4.5- Carboxylic acids and their derivatives
1/97
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
98 Terms
What group do carboxylic acids contain?
COOH
What two functional groups is this comprised of?
Carbonyl (C=O)
Hydroxyl (OH)
What are the physical properties of carboxylic acids governed by?
Their ability to form hydrogen bonds
In a pure carboxylic acid, what can occur?
Hydrogen bonding between two molecules of acid to form a dimer, as the OH groups can hydrogen bond to one another
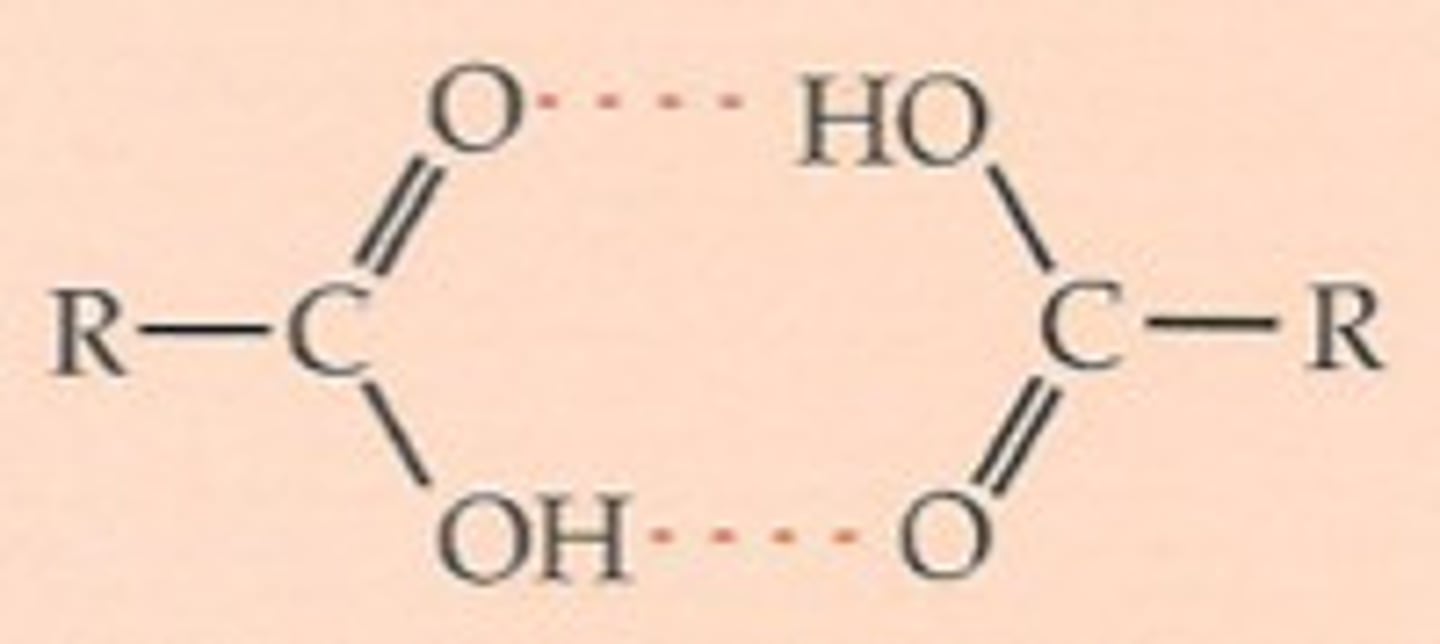
What does this result in?
Doubles the size of the molecule, so more Van der Waals forces between the dimers, so a higher boiling point
Why is ethanoic acid less volatile than butane?
Molecules of ethanoic acid can hydrogen bond to one another as they have OH group to form a dimer. The size of the dimer increases the VdW forces between dimers, so they have a higher boiling point.
As hydrogen bonds are stronger than VdW and butane only has VdW, carboxylic acid has higher boiling point.
What don't carboxylic acids do in the presence of water?
Form dimers
What form instead?
