Rates of Reaction and Energy Changes (1)
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
What is the equation for rate of reaction?
Rate of reaction = Amount of reactant used or product formed / Time (s)
What units could be used for rate of reaction?
g/s, cm3/s, mol/s
How can you measure the rate of reaction when a gas is given off?
Measure time and collect gas in an upside down measuring cylinder in a trough of water or in a gas syringe to measure the volume of gas produced.
Measure time and the change in mass.
How can you measure the rate of reaction when a precipitate is formed?
Put a black cross below a beaker containing one reactant.
Time how long it takes for the cross to disappear after the second reactant is added.
Why is using the precipitation method to investigate rate of a reaction not very accurate?
It’s subjective so people are likely to disagree over the exact point at which the cross is no longer visible.
How can you measure rate of reaction using a digital balance?
When a gas is produced as this will cause mass to decrease.
The experiment can be carried out on a digital balance and the rate of reaction can be calculated by recording the mass at regular time intervals.
What does the term ‘activation energy’ mean?
The minimum amount of energy required for a reaction to occur between two reacting particles.
What must happen for a reaction to occur?
Particles must collide at the correct orientation with sufficient energy to react.
In terms of particles, what 2 things could happen when the rate of reaction increases?
More frequent collisions.
Energy of collisions increases.
How can the rate of a reaction be increased?
Increase the temperature.
Increase the concentration.
Increase surface area to volume ratio.
Add a catalyst.
Increase the pressure (for gases).
How does temperature affect the rate of reaction?
Increasing temperature increases the rate of reaction.
This is because the reactants have more energy so more particles have energy above the activation energy meaning more collisions will be successful.
Collisions also occur more frequently because the particles have more kinetic energy.
How does surface area affect the rate of reaction?
Increasing the surface area of reactants increases the rate of reaction.
This is because a greater surface area means there are more exposed particles so more frequent successful collisions.
How