CH2 (Atomic Theories and the Periodic Table)
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
2.1 Development of Theories of Structure or Matter
Democritus: matter is composed of tiny indivisible particles called atoms.
Aristotle: vacuum doesn’t exist & matter is composed of earth, air, fire, and water.
Atomic Models:
Dalton (compact atom): all matter is composed of tiny indivisible particles called atoms.
Thompson (charged atom): atoms contain positive and negative charges.
Rutherford (nuclear atom): atom has positive, small, and massive nucleus.
Bohr (planetary atom): atom contains nucleus (protons and neutrons) surrounded by shells with electrons.
Modern Model (electron cloud): central positive nucleus and negative electron cloud.
2.2 The Atom and Electrons
Atom is the smallest part that keeps the element properties.
extremely small atomic volume that can only be imaged by Scanning Tunneling Microscope (STM)
Electron is a small negative subatomic particle moving around the nucleus.
discovered by Thompson using Cathode Ray Tube.
Electron Cloud is an instant shot that describes the movement of an electron around the nucleus.
Energy Level is the region with high probability to find the electron around the nucleus.
2.3 Main Energy Levels
Principal Quantum Number (n = 1, 2, .. 7): represents the different levels.
Main energy level (shell) can contain different sublevels (subshells).
Sublevels are divided into orbitals and every orbital can be filled by 2 electrons.
s: 1 oribtal and 2e (spherical)
p: 3 orbital and 3×2=6e (dumbbell)
d: 5 orbital and 5×2=10e (cloverleaf)
f: 7 orbital and 7×2=14e (complex)
Max number of electrons in a shell = 2n². (ex. n=4 is 2(4)² = 32e)
2.4 Electron Configuration in the Ground State
Aufbau Principle: electrons fill lower-energy atomic orbitals before filling higher-energy ones.
Pauli’s Exclusion Principle: orbitals can be filled by a maximum of 2 electrons in OPPOSITE spins.
Hund’s Rule: every orbital in a sublevel is SINGLY occupied before any orbital is doubly occupied.
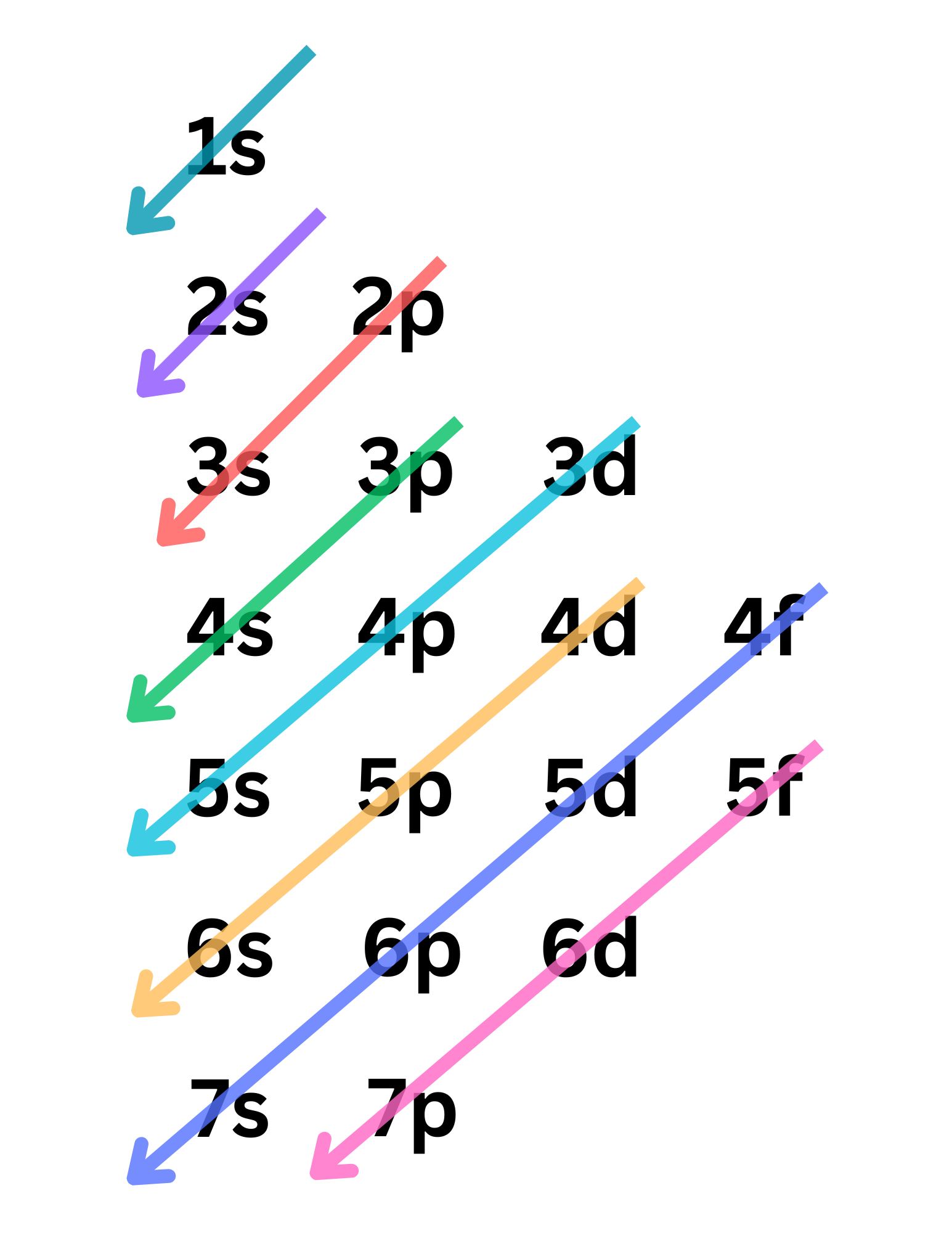
2.5 Some Special Electron Configuration
24Cr = [Ar] 4s1 3d5
29Cu = [Ar] 4s1 3d10
47Ag = [Kr] 5s1 4d10
11Na = 1s2 2s2 2p6 3s1 → Na+ = 1s2 2s2 2p6 3s1
22Ti = [Ar] 4s2 3d2 → Ti2+ = [Ar] 3d2
29Cu = [Ar] 4s1 3d10 → Cu2+ = [Ar] 3d9
2.6 Valence Electrons and Lewis Structures
Valence Electrons: are electrons in the last energy level. (ex. 7N 1s2 2s2 2p3 has 5e)
Electron dot diagrams: atom symbol surrounded by valence electrons as dots, called a Lewis Structure.
