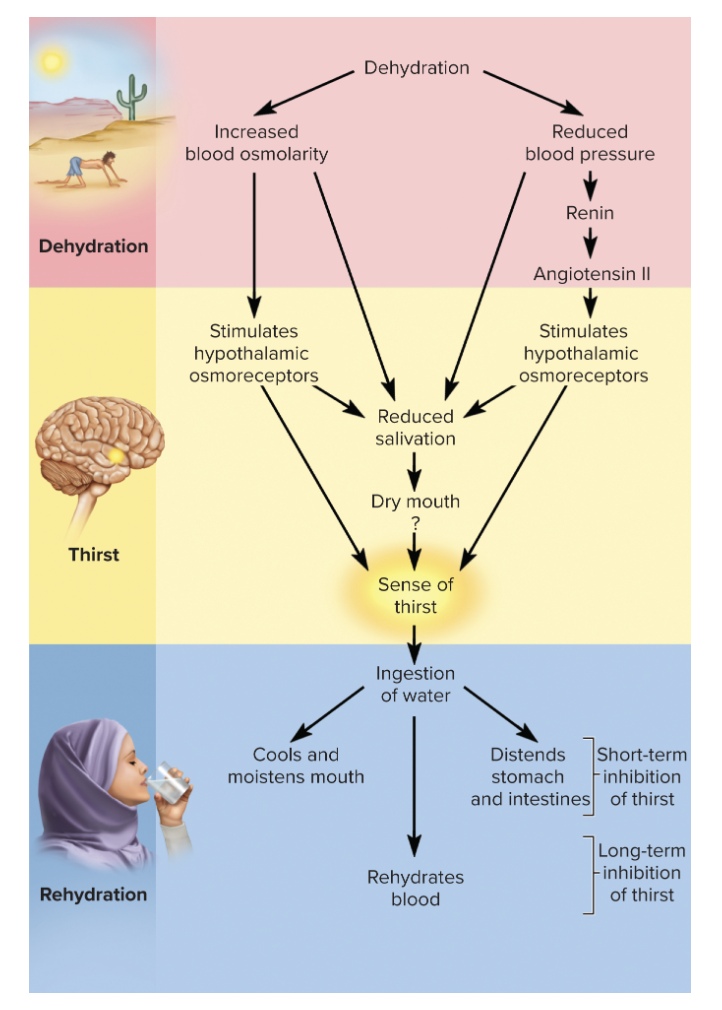
short term satiation of thirst
fast acting stimuli
coolness
moisture
filling of stomach
prevent an animal from drinking an excessive amount of liquid (effective for 30-45 mins)
regulation of output
only significant control water output is through variations in urine volume - usually linked to adjustments in sodium reabsorption
when sodium is reabsorbed or excreted, proportionate amounts of water accompany it
total volume of fluid remaining in body may change but osmolarity remains stable
kidney’s limitations in regulation of output
cant completely prevent water loss
cant replace lost water or electrolytes
never restore fluid volume or osmolarity
kidneys role in dehydration
support existing fluid levels and slow down the rate of loss until water and electrolytes are ingested
ADH in output regulation
provides control of water output independently of sodium
helps kidneys retain water
slows down decline in blood volume and rise in osmolarity
forms a negative feedback loop
Steps of how ADH contributes to output regulation
increased osmolarity of blood stimulates hypothalamic osmoreceptors - stimulate posterior pituitary to release ADH
cells of collecting ducts of kidneys synthesize aquaporins
serve as channels that allow water to diffuse out of duct into hypertonic tissue fluid of renal medulla
kidneys reabsorb more water and produce less urine
sodium continues to be excreted so the ratio of sodium to water in urine increases (urine becomes more concentrated)
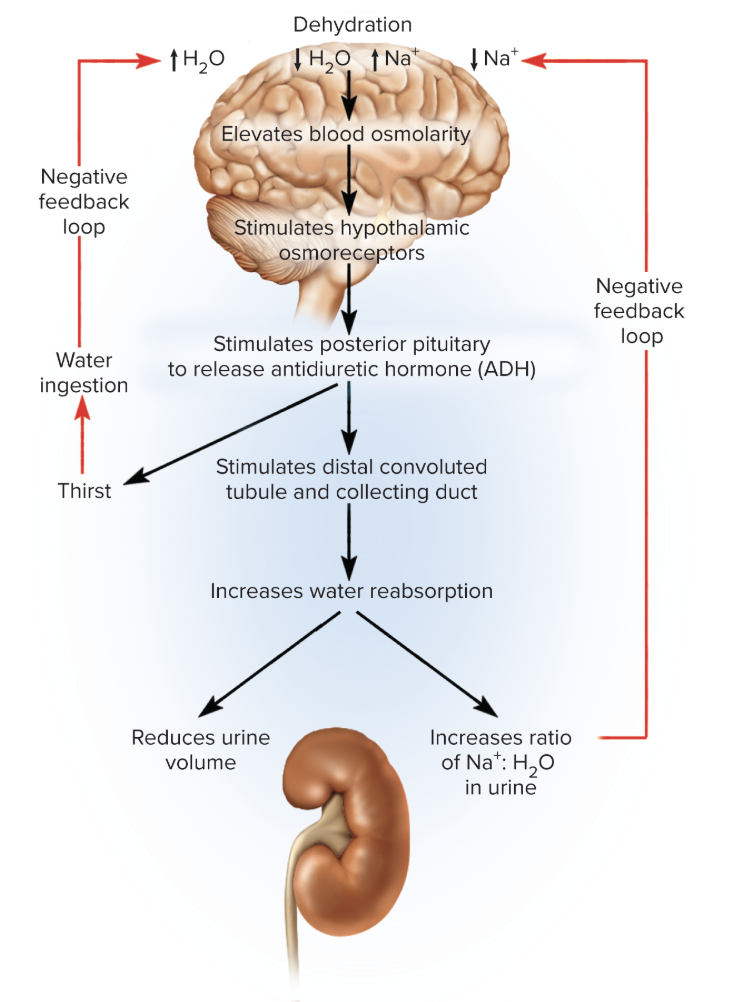
how does ADH create an effective way of compensating for hypertension?
if blood volume and pressure are too high, or blood osmolarity is too low, ADH release is inhibited
causes renal tubules to reabsorb less water
urine output increases, and total body water declines
lack of ADH increases the ratio of water to sodium in the urine, raising the sodium concentration and osmolarity of the blood
fluid imbalance
abnormality of total volume, concentration, or distribution of water among the compartments
kinds of fluid deficiency
differ in relative loss of water and electrolytes and the resulting osmolarity of the ECF; require different strategies of fluid replacement therapy
volume depletion
dehydration
volume depletion (hypovolemia)
occurs when proportionate amounts of water and sodium are lost without replacement
total body water declines but osmolarity remains normal
what is hypovolemia (volume depletion) caused by
