10 Elementary Steps
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
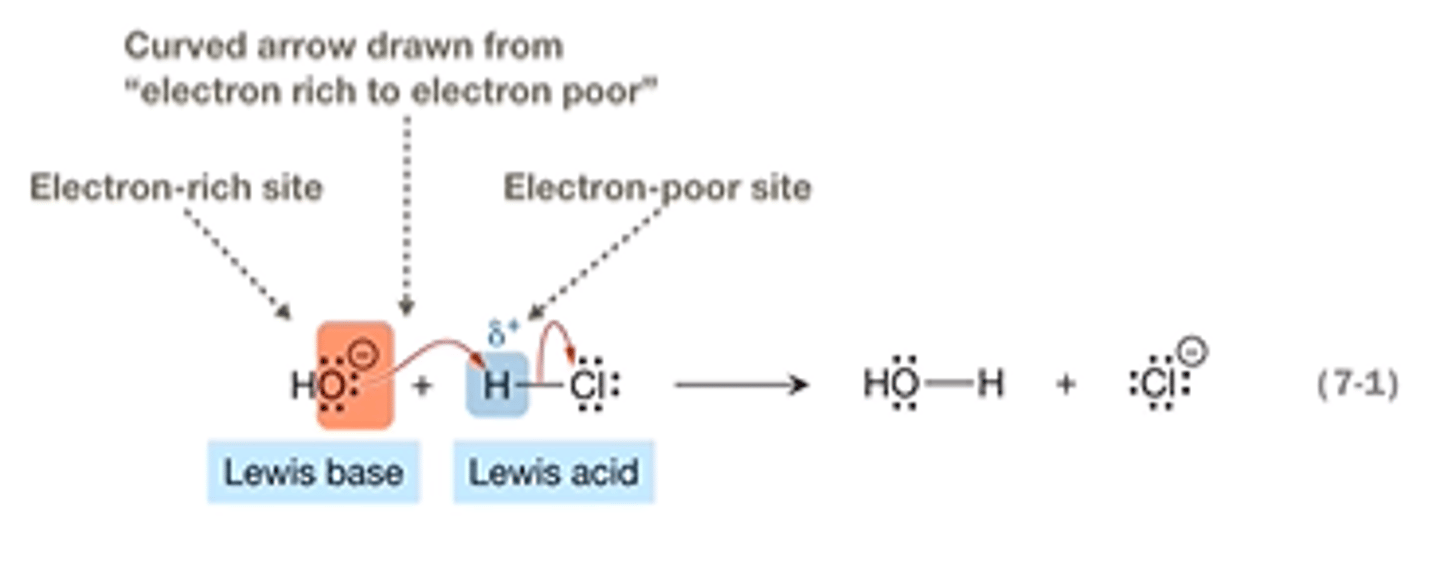
proton transfer step
bronsted acid donates H+ to a base forming a conjugate acid and conjugate base.
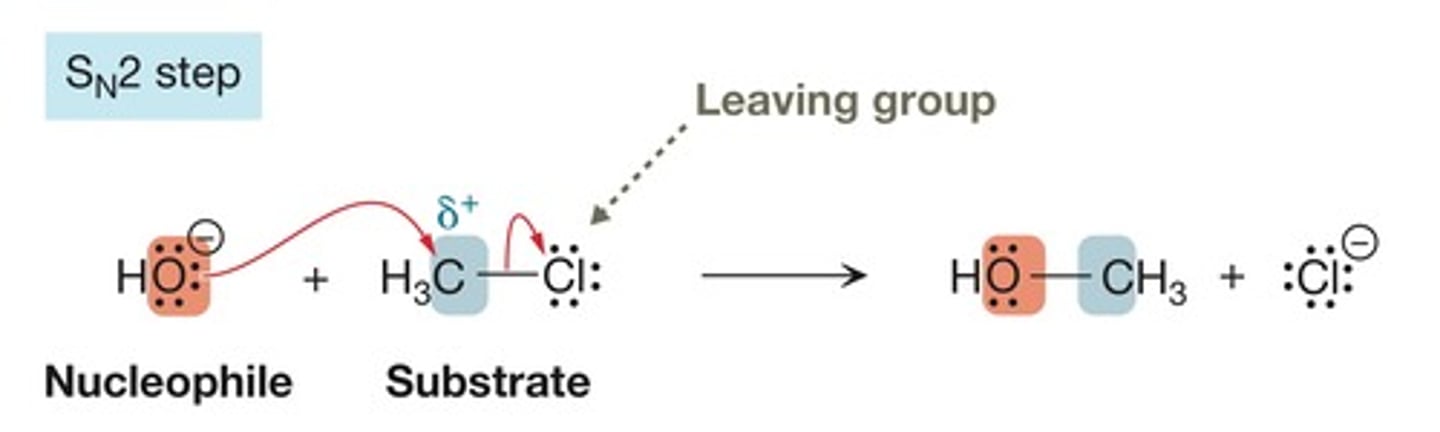
