Chem of Life Final (Biochem)
1/121
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
122 Terms
What is a polypeptide?
Unit 1
a linear chain of many amino acids (typically linked together by peptide bonds). It is the raw chain that folds into a functional protein.
What is the peptide bond?
Unit 1
a covalent bond (C–N) formed between the carboxyl group of one amino acid and the amino group of the next, via a condensation reaction (loss of water).
What are the two termini of a polypeptide?
Unit 1
N-terminus (amino terminus): the free amino group at the beginning of the chain.
C-terminus (carboxyl terminus): the free carboxyl group at the end of the chain.
What makes up the backbone of a polypeptide?
Unit 1
the repeating sequence of covalently linked –N– Ca–C– atoms, consisting of the amino nitrogen (N), alpha carbon (Ca), and carbonyl carbon (C). The side chains (R-groups) protrude from this backbone at the alpha carbon.
What is a zwitterion?
Unit 1
a molecule, such as a polypeptide, that contains both positive and negative charges; at physiological pH, the amino terminus exists as a positively charged ammonium ion (NH3+) and carboxyl terminus as a negatively charged carboxylate ion (COO–)
What is a peptide residue?
Unit 1
an amino acid that has been incorporated into a polypeptide chain (it loses a water molecule during formation).
What are the three components of a nucleotide?
Unit 1
a phosphate group, a deoxyribose sugar, and a nitrogenous base.
Which bases pair together in DNA?
Unit 1
• adenine (A) pairs with thymine (T), and cytosine (C) pairs with guanine (G).
What type of bond holds the base pairs together?
Unit 1
hydrogen bonds (weak bonds).
What type of bond holds the sugar-phosphate backbone together?
Unit 1
covalent bonds (phosphodiester bonds).
Which base pairs have two hydrogen bonds and which have three?
Unit 1
A-T pairs have two hydrogen bonds, while C-G pairs have three.
What is the difference between purines and pyrimidines?
Unit 1
purines (adenine, guanine) have a double-ring structure, while pyrimidines (thymine, cytosine) have a single-ring structure. A purine pairs with a pyrimidine in the Watson-Crick base-pair scheme.
Why is the DNA backbone considered "sugar-phosphate" rather than just "sugar"?
Unit 1
the phosphate group links the 3' carbon of one sugar to the 5' carbon of the next, providing the structural link.
What is the difference between ribose and deoxyribose?
Unit 1
ribose (in RNA) has hydroxyl (-OH) groups on both the 2 ́and 3 ́carbons, whereas deoxyribose (in DNA) lacks the 2 ́-oxygen (it has only a hydrogen at the position).
What makes RNA different from DNA chemically?
Unit 1
sugar: RNA contains ribose, while DNA contains deoxyribose.
base: RNA contains uracil instead of thymine (with a hydrogen in place of a methyl group)
How does deoxyribose connect to other components?
Unit 1
the 1 ́ carbon binds to the nitrogenous base, while the 3 ́ and 5 ́ carbons link to phosphate groups to form the sugar-phosphate backbone.
The leading strand of a DNA molecule has the following sequence:5’-CGCATG-3’. What is the sequence of the complementary strand?
Unit 1
5 ́-CATGCG-3 ́
How are the carbon atoms in deoxyribose numbered?
Unit 1
they are numbered 1 ́ to 5 ́ to distinguish them from the atoms in nitrogenous bases.
Which carbon atom in the deoxyribose ring is located outside the five-membered ring?
Unit 1
the 5 ́ carbon is the one located outside the ring structure.
To which carbon atoms do the phosphate group and nitrogenous base attach?
Unit 1
the nitrogenous base attaches to the 1 ́ carbon, and the phosphate group attaches to the 5 ́ carbon
(and the 3 ́ carbon of the preceding nucleotide).
Which of the following statements about deoxyribose is true?A) It contains six carbon atoms.
B) It has a hydroxyl group at the 2 ́ position.
C) It is the sugar component found in both DNA and RNA.
D) It forms part of the sugar-phosphate backbone in DNA.
Unit 1
D) It forms part of the sugar-phosphate backbone in DNA.
