Exam II Content
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
What are the primary components of gas chromatography?
gas cylinder, column, injector, detector, and data system
What is the importance of the injector?
the port is at a higher temperature so the sample vaporizes instantly when injected
the whole system is kept at an elevated temp to keep everything in the gas phase
Advantages of the Open Tubular (Capillary) Columns over packed columns in GC
higher efficiency (more theoretical plates) → produce narrower peaks
better resolution → peaks are narrower so they can be separated more easily
faster analysis
greater sensitivity → narrow peaks give higher peak heights, making small amounts of analyte easier to detect
What are the 3 main types of carrier gases?
Helium, Hydrogen, and Nitrogen
What is the main advantage and disadvantage of Helium as a carrier gas?
A: inert, good efficiency
D: expensive
What is the main advantage and disadvantage of Hydrogen as a carrier gas?
A: fast separations
D: flammable
What is the main advantage and disadvantage of Nitrogen as a carrier gas?
A: cheap, good for electron capture
D: slow separations
What is the general elution problem?
when a mixture contains compounds with very different boiling points so one temp cannot give a good separation for both early and late eluents
What are ways to solve the General Elution problem?
Temperature programming → gradually increase temp
Use different columns/stationary phase
Gradient Program
changing the composition of MP during the separation
does not work for GC because since the carrier gas does not interact strongly with analytes, changing the composition would not significantly affect separation
What are the 2 main parameters for retention time?
boiling point (volatility) and polarity (interaction with the stationary phase)
What is the relationship between boiling point and retention time?
higher boiling point → higher retention time
Order these from least to most polar: Water, acetonitrile, methanol, THF, hexane
Hexane, THF, Methanol, Acetonitrile, Water
Normal phase: Order these from weakest to strongest eluent: Water, acetonitrile, methanol, THF, hexane
Hexane, THF, Acetonitrile, Methanol, Water
Common Stationary Phases and their characteristics
Diphenyl: nonpolar, intermediate van der waals forces, high temperature
Cyanopropylphenyl: intermediate, dipole-dipole + van der waals, lower temps
Carbowax: strongly polar, dipole-dipole, hydrogen bonding, van der waals
Biscyanopropyl: strongly polar, dipole-dipole + van der waals
What are the 3 most common GC detectors?
Thermal conductivity, flame ionization, and electron capture
Thermal Conductivity Detector
GC
Nondestructive
Linear
Nonspecific- least sensitive
Fluorescence Detector
LC
Nondestructive
Linear
Specific
UV-Vis Detector
LC
Nondestructive
Linear
Nonspecific
Electron Capture Detector
GC
Nondestructive
Nonlinear
Specific → extremely sensitive
Flame Ionization Detector
GC
Destructive
Linear
Specific → very sensitive
Split Injection vs. Splitless Injection
in a split injection only a small fraction enters the column
split injection has high concentration samples
in a split injection there is low risk for column overload
split injection → lower sensitivity
Kovats Retention Index
standardized value based on the retention times of n-alkanes that allows retention data to be compared across different GC systems and helps identify all compounds
Advantages of Open Tubular (Capillary columns) over packed columns in LC
higher efficiency → more theoretical plates, narrower peaks, and better separation
less Eddy diffusion → no packing particles so MP flows more uniformly
lower pressure requirements → less resistance to flow
better resolution
Disadvantages of Open Tubular (Capillary columns) over packed columns in LC
small sample capacity, low flow rates, and more difficult instrumentation
Isocratic Elution
mobile phase composition remains constant throughout entire separation
LSC vs LLC
LSC is solid absorbent and LLC is liquid film on support
LSC uses adsorption while LLC uses partitioning
LSC has surface interactions and LLC has solubility differences
Normal phase vs Reversed phase LC
in normal phase the stationary phase is polar but in reversed it is nonpolar
in normal phase the mobile phase is nonpolar and in reversed it is polar
in normal phase nonpolar compunds elute first and vice versa
in normal phase polar compounds elute last and vice versa
What are the most common LC Detectors?