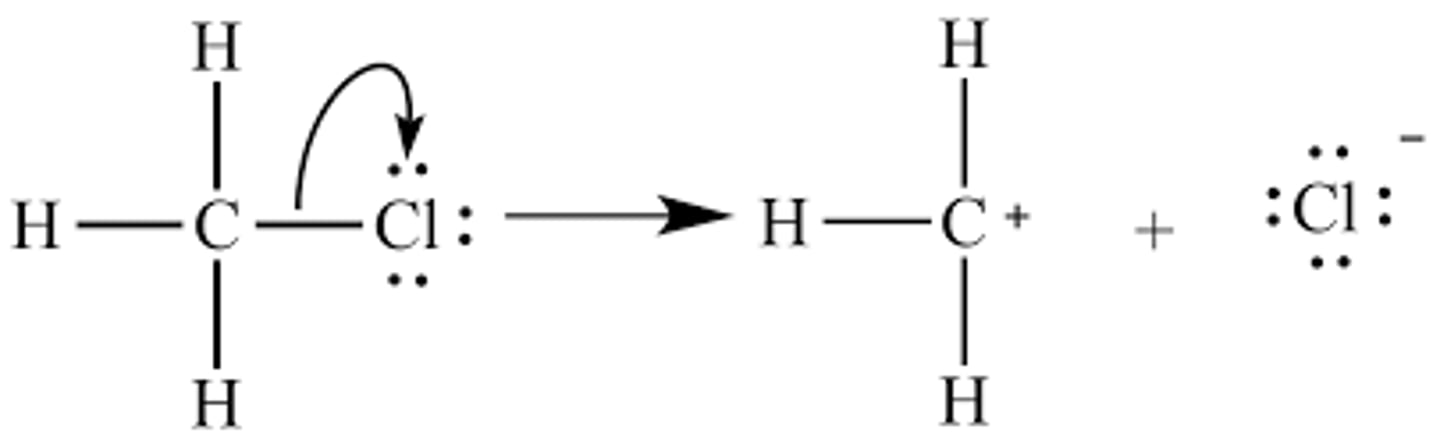
Sn2 step
substitution, nucleophilic, bimolecular
nucleophile bonds to substrate while bond to leaving group is broken
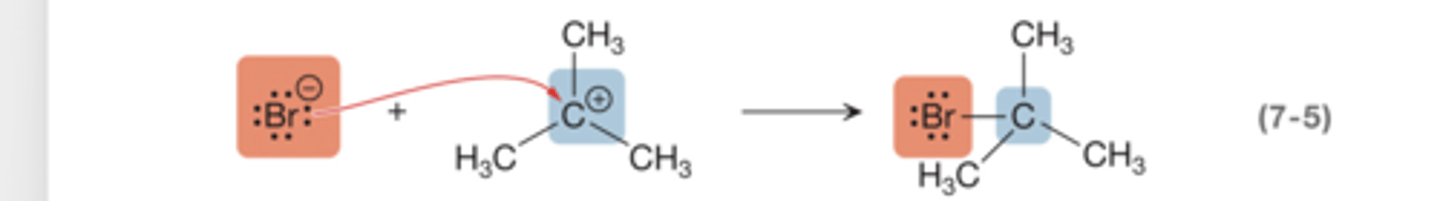
coordination
forms a single covalent bond (no bond breaking)