A) is incorrect because it is a pentose (5-carbon sugar).
B) is incorrect because deoxyribose specifically lacks the hydroxyl group at the 2' position.
C) is incorrect because RNA contains ribose, not deoxyribose.
What are the three types of chemical variables used to describe biopolymer structure?
Unit 1
the bond lengths between covalently linked atoms
the valence angles between successive chemical bonds
the torsion angles about rotatable (single) bonds.
What are two conflicting roles of DNA structure?
Unit 1
protect the genetic information encoded in the primary sequence of bases
expose the bases to the cellular machinery that copies/manipulates the genetic information
What is the structural brilliance behind the Watson-Crick model of base pairing?
Unit 1
four possible base pairs fit into the same space
What is transcription?
Unit 1
the fundamental biological process of copying a specific segment of DNA into a new, complementary messenger RNA (mRNA) molecule
What is the Protein Data Bank?
Unit 1
a worldwide repository of 3D structural data for large biological molecules (proteins, DNA, RNA)
Why is the sugar ring of DNA or RNA flexible?
Unit 1
it is a five-membered ring (pentose) that cannot lie completely flat, forcing it to "pucker
Which component of DNA carries the genetic features?
Unit 1
the distribution of electrons (charge accumulation) on the neutral bases
What are the sides of the DNA ladder made of?
Unit 2
sugar and phosphate molecules
Which bases pair in DNA?
Unit 2
adenine pairs with thymine (A-T), and guanine pairs with cytosine (G-C).
How do the bases bond together?
Unit 2
Through hydrogen bonds, with A-T forming two bonds and C-G forming three.
How many base pairs are in one turn of the Watson-Crick DNA helix?
Unit 2
• One turn consists of 10 base pairs.
What does "antiparallel" mean?
Unit 2
The two strands of the double helix run in opposite directions.
Who discovered the structure of DNA?
Unit 2
James Watson and Francis Crick, utilizing X-ray fiber diffraction data from Rosalind Franklin.
What is the structure of the DNA backbone?
Unit 2
The backbone is composed of alternating phosphate groups and deoxyribose sugars.
What is the sequence of the strand complementary to 5 ́-ATGCGTA-3 ́?
Unit 2
3 ́-TACGCAT-5 ́
How many hydrogen bonds hold a segment with five adenines (paired with five thymines) and ten guanines (paired with ten cytosines in place?
Unit 2
5 A-T ́ 2 hb/A-T + 10 G-C ́ 3 hb/G-C = 10 hb + 30 hb = 40 hb
How many base pairs (bp) constitute five turns of a Watson-Crick DNA double helix?
Unit 2
• 10bp/turn * 5turns=50bp
Why is DNA in a double helical form, i.e., a twisted ladder, rather than a straight ladder ?
Unit 2
twisting allows the hydrophobic (water-hating) nitrogenous bases to stack against one another on the inside of the structure and keep the hydrophilic (water-loving) sugar-phosphate backbone remains on the outside.
What stabilizes the DNA helix?
Unit 2
The helix is stabilized by hydrophobic interactions (base stacking) on the inside and hydrophilic interactions with water on the outside.
What is Chargaff's rule?
Unit 2
The amount of adenine equals thymine (A = T), and guanine equals cytosine (G = C).
What is Z-DNA?
Unit 2
a left-handed double helix narrower than B-DNA, with 12 base pairs per turn.
What is fundamental difference between a right-handed and left-handed helix?
Unit 2
the direction of rotation (chirality) in which the spiral twists as it moves away from an observer.
a right-handed helix coils in a clockwise direction when viewed moving away from the observer. It is
shaped like a standard screw thread or a typical spiral staircase that you climb while turning to the
right.
a left-handed helix coils in a counterclockwise (anticlockwise) direction moving away from the
observer. It is the mirror image of a right-handed helix and cannot be rotated to look like one.
What is the primary difference between the major and minor grooves in the Watson-Crick model of DNA?