Hydrogen bonds and permanent dipole-dipole interactions between water molecules and individual molecules of acid
What happens to the solubility of larger acids?
Decreases with size
Why?
The longer hydrocarbon tails get between water molecules and break the hydrogen bonds. The hydrogen bonds are replaced with weaker VdW
Are carboxylic acids strong or weak acids and what does this mean?
weak so partially dissociate
How can they act as weak acids?
Due to the stability of the R-COO- anion
What does the ion have and what does this mean?
A delocalised system so it has a relatively high stability
Are they stronger or weaker acids than phenols?
Stronger
How can this be shown?
By their reaction with NaHCO3 to produce CO2- phenols are not strong enough acids to react
What do both carboxylic acids and phenols react with?
Strong alkalis
What is the order of acidity for water, carboxylic acids, alcohols and phenols?
Most- carboxylic acids, phenols, water/alcohols- least
What don't?
Alcohols and water
How can the acidity of carboxylic acids and phenols be altered?
By the presence of a substituent group- eg. substituting a H with a Cl, which increases its dissociation into ions
What are some typical reactions of carboxylic acids?
Acid + Metal -> Salt + Hydrogen
Acid + Base -> Salt + Water
Acid + Alkali -> Salt + Water
Acid + Carbonate -> Salt + Water + CO2
What type of reaction is acid + metal
Redox
2CH3COOH + Mg
(CH3COO-)2Mg2+ + H2
What type of reaction is acid + base
Neutralisation
Acid + metal hydroxide
ethanoic acid + NaOH
salt + water
sodium ethanoate + water
Acid + metal oxide
2ethanoic acid + CaO
salt + water
calcium ethanoate + water
What is a standard test for the carboxyl group?
Sodium hydrogencarbonate (fizzing)
Why?
Acid + carbonate
Acid + hydrogencarbonate
salt + water + CO2
2CH3COOH + Na2CO3
2CH3COO-Na+ + H2O + CO2
CH3COOH + NaHCO3
CH3COO-Na+ + H2O + CO2
How can carboxylic acids be formed?
Through the complete oxidation of primary alcohols heated under reflux with acidified potassium dichromate (orange to green)
What else could be used?