Lewis acid: accepts e's
Lewis base: donates e's
Lewis acid and base combine
Heterolysis
opposite of coordination, breaks a single covalent bond
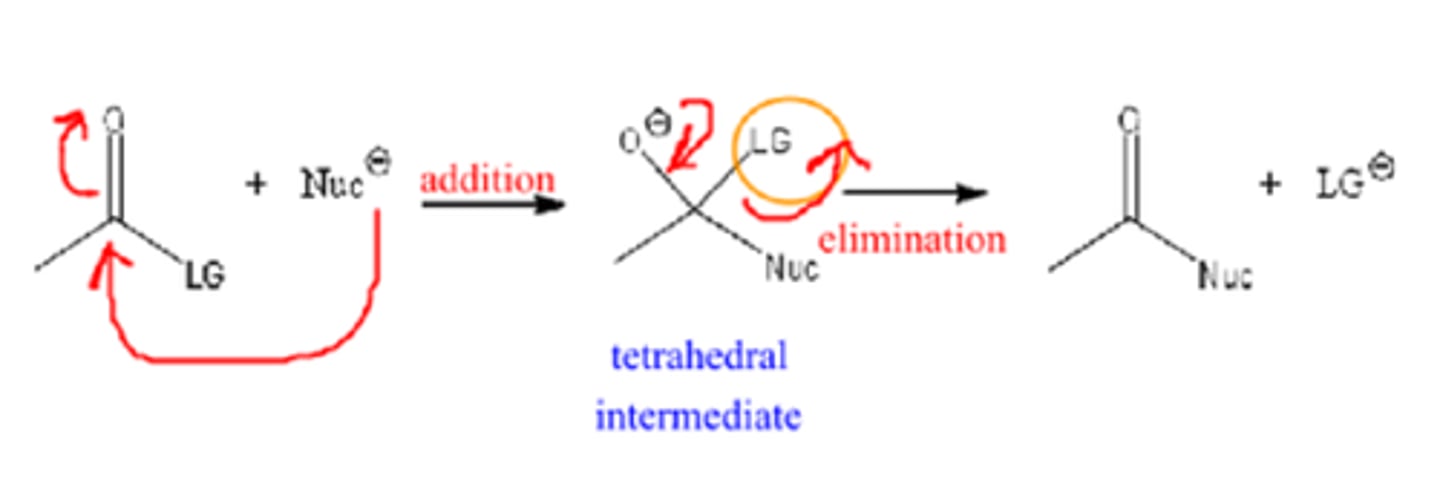
nucleophilic addition
forms a covalent bond and breaks a pi bond