Unit 2
The primary difference between the major and minor grooves of DNA is their width and accessibility to
genetic information. The major groove is significantly wider (approx. 22 Å) and deeper than the minor groove (approx. 12 Å), allowing proteins to more easily "read" specific base-pair sequences without unwinding the helix.
How do the dimensions of these grooves change in different forms of DNA?
Unit 2
in A-DNA, the major groove becomes very deep and narrow, while the minor groove becomes wide and shallow
What is the difference between A and B DNA?
Unit 2
The primary difference between A-DNA and B-DNA is that B-DNA is the standard, relaxed, right- handed helix found in living cells, while A-DNA is a more compact, dehydrated right-handed form that occurs under low-humidity conditions or during certain biological interactions. B-DNA is longer and thinner, whereas A-DNA is shorter and wider.
How are base pair positioned in A vs. B DNA?
Unit 2
Base pairs in A-DNA are tilted, shifted away from the center, and stacked closely together, whereas in B-DNA they are perpendicular to the axis, centered, and stacked farther apart. A-DNA has a distinct hollow core because of this displacement.
Why are there 10 rather than 16 unique base pair steps (or dinucleotide steps) in B-DNA?
Unit 2
While there are 16 possible combinations of two consecutive base pairs, the structural symmetry of the DNA double helix reduces the number of unique pairs to 10. Certain steps are structural equivalents of their reverse complement (e.g., CA/TG is the same as TG/CA).
What is a DNA palindrome?
Unit 2
a double-stranded sequence that reads the same 5 ́ to 3' ́on both strands, matching its own reverse complement. The 5 ́-3 ́ sequence of the top strand is identical to the 5 ́-3 ́ sequence of the bottom (complementary) strand.
What is a DNA junction, also called a Holliday junction?
Unit 2
a four-way cross- or X-shaped structure where two double-stranded DNA molecules are linked by the exchange of strands
What is a G-quadruplex, and which metallic cation is most effective at stabilizing its structure?
Unit 2
a four-stranded secondary structure formed in guanine-rich nucleic acid sequences, composed of stacked G-tetrads (four guanines arranged in non-Watson-Crick base pairs). Potassium (K+) ions are most effective for stabilization
A sample of double-stranded DNA is analyzed and found to contain 20% cytosine. What percentage of the DNA is adenine?
Unit 2
If C=20%, then G=20%. The remaining 60% is split equally between A and T (30% each)
How long is the DNA in one human cell if the DNA is fully stretched out as a B-form double helix?
Unit 3
If you uncoil the DNA from a single diploid cell (a cell with two complete sets of chromosomes, one set inherited from each parent) and lay it end-to-end, it would measure about 6.7 feet.
2 chrm-set ́ 3,000,000,000 bp/chrm-set ́ 3.4 Å/bp ́ (1 cm/108 Å) ́ (1 in/2.54 cm) ́ (1 ft/12 in) = 6.7 ft
How long is the DNA in the entire human body?
Unit 3
Estimates vary based on cell count, but with 3 x10^13 cells, the total length of all DNA strands in your body would stretch about 38 billion miles. This is enough to travel from the Earth to the Sun (~90 million miles one way) over 400 times.
3 ́1013 cells ́ 6.7 ft/cell ́ (1 mi/5280 ft) = 38,027,916,965 mi ́ (1 trip/90,000,000 mi) = 422.5 trips
What is a histone?
Unit 3
is a highly conserved, positively charged protein that binds to negatively charged DNA. Assemblies of histones act as spools around which DNA wraps to form structural units called nucleosomes, making it possible for long DNA molecules to be packed into the cell nucleus.
What are the components of a nucleosome?
Unit 3
• A nucleosome wraps ~150 base pairs of DNA around eight histone proteins, two copies each of H2A, H2B, H3, and H4.
What is the basic structural unit of chromatin called?
Unit 3
Nucleosome
What are the main functions of chromatin?
Unit 3
DNA packaging and the regulation of gene expression.
What is the role of nucleosomes in gene expression?
Unit 3
condense DNA and regulate access to genetic information; tightly packed nucleosomes generally inhibit transcription, while looser ones allow it.
What are the two types of chromatin?