Acidified potassium manganate purple to colourless
Or?
Alkaline potassium manganate (purple to brown-black sludge of manganese oxide)
Describe the process of the reduction of carboxylic acids
As relatively stable, have to be reduced with LiAlH4 dissolved in ethoxyethane
Propanoic acid + 4[H] -> Propan-1-ol + water
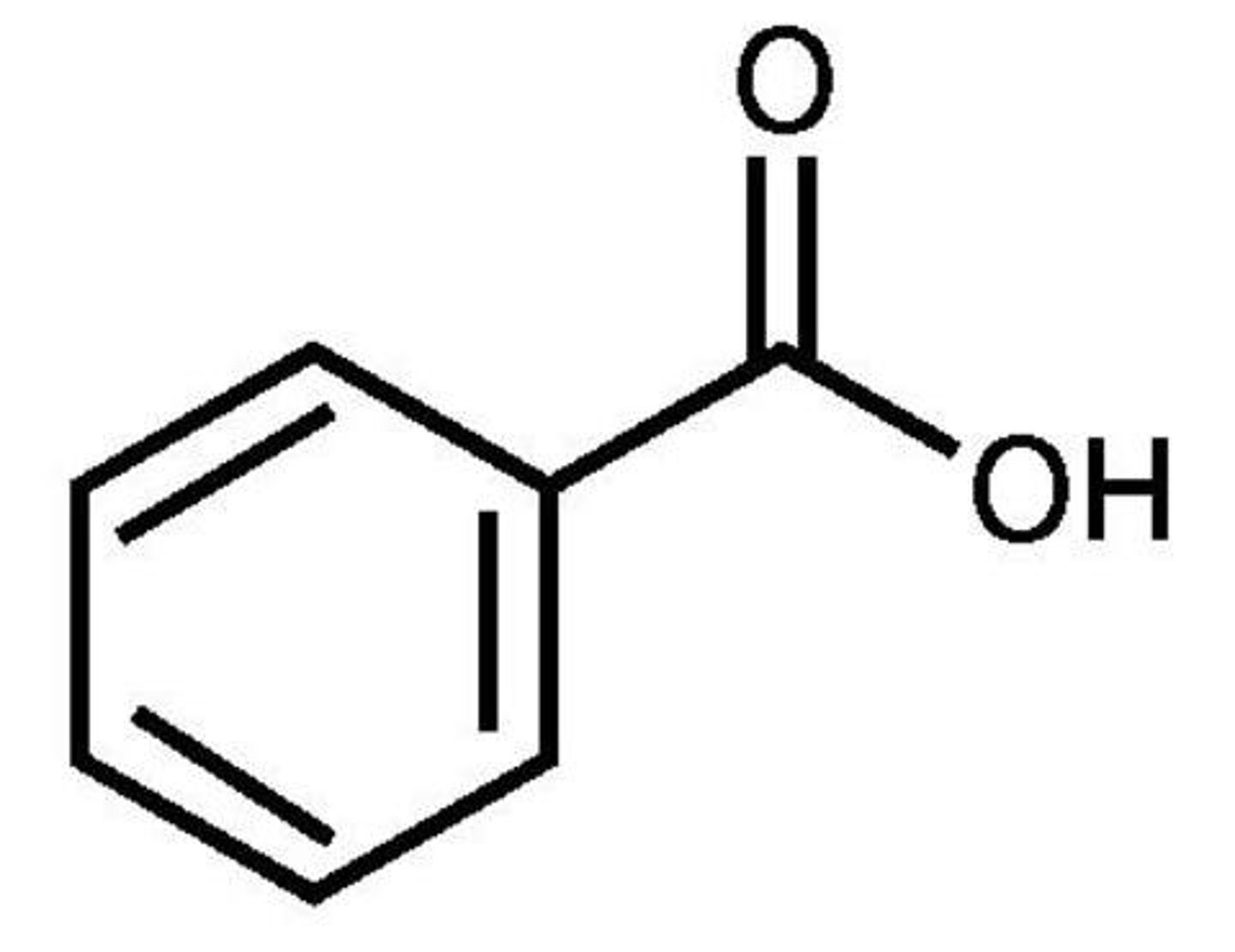
How can an aromatic carboxylic acid be made?
When a primary alcohol or aldehyde attached to a benzene ring is oxidised using acidified potassium dichromate/manganate
What is another method?
Oxidise a methyl chain on a benzene ring with alkaline potassium manganate to form benzenecarboxylic acid
Propan-1-ol + acidified potassium dichromate under reflux?
propanoic acid + water
orange to green
2 methyl propan-1-ol with alkaline potassium manganate
2 methyl propanoic acid + water
purple to brown sludge
phenylethanol with acidified potassium manganate
phenylcarboxylic acid + H2O
orange to green
What is decarboxylation?
The removal of carbon dioxide from a molecule
What is used for this process and why?
As CO2 is an acidic oxide, it will react with a base.
The base used is 'soda lime' (Ca(OH)2 with NaOH)
What will the resulting product contain?
One less carbon
What is the equation for the decarboxylation of benzenecarboxylic acid with Ca(OH)2?
benzenecarboxylic acid + Ca(OH)2 -> benzene + CaCO3 + H2O
Decarboxylation of 3-methyl 3-phenyl propanoic acid with CaO?
benzene with an ethyl group and a methyl group on that?? CaCO3
When can decarboxylation also occur?
When a calcium salt of an acid is heated in the absence of soda lime (CH3CH2COO-)2Ca2+
What will be formed?
pentan-3-one and CaCO3
What functional group do esters have?
RCOOR'
The one bonded to the C=O is from the...
acid
General name for ester: methyl methanoate.
If you reacted propanol with ethanoic acid, what would the name be?
Propyl ethanoate
methyl ethanol with methanoic acid
methylethyl methanoate
How are acid chlorides formed?
By reacting carboxylic acid with sulfur dichloride oxide (SOCl2)
What does them being extremely reactive mean?