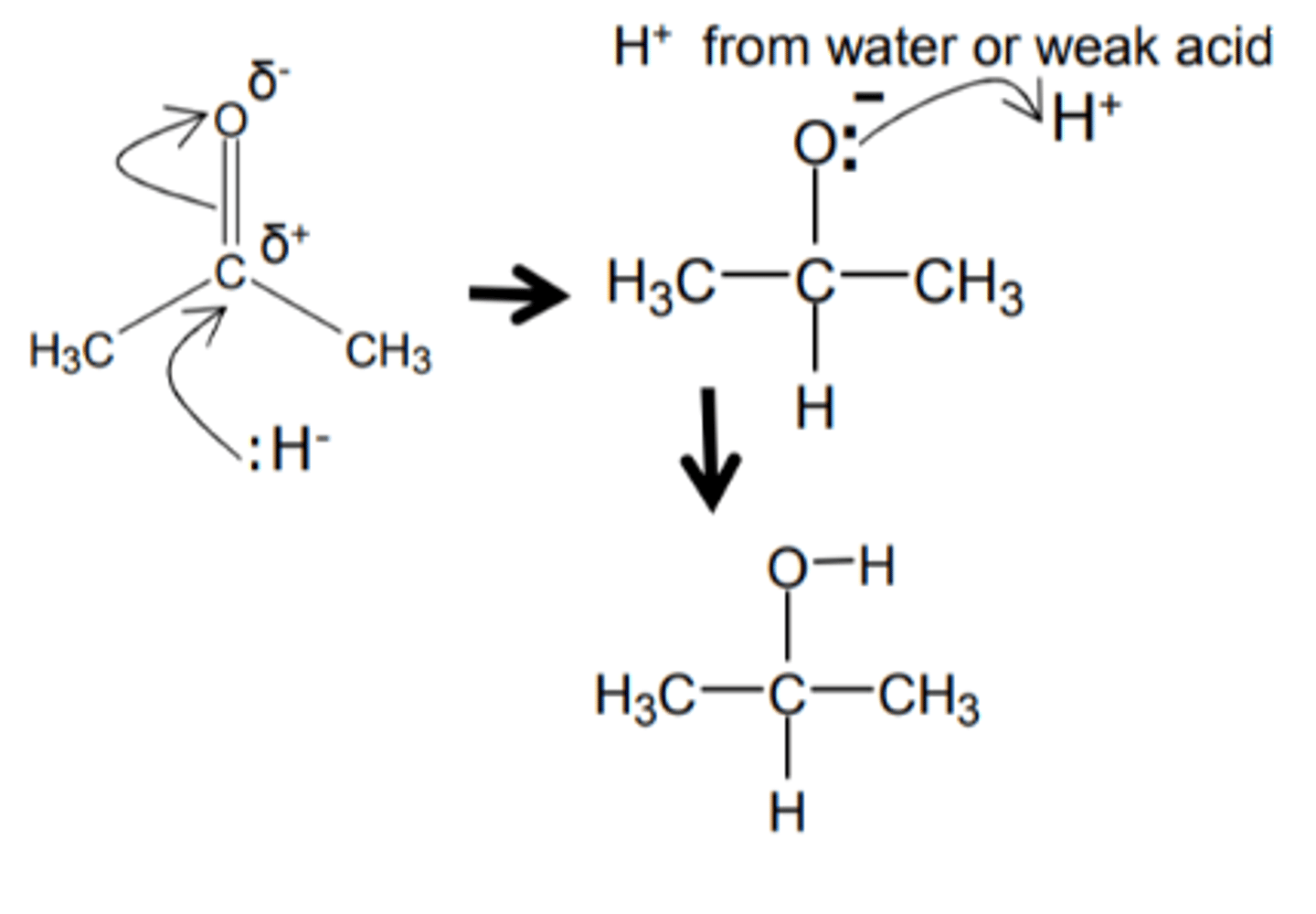
nucleophilic elimination
form a pi bond and break a covalent sigma bond
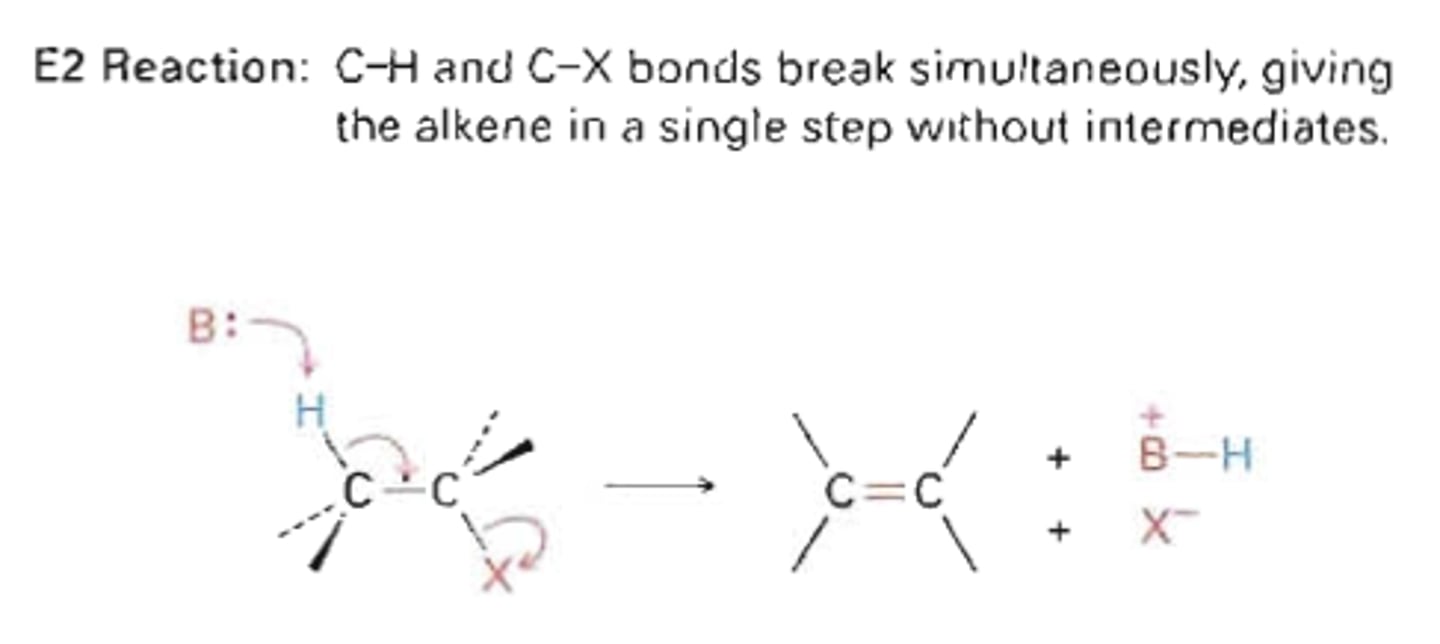
bimolecular elimination (E2)
proton and a leaving group are removed from substrate resulting in a new double or triple bond