Atom is surrounded by 8e except for hydrogen.
CO2, CH4, NH3, H2O.
Some can be stable with fewer or more than 8e.
BH3 surrounded by 6e, PCl5 surrounded by 10e.
2.7 The Modern Periodic Table
Periodic Table consists of 7 periods and 17 groups.
3 Major Groups are: metals, nonmetals, semimetals.
4 Blocks: s, p, d, f
Group Names:
G1 (alkali metal): highly reactive, lose 1e to form +1
G2 (alkaline metal): reactive, lose 2e to form +2
G3-12 (transition metals): have multiple positive chargers and colored salt
G17 (halogens): highly reactive, gain 1e to form -1
G18 (noble gases): completely unreactive, have 8 valence electrons
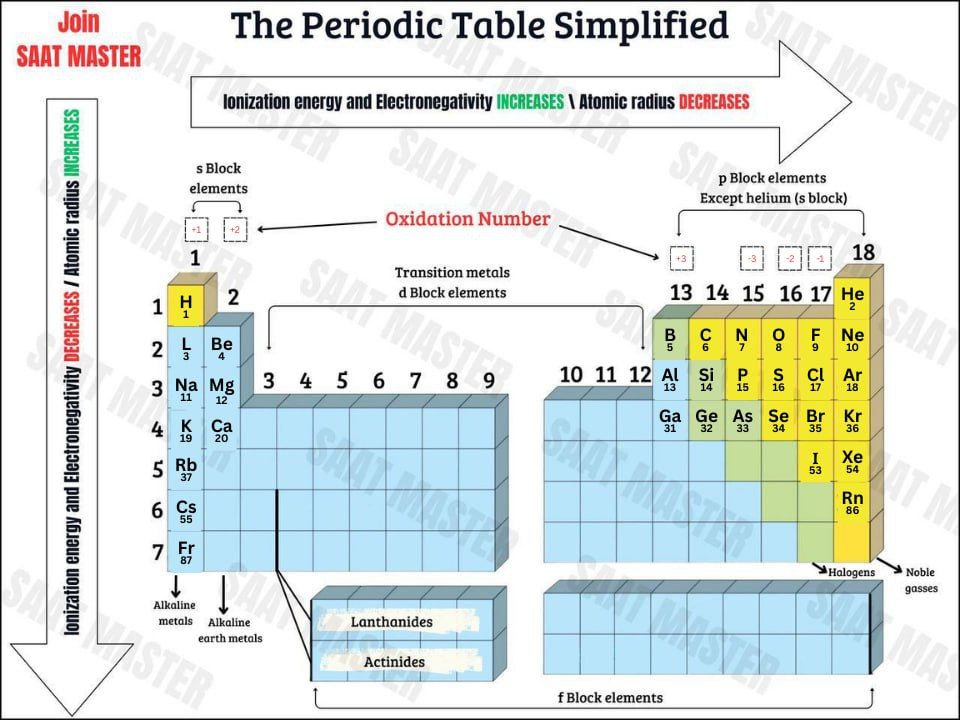
2.8 Blocks and Position of Element in the Period Table
s-block: 2 groups (1 and 2) & ends in s1 to s2
p-block: 6 groups (13 to 18) & ends in p1 to p6
d-block: 10 groups (3 to 12) & ends in d1 to d10
f-block: 14 groups (inner transition metals) & ends in f1 to f14
period number: number of the highest level in electron configuration.
group number: s block (electrons in last shell), p block (electrons in last shell of s and p + 10), d-block (electrons of last s and d).
Trends in the Periodic Table
Atomic Radius: half the distance between two nuclei in an element.
Increases diagonally up.
Ionization Energy: energy required to remove an electron completely from an atom in gaseous state.
Decreases diagonally down.
First ionization energy: remove electron from neutral atom to become +1 ion.
Second ionization energy: remove second electron from +1 ion.
Second is larger than first due to difference in charge with higher positive charge.
Electronegativity: ability of an atom to attract the bonding electrons.
Decreases diagonally down.