UV-Vis, Refractive Index, and Fluorescence
UV Vis Detector
very good linear range
good sensitivity
moderately universal (only compounds that absorb UV)
Refractive Index Detector
moderate linear range
low sensitivity
most universal
Fluorescence Detector
moderate linear range’
highest sensitivity
least universal (only fluorescent compounds)
The Partition Coefficient (K)
how a compound distributes between 2 immiscible phases
large K: solute prefers the stationary phase → longer retention time
small K: solute prefers the mobile phase → shorter retention time
The Distribution Coefficient (D)
similar to K but includes all forms of the solute present
K and D are only the same when only one form of the compound exists
Organic Acids D vs pH plots
at low pH → mostly neutral HA; D is large
at high pH → mostly ionized A- ; D decreases
the partition coefficient K corresponds to the plateau region where compound is fully neutral
the pKa occurs at the midpoint of the curve where pH=pKa
Organic Bases D vs pH plots
at low pH → mostly protonated BH+; D is small
at high pH → mostly neutral B; D increases
Solvent Extraction vs Partition Chromatography
in solvent extraction there are one or few partition steps but in partition there are many repeated partition steps
solvent uses a separatory funnel but partition uses a chromatography column
solvent has coarse separation but partition uses high resolution separation
Theoretical Plates (N)
are a measure of column efficiency in chromatography
more theoretical plates → narrower peaks
Van Deemter Equation
H=A+B/u+Cu
A: Multiple paths (Eddy diffusion)
B: Longitudinal diffusion → faster flow rates don’t give solute band as much time to diffuse (broaden) → solute bandwidth is proportional to the square root of time → D is proportional to temp
C: Equilibration (resistance to mass transfer) → inversely proportional to temp
What is fronting?
occurs when the front (leading edge) of a peak becomes stressed out instead of being symmetric
most common cause is column overload
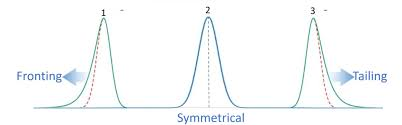
What is Tailing?
when the back of the peak becomes stretched out
caused by strong adsorption to the stationary phase (interact strongly)
Retention Time (tr)
total time it takes a compound to travel from injection point to the detector
Adjusted Retention Time (t’r)
retention time corrected for dead time
Migration Time
the amount of time it takes a solute to reach the detector in Capillary Electrophoresis
Reverse phase: Order these from weakest to strongest eluent: Water, acetonitrile, methanol, THF, hexane
Water, methanol, acetonitrile, THF, Hexane
Ion exchange Chromatography
separates compounds based on charge
common applications are protein purification, amino acid analysis, pharmaceuticals
Electroosmosis
the movement of liquid through a capillary when an electrical field is applied
only effective in small diameter capillaries because of large surface area to volume ratio, the charged layer affecting a larger portion of the solution and allows heat to dissipate quickly
Ways to increase the number of Theoretical Plates
increase column length
smaller stationary phase particles
improve column packing quality
optimize flow rate
LC Separation on C18 SP Column
SP is nonpolar
most polar compounds elute first
Electrophoretic Mobility
describes how fast an ion moves in an electric field due only to its charge and size
Apparent mobility
the actual mobility observed experimentally in the capillary
Capillary Electrophoresis has very high efficiency because…
no stationary phase (separation occurs in electric field)
all molecules move at same velocity
very small diameter → allows efficient heat dissipation and use of high electric fields
minimal eddy diffusion
What are the most common detectors in Capillary Electrophoresis
UV-Vis, Fluorescence, and Conductivity
Advantages of Capillaries for Electrophoresis
much higher efficency
faster separations
better heat dissipations
very small sample consumption
quantitative analysis
Relationship between resolution and theoretical plates
Rs scales to √N
Internal Standard Method
quantitative technique where a known amount of a compound (the internal standard) is added to all samples, calibration standards, and blanks
a constant, known amount of internal standard is added to every solution
chromatogram is run and peak areas are measured
calibration curve is made using known concentrations
ratio used to determine analyte concentration
To increase N in CE…
increase applied voltage
use a longer capillary
reduce diffusion
use smaller injection volumes
Problem with large relative retention
very long analysis times
peak broadening
poor peak shape and sensitivity