hemorrhage
severe burns
chronic vomiting/diarrhea
aldosterone hyposecretion (results in inadequate sodium and water reabsorption by the kidneys)
Addison disease
aldosterone hyposecretion leading to inadequate sodium and water reabsorption by kidneys
dehydration (negative water balance)
occurs when body eliminates significantly more water than sodium
raises ECF osmolarity
causes of dejudration
lack of drinking water
diabetes mellitus
ADH hyposecretion (diabetes insipidus)
profuse sweating
overuse of diuretics
reasons infants are more vulnerable to dehydration
high metabolic rate produces toxic metabolites faster, excrete more water to eliminate them
kidneys aren’t fully mature and can’t concentrate urine as effectively
greater ratio of body surface to volume
lose twice as much water per kg of body weight by evaporation
what does dehydration affect
all fluid compartments
as blood loses water - osmolarity rises ad water from tissue fluid enters bloodstream to balance loss
high osmolarity of tissue fluid moves water out of cells to balance
all 3 fluid compartments (ICF, blood, tissue fluid) lose water
most serious effect of fluid deficiency
circulatory shock due to loss of blood volume and neurological dysfunction due to dehydration of brain cells
fluid excess
less common because kidneys are very effective at compensating for excessive intake by excreting more urine
renal failure and other causes can lead to excess fluid retention
2 types of fluid excess
volume excess
hypotonic hydration
volume excess
both sodium and water are retained and the ECF remains isotonic
results from aldosterone hypeorsecretion or renal failure
hypotonic hydration (water intoxication/positive water balance)
more water than sodium is retained or ingested and the ECF becomes hypotonic
can occur if you lose a large amount of water and salt through urine and you replace it by drinking plain water
without proportional intake of electrolytes, water dilutes ECF, and makes it hypotonic - inducing cellular swelling
how can ADH cause hypotonic hydration
hypersecretion stimulates excessive water retention as sodium continues to be excreted
fluid sequestration
condition in which excess fluid accumulates in a particular location
total body water and osmolarity may be normal, but volume of circulating blood may drop to the point of causing circulatory shock
causes of fluid sequestration
edema - most common
hemorrhage
pleural effusion
electrolyte balance