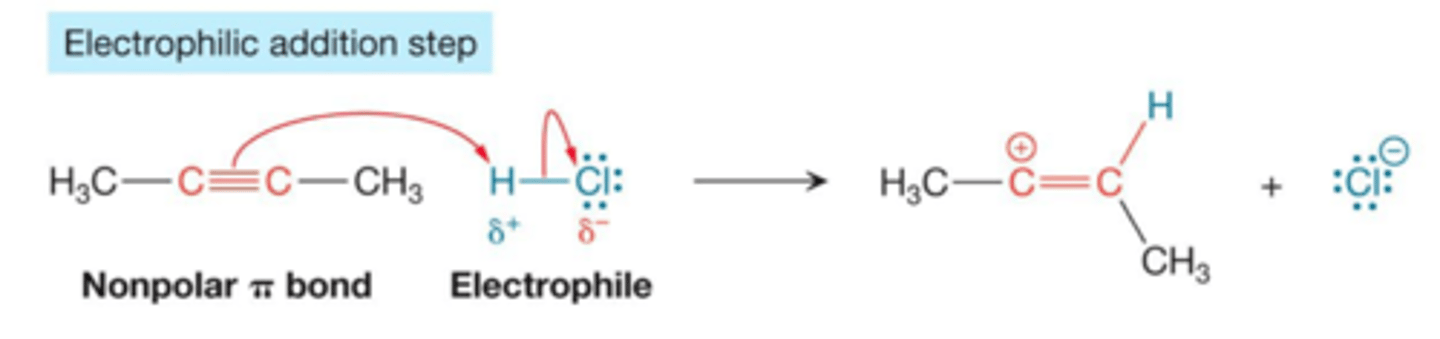
electrophile addition
use non polar pi bonds to engage w/ electrophiles.
creates a carbocation
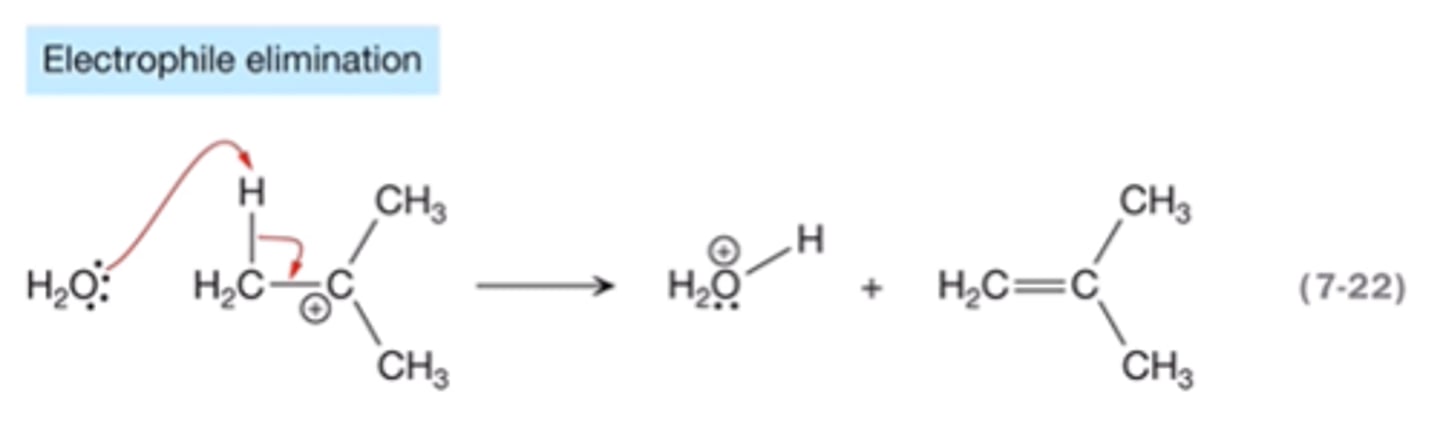
electrophile elimination