Unit 3
Euchromatin, which is open and active, and heterochromatin, which is highly condensed and inactive.
What features of histone proteins allow them to bind tightly to DNA?
Unit 3
Histones are rich in basic amino acids, giving them a net positive charge that attracts the negatively charged phosphate backbone of DNA. They also contain short a-helices that fit into the DNA grooves.
Which amino acids carry a positive charge?
Unit 3
The basic amino acids are lysine (Lys, K), arginine (Arg, R), and histidine (His, H). They are positively charged (cationic) because their side chains are protonated at pH 7.
What is the role of linker DNA?
Unit 3
connects adjacent nucleosomes. The length of the linker determines the orientation of successive nucleosomes and the overall state (open or closed) of chromatin.
What stabilizes the a-helix structure?
Unit 3
• The a-helix is primarily stabilized by hydrogen bonds between the carbonyl oxygen of one amino acid and the amide hydrogen of the amino acid four residues ahead (i to i+4).
How are the R groups (side chains) of an a-helix oriented?
Unit 3
The R groups point radially outward from the helix axis to minimize steric hindrance and maximize stability.
What is the "handedness" of a typical a-helix?
Unit 3
Nearly all a-helices in Nature are right-handed.
How many residues make up a complete turn of an a-helix?
Unit 3
There are exactly 3.6 amino acid residues per complete turn of the a-helix.
What is the total length of an a-helix consisting of 36 amino acids given the rise (height per residue) of an a-helix is 1.5 Å?
Unit 3
Number of turns: 36 aa ́ (1 turn/3.6 aa) = 10 turns
• Total length: 36 × 1.5 Å/aa = 54 ÅÅ
How many water molecules are lost when forming a polypeptide of 100 residues?
Unit 3
• 99 (always n–1)
Why can't peptide bonds rotate freely?
Unit 3
Because they have partial double-bond character
Which residues lie on the same face of an a-helix?
Unit 3
a-helices have 3.6 residues per turn. Side chains separated every 3rd or 4th residues lie on the same face of the helix. The side chains form "ridges" that run nearly parallel or slightly inclined to the helix axis. The helices pack by fitting ridges formed by one set of side chains on one helix into grooves on the opposite helix.
What is responsible for the dipole of the a-helix?
Unit 3
• Because all hydrogen bonds point in the same direction, the a-helix has a net dipole with a partial positive charge at the N-terminus and a partial negative charge at the C-terminus.
What is the helix-turn-helix motif?
Unit 3
is a stretch of amino acids that form two a-helices separated by a short turn. The motif acts as a DNA-binding domain, where one helix fits into the DNA major groove while the other stabilizes the structure.
How does RNA differ from DNA?
Unit 4
RNA is single-stranded, contains a ribose sugar, and contains uracil (U) instead of thymine (T).
What is the name of the sugar molecule found in the backbone of RNA?
Unit 4
The "R" in RNA stands for ribonucleic acid, referring to its ribose sugar.
Which type of RNA is responsible for bringing specific amino acids to the ribosome during protein synthesis?
Unit 4
tRNA molecules have an "anticodon" on one end and a specific amino acid on the other, which they deliver to the ribosome.
What is the sequence of three nucleotides on an mRNA molecule that codes for a specific amino acid called?
Unit 4
The genetic code is read in groups of three bases called codons.
What is the shape of tRNA?
Unit 4
tRNA has a cloverleaf secondary structure and a functional three-dimensional inverted L-shape, which is essential for its role in translation.
Where is the amino acid attached on tRNA?
Unit 4
Amino acids attach to the 3' end of the tRNA molecule. This site is known as the acceptor stem and specifically ends with the sequence CCA.
What is the function of the anticodon loop?
Unit 4
Located opposite the ends, this loop contains a triplet of nucleotides (anticodon) that forms base pairs with the complementary mRNA codon during translation.
How does the 2D "cloverleaf" structure differ from the 3D structure?
Unit 4
In two dimensions, tRNA appears as a cloverleaf with four main arms: the acceptor stem, the D-arm, the TYC arm, and the anticodon arm. In three dimensions, the molecule folds into a compact L- shape, which allows it to fit precisely into the A, P, and E sites of a ribosome during translation.