state in which the amount of electrolytes absorbed by the small intestine balances the amount lost from the body (mainly through urine) and in which electrolyte concentrations in the body fluids are regulated within homeostatic limits
why are electrolytes physiologically important
chemically reactive and participate in metabolism
determine the electrical potential (charge difference) across cell membranes
strongly affect osmolarity of the body fluids and body’s water content and distribution
major cations of electrolytes
sodium Na+
potassium K+
calcium Ca2+
magnesium Mg2+
hydrogen H+
major anions of the electrolytes
chloride Cl-
bicarbonate HCO3-
phosphates Pi
what is the most accessible fluid for measurements of electrolyte concentration?
blood plasma
does electrolyte concentration affect osmolarity between the two fluid compartments
no lol they have the same osmolarity (300 mOsm/L)
Sodium functions
principal ion responsible for resting membrane potentials of cells
inflow of sodium into cell is essential in depolarization driving nerve and muscle function
principal cation of ECF
most significant solute in determining total body water and the distribution of water among fluid compartments
sodium gradients across membrane provide potential energy needed for cotransport of other solutes
Na+-K+ pump is important mechanism in creating body heat
sodium bicarbonate plays major role in buffering pH of ECF
Sodium homeostasis
multiple mechanisms
tied to effects on blood pressure and osmolarity
coordinated by aldosterone, ADH, and natriuretic peptides
Aldosterone in Sodium Homeostasis
“salt retaining hormone”
primary role in adjustment of sodium excretion
primary effect = urine contains less NaCl and more potassium, decreasing its pH
hyperkalemia directly stimulates adrenal cortex to secrete aldosterone
hypotension stimulates secretion through renin-angiotensin-aldosterone mechanism
where are aldosterone receptors?
cells in:
ascending limb of nephron loop
DCT
cortical part of the CD
Mechanism of Aldosterone’s effect in Sodium Homeostasis
aldosterone binds to nuclear receptors and activates transcription of a gene for the Na+-K+ pump
enough pumps are synthesized and installed in plasma membrane to produce a noticeable effect
sodium concentration in urine falls and potassium concentration rises as tubules reabsorb more sodium and secrete more hydrogen & potassium ions
water and Cl passively follow sodium
what does aldosterone strongly influence?