eliminates a proton from a carbocation. Froms a new double or triple bond
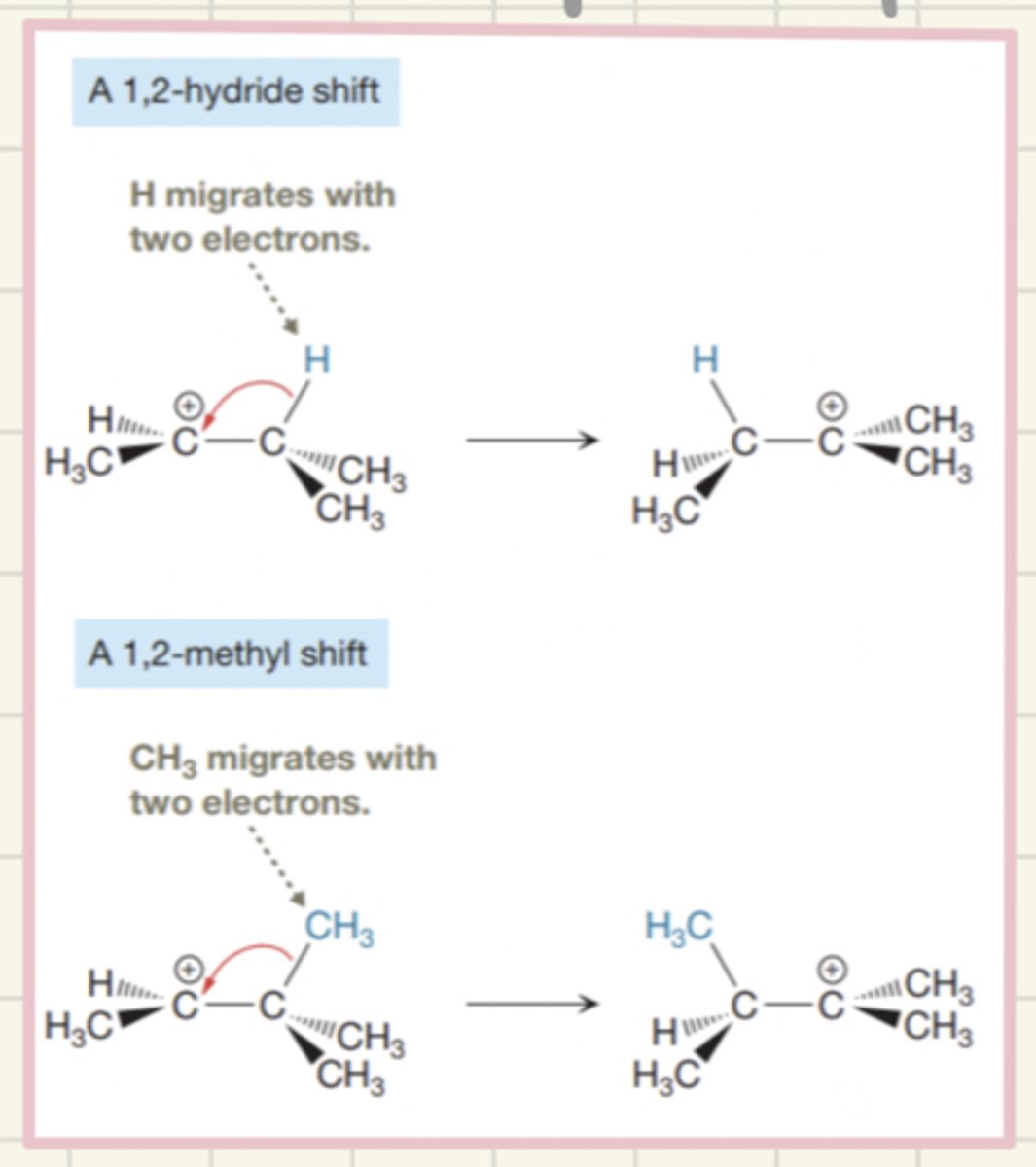
carbocation rearrangements
hydride shift: H atom moves
alkyl shift: anything migrating that isn't H.
only rearrangements that give more stable carbocation (3) will occur.