Why is tRNA often described as having "unusual" bases?
Unit 4
tRNA contains many chemically modified nucleotides beyond the standard A, U, G, and C. These modifications, such as pseudouridine Y, help to stabilize the structure, aid in proper folding, and facilitate accurate codon-anticodon pairing, including "wobble" base pairing.
What is a wobble base pair?
Unit 4
A wobble base pair is a non-Watson–Crick base pairing between two nucleotides in RNA molecules that occurs at the third position of a codon and the first position of an anticodon. The most common wobble pair is G–U.
How is mRNA different from DNA?
Unit 4
Unlike DNA, which is double-stranded and stable, mRNA is single-stranded and breaks down quickly after its message is read. It also uses the base Uracil (U) instead of Thymine (T).
What are codons?
Unit 4
These are groups of three nucleotides on the mRNA strand that specify which amino acid should be added next during protein synthesis.
What is the primary function of rRNA?
Unit 4
rRNA forms the structural backbone of ribosomes and plays a catalytic role (ribozyme) in forming peptide bonds between amino acids during translation.
What is the composition of a ribosome?
Unit 4
Ribosomes are composed of ribosomal RNA (rRNA) and various proteins. They consist of two distinct subunits: a large subunit and a small subunit.
What are the key functions of rRNA?
Unit 4
rRNA serves as a structural framework for the ribosome and acts as a ribozyme to catalyze the formation of peptide bonds between amino acids.
What are the differences between canonical and non-canonical base pairs?
Unit 4
Canonical pairs follow the Watson-Crick model, whereas non-canonical pairs involve alternative hydrogen bonding schemes.
Why is RNA more fragile than DNA?
Unit 4
RNA is more fragile than DNA because of its chemical structure, which makes it highly susceptible to self-cleavage (hydrolysis), and its single-stranded nature, which leaves it exposed to degradation. The extra -OH group acts as a "chemical weapon," allowing the RNA to break its own phosphate backbone when exposed to heat or alkaline (basic) conditions.
What are the key features of RNA secondary structure?
Unit 4