sodium reabsorption - little effect on plasma sodium concentration because it is accompanied by proportional amount of water
what inhibits renin-angiotensin-aldosterone mechanism
high blood pressure
kidneys reabsorb almost no sodium beyond PCT
aldosterone only has small effects on:
urine volume
blood volume
blood pressure
increase in blood volume increases blood pressure and GFR
even tho aldosterone increases tubular reabsorption of sodium and water, it is offset by rise in GFR with only a small drop in urine output
Antidiuretic hormone and Sodium homeostasis
modifies water excretion independently of sodium excretion - enables it to change sodium concentration
stimulated by high concentration of sodium in blood
kidneys reabsorb more water, slows down further increase in blood sodium concentration
can’t lower concentration alone
inhibited by drop in sodium concentration
more water is excreted and raises the relative amount of sodium remaining in blood
secreted from posterior lobe of pituitary gland
natriuretic peptides and sodium homeostasis
inhibit sodium and water reabsorption and secretion of renin and ADH
eliminate more sodium and water and lower the blood pressure
angiotensin II and sodium homeostasis
activates Na+-H+ antiport in PCT and increases sodium reabsorption, reducing urinary sodium output
sodium imbalances
hypernatremia
hyponatremia
hypernatremia
plasma sodium concentration excess pf 145 mEq/L
results from administration of IV saline
major consequences = water retention, hypertension, and edema
hyponatremia
plasma sodium concentration of less than 130 mEq/L
usually result of excess body water instead of excess sodium secretion
quickly corrected by excretion of excess water
Potassium functions
most abundant cation of ICF
greatest determinant of intracellular osmolarity and cell volume
produces resting membrane potentials and action potentials of nerve and muscle cells
as important as sodium to Na+-K+ pump and its functions of cotransport and thermogenesis
essential cofactor for protein synthesis and other metabolic processes
Potassium Homeostasis
closely linked to sodium’s homeostasis (90% of K+ filtered by glomerulus is reabsorbed by PCT; rest excreted in urine)
excretion controlled later in nephron by changing amount of potassium returned to tubular fluid by DCT and cortical potion of CD
what happens when potassium concentration is high
tubules secrete more potassium into filtrate and the urine may contain more potassium than glomerulus can filter from blood
what happens when blood potassium level is low
tubules secrete less
DCT and CD reabsorb potassium through intercalated cells
aldosterone and potassium homeostasis
regulates potassium balance along with sodium
aldosterone secretion by adrenal cortex is stimulated by a rise in potassium concentration
aldosterone stimulates renal secretion of potassium at the same time it stimulates reabsorption of sodium
the more sodium the less potassium
potassium imbalances
most dangerous of all electrolyte imbalances
hyperkalemia
hypokalemia
hyperkalemia (quick and slow rises)
(>5.5 mEq/L) can have completely opposite effect depending on whether potassium concentration rises quickly or slowly
quick rise in extracellular potassium tends to make nerve and muscle cells abnormally excitable
less concentration difference between ICF and ECF - outward diffusion of K+ is reduced
normally it passes into and out of cells at equal rates through Na+-K+ pump
more K+ remains in cell than normal, plasma membrane has less negative resting potential and is closer to the threshold at which it will set off action potential
slow rise in extracellular potassium concentration - nerve and muscle become less excitable
slow depolarization of a cell inactivates voltage gated sodium channels
dont become excitable again until membrane repolarizes
inactivated sodium channels can’t produce action potentials
hypokalemia
(<3.5 mEq/L)
ECF concentration falls, more potassium moves from ICF to ECF
cells become hyperpolarized and nerve and muscle cells are less excitable
occurs in people with depressed appetite - heavy sweating, chronic vomiting, diarrhea, aldosterone hyposecretion, alkalosis, laxative abuse etc
functions of calcium
lends strength to skeleton
activates sliding filament mechanism of muscle contraction
serves as second messenger for some hormones and neurotransmitters
activates exocytosis of neurotransmitters and other cellular secretions
essential factor in blood clotting
** sustains ventricular contraction long enough to ensure effective ejection of blood
why do cells maintain low intracellular calcium concentration?