That due to the instability of methanoyl chloride, ethanoyl chloride is the simplest acid chloride
How do you name acid chlorides?
Remove the 'oic acid' part and replace it with 'oyl chloride'
eg
propanoic acid -> propanoyl chloride
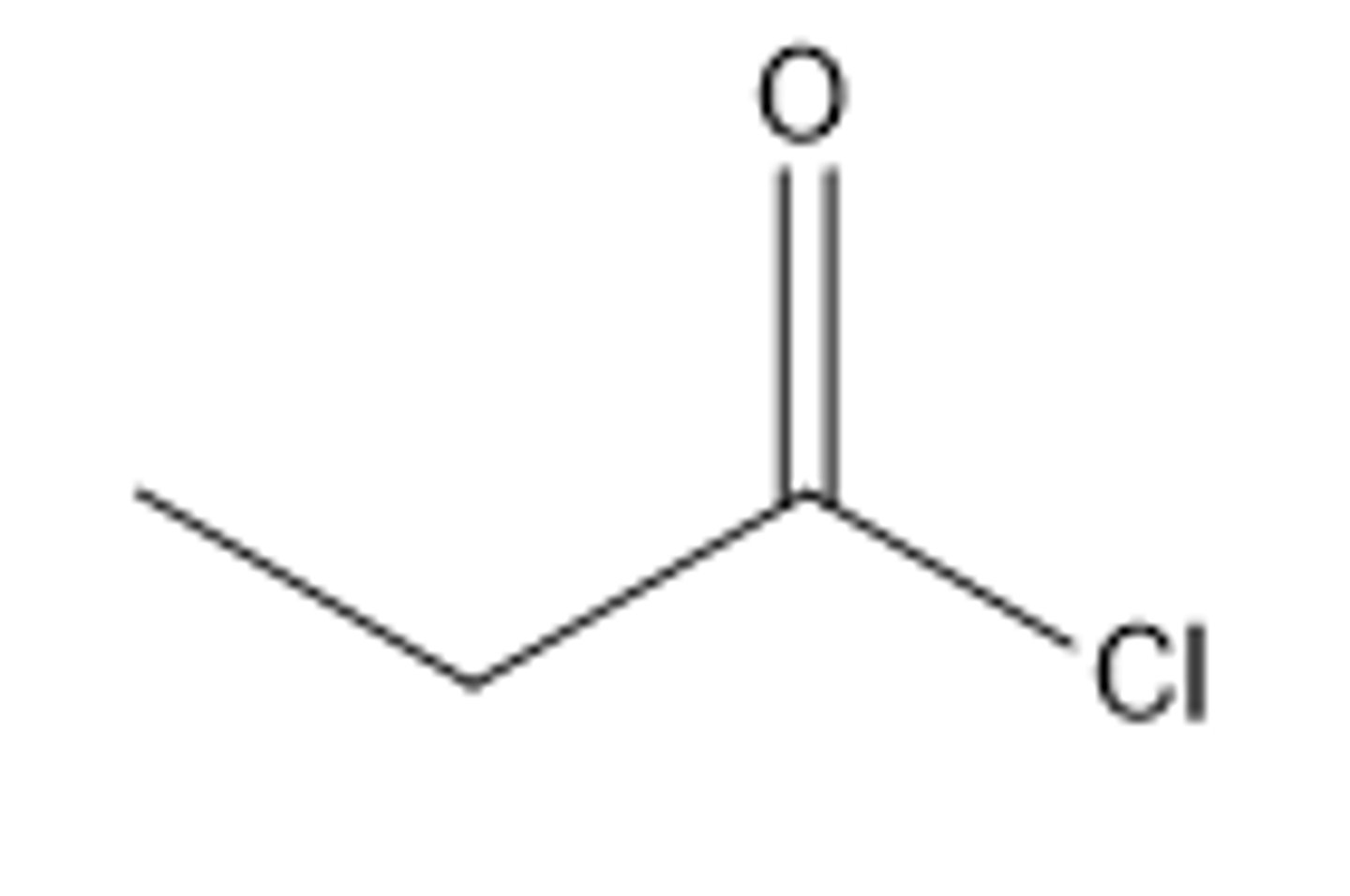
What is the skeletal formula of propanoyl chloride?