Stems, or regions where base pairing occurs, usually in an A-form helix; hairpin loops, which connect the ends of a stem; internal loops/bulges, or unpaired nucleotides within a stem; junctions, where three or more stems meet; single-stranded, unpaired regions, that allow for flexibility/interaction.
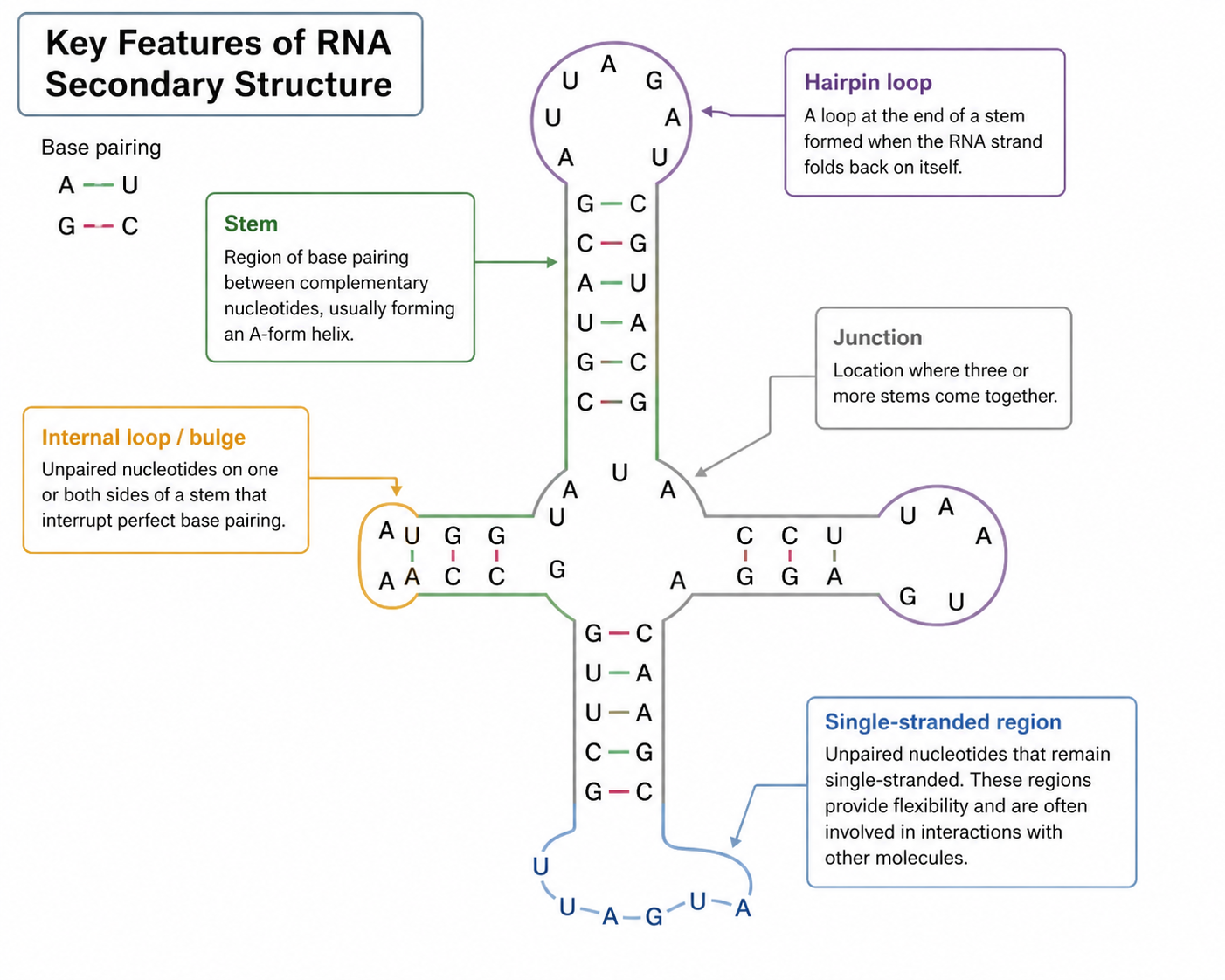
Why is RNA structure important?
Unit 4
RNAs rely on specific folding to function.
What is the difference between parallel and antiparallel b-sheets?
Unit 4
The strands in parallel b-sheets run on the same direction strands, with weaker H-bonds; The strands in antiparallel b-sheets run in opposite direction, with stronger linear H-bonds.
How are the side chains (R-groups) positioned in a standard b-pleated sheet?
Unit 4
They alternate projecting above and below the plane of the sheet. This arrangement minimizes steric hindrance between bulky side chains, making b-sheets flexible structures.
What is a Beta-turn?
Unit 4
A b-turn is a type of protein secondary structure that causes the polypeptide chain to reverse direction and fold back on itself. It is a compact, four-amino-acid segment that turns the backbone by ~180°, crucial for creating globular shapes of protein.
What is the basic structure of an amino acid?
Unit 5
Every amino acid consists of a central carbon atom bonded to a hydrogen atom, an amino group (NH2), a carboxyl group (COOH), and a variable side chain (R group) that determines its identity.
What is a zwitterion?
Unit 5
At physiological pH (~7.4), amino acids exist as dipolar ions called zwitterions. The amino group is protonated (NH3+) and the carboxyl group is deprotonated (COO–), resulting in a net neutral charge unless the R group is charged.
How are peptide bonds formed?
Unit 5
A peptide bond forms through a condensation reaction where the carboxyl group of one amino acid reacts with the amino group of another, releasing a molecule of water.
What is a key difference between parallel and antiparallel b-sheets?
Unit 5
Antiparallel sheets are more stable because their hydrogen bonds are linear.
Which type of bond is primarily responsible for stabilizing a-helix and b-pleated sheet structures?
Unit 5
Secondary structures like a-helices and b-sheets are stabilized by hydrogen bonds between the N-H group of one amino acid and the C=O group of another amino acid along the peptide backbone.