they require a high concentration of phosphate ions, and if calcium and phosphate were both concentrated inside a cell, calcium phosphate crystals would precipitate in cytoplasm
how do we avoid calcium phosphate crystals
to keep a high phosphate concentration and avoid crystallization of calcium phosphate
cells pump out Ca2+and keep it at a low intracellular concentration
also sequester Ca2+ in smooth ER and release it only when needed
calsequestrin
protein that binds stored Ca2+ and keeps it chemically unreactive
Calcium homeostasis
concentration regulated chiefly by:
parathyroid hormone
calcitriol (calcitonin in children)
work through effects on bone deposition and resorption, intestinal absorption of calcium, and urinary excretion
calcium imbalances
hypercalcemia
hypocalcemia
hypercalcemia
reduces sodium permeability of plasma membranes and inhibits depolarization of nerve and muscle cells
hypocalcemia
increases sodium permeability of plasma membranes, causing nervous and muscular systems to be overly excitable
Magnesium functions
second most abundant intracellular cation (after potassium)
has wide range of effects on membrane transport, membrane electrical potentials, cell metabolism, and DNA replication
magnesium homeostasis in intestine
intestinal absorption mainly regulated by vitamin D (only 30-40% gets reabsorbed, rest passes through)
2/3 lost via feces, 1/3 in urine
magnesium homeostasis in nephron
retention/loss of plasma magnesium is regulated by thick segment of ascending limb of nephron loop
smaller amounts reabsorbed in other segments of nephron
mainly through paracellular route (driven by positive electrical potential of tubular fluid repelling the positive magnesium ions)
parathyroid hormone effect on magnesium homeostasis
governs rate of reabsorption through paracellular route, being the primary regulator of plasma Mg2+ level
magnesium imbalances
usually due to excessive loss from the body
hypermagensemia
hypomagnesemia
hypermagnesemia
rare except in renal insufficiency
sedative effect, depresses everything
leading to lethargy, weakness, respiratory depression/failure, hypotension, cardiac arrest
hypomagnesia
results in hypoerirritability of nervous and muscular systems
chloride functions
most abundant anions of ECF and make a major contribution to osmolarity
required for formation of stomach acid
involved in chloride shift that accompanies CO2 loading and unloading by erythrocytes
major role in regulation of body pH
chloride homeostasis
strongly attracted to sodium, potassium, and calcium
achieved primarily as side effect of sodium homeostasis
(when sodium is retained or excreted, chloride ions passively follow)
chloride imbalances
primary effects are disturbances in acid-base balance
hyperchloremia
hypochloremia
hyperchloremia
result of dietary excess or administration of IV saline
hypochloremia
usually side effect of hyponatremia
sometimes results from hyperkalemia or acidosis
kidneys retain potassium by excreting more sodium
sodium takes chloride with it
phosphate functions
activate many metabolic pathways by phosphorylating enzymes and substrates
important buffers that help stabilize pH of body fluids
relatively concentrated in ICF
generated by hydrolysis of ATP and other phosphate compounds
component of phospholipids, DNA, RNA, ATP, GTP, cAMP, creatine phosphate, etc
inorganic phosphates of the body fluids are an equilibrium mixture of phosphate, monohydrogen phosphate, and dihydrogen phosphate
phosphate homeostasis
usually maintained with a continual loss of excess phosphate through glomerular filtration
readily absorbed by small intestine
a plasma phosphate concentration drop causes renal tubuls to reabsorbed all filtered phosphate
parathyroid hormone and phosphate homeostasis
increases excretion of phosphate as part of mechanism for increasing concentration of free calcium ions in the ECF
lowering ECF phosphate concentration minimizes formation of calcium phosphate and helps support plasma calcium concentration
phosphate excretion rates strongly affected by pH of urine
phosphate imbalances
not as critical as other electrolytes
body can tolerate broad variations with little immediate effect on physiology