How are acid anhydrides formed?
removal of water from two carboxylic acid molecules
How do you name them?
Remove the 'acid' and replace with 'anhydride'
eg. ethanoic acid-> ethanoic anhydride
What are the 3 ways of making esters?
Carboxylic acids + alcohol -> ester + water
acid anhydride + alcohol -> ester + carboxylic acid
acid chloride + alcohol/phenol -> ester + hydrogen chloride
What conditions are carboxylic acids and alcohols reacted under?
Heated under reflux with concentrated H2SO4 as a catalyst
What is this reaction know as?
Esterification
What is it an example of?
Condensation
Is it reversible?
Yes
ethanoic acid + methanol ->
Methyl ethanoate + H2O
What are the conditions that acid anhydrides are reacted with alcohols and phenols under?
Gently heated
What are the advantages?
Non-reversible, high yield, occurs readily
Acid anhydride + alcohol ->
ester + carboxylic acid
methanol + ethanoic anhydride ->
methyl ethanoate + ethanoic acid
propanoic anhydride + ethanol ->
ethyl propanoate + propanoic acid
What are the conditions for acid chloride + alcohol/phenol and what is formed?
ester + hydrogen chloride
react readily without need for catalyst
ethanoyl chloride + butan-1-ol ->
butyl ethanoate + HCl
propanoyl chloride + phenol ->
phenyl propanoate + HCl (g)
What are the two types of hydrolysis of esters?
Acid and alkaline hydrolysis
What does acid hydrolysis entail?
Refluxing an ester with an acid
What is the equation?
Ester + H2O -> carboxylic acid + alcohol
Why is it not ideal?
Not got a good yield as reversible
Alkaline?
Ester + NaOH -> Carboxylate salt + alcohol
What is it also known as?
Saponification
What are the conditions?
Heated under reflux with aqueous NaOH
Why is it good?
Irreversible so high yield
What can be done after cooling?
Carboxylate ion can be acidified to form a carboxylic acid
What are the properties and uses of esters?
Sweet smelling
Used in food flavourings and perfume
Methyl propanoate + water ->
methanol + propanoic acid
propyl methanoate + water ->
propanol + methanoic acid
methyl propanoate + NaOH ->
sodium propanoate + methanol
propyl methanoate + NaOH->
sodium methanoate + propan-1-ol
How do you prepare acid chlorides?
React a carboxylic acid with SOCl2
Where must this reaction be carried out?
In a fume cupboard
Why?
As harmful gases SO2 and HCl are evolved
What is the reaction between propanoic acid and sulfur dichloride oxide?
Propanoyl chloride + sulfur dioxide + hydrogen chloride
What reagents can be used instead?
PCl3 and PCl5
What are the equations for these reactions with propanoic acid?
3CH3COOH + PCl3 -> 3CH3COCl + H3PO3
CH3COOH + PCl5 -> CH3COCl + POCl3 + HCl
What are the 3 reactions of acid chlorides?
1. Reaction of acid chlorides to form esters
2. Reaction of acid chlorides with water to form carboxylic acids
3. Reaction of acid chlorides with ammonia and amines to form amides
Are acid chlorides very reactive?
Yes
What do acid chlorides react with?
Nucleophiles, substituting the chlorine through the loss of a chloride ion whilst retaining the carbonyl bond
What can acid chlorides form esters with and what will they form?
Alcohols/phenols and will form
What does the reaction between ethanoyl chloride and water form?
Ethanoyl chloride + water -> ethanoic acid + hydrogen chloride
Why are acid chlorides not described as soluble in water?
Because they react so vigourously
Why can acid chlorides react with ammonia and amines?
Because they are both good nucleophiles as they have a lone pair of electrons which can readily be donated
Ammonia reacts with acid chlorides to form what?
A primary amide
Ethanoyl chloride + ammonia ->
Ethanamide + NH4Cl