organic chem yo
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms

alkene
H2 Ni catalyst ; recduction
alkenes → alkanes
(alkanes) CH4 + = CO2 + 2H2O
[E] O2 ; combustion
(alkanes) [E] CH4 + = CH3X + HX (halogenalkanes)
Limited X2 (Cl/Br) uv light ; FRS

alkene → HALOGENALKANES
HX(g) (Cl/Br/I) rt HI > HBr > HCl ; EA
HI has highest reactivity bc of lowest bond energy
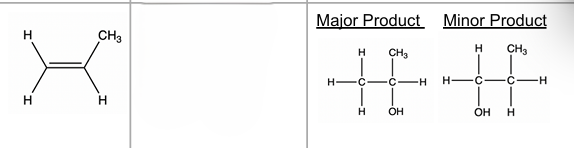
alkene → ALCOHOL
H2O (g), conc H3PO4, high T&P ; EA

alkene → HALOGENALKANES
X2 (Cl/Br) in CCl4, rt ; EA
pale yellow Cl2 decolourise
Orange-red Br2 decolourises
(absence of uv light to avoid FRS)

nt v imporatant
ICl in CCl4, rt ; EA

alkene → HALOGENALKENE & ALCOHOL
Br2 (aq), rt ; EA
orange Br2(aq) decolourises
test of presence of alkene

alkene → DIOL
Dilute KMnO4 (aq), NaOH (aq) COLD ; mild oxidation
……………………..H2SO4 (aq)

alkene → ketone, carb acid, produce CO2
KMnO4 (aq), H2SO4 (aq) Heat ; Oxidative cleavage
Step Down
C=C → C=O
H → OH
R → R
=CH2 → CO2 + H2O

benzene → HALOGENOARANE
X2, anhydrous FeX3 / AlX3 catalyst, rt ; ES

benzene → NITRObenzene
Benzene : conc HNO3, conc H2SO4, 50dc ; ES
alkylbenzene : conc HNO3, conc H2SO4, 30dc ; ES
electron-donating methyl grp 2,4-directing

nt important
conc HNO3, conc H2SO4, >50dc ; ES
COOH is electron-withdrawing grp 3-directing

benzene → ALKYLbenzene
RX (halogenoalkane), anhydrous AlX3 catalyst, rt ; ES

side chain on benzene to -COOH
KMnO4 (aq), H2SO4 (aq) Heat under reflux ; Side chain oxidation
first carbon directly attached to benzene must have at least one H
If alkaline KMnO4, -COOH grp → -COO- (acid base rxn)
Side Products :
R has x1 C, extra CO2 + H2O
R has x2 C, extra CH3COOH

ALKANE (side chain) → halogenalkane
Limited X2 (Cl/Br), uv light ; FRS

Halogenoalkane → alcohol
NaOH (aq), Heat ; NS
KOH (aq)

halogenoalkane → alkene
NaOH in ethanol, heat ; Elimination
Ethanolic KOH

halogenoalkane → AMINE
[E] ethanolic NH3, heated under pressure (heat in sealed tube) ; NS
2 steps :
NS of RX → R-NH3+
rmv H+ to form R-NH2

amine (not important)
Ethanolic MEDIUM, heated under pressure (heat in sealed tube)

halogenoalkane → -CN (nitrile)
Ethanolic NaCN, heat ; NS
STEP UP RXN
ethanolic KCN can be used

resistance to NS
NaOH (aq) Heat
OR
[E] ethanolic NH3, heat under pressure/sealed tube
OR
ethanolic NaCN heat
partial double bond
additional rsn : hgih electron density of ring repels necleophile

Xtra
conc H2SO4, conc HNO3, >50dc ; ES
electron-withdrawing halogen grp is 2,4-direction

-CN → amine
LiAlH4 (in dry ether) ; Reduction
OR
H2(g), nickel catalyst

-CN → -COO- (deprotonated form)
NaOH (aq), heat ; Alkaline Hydrolysis
to obtain protonated form,
use aqueous acid such as HCl (aq) or H2SO4 (aq) (AB rxn)

-CN → -COOH
HCl (aq), heat ; Acidic Hydrolysis
H2SO4 (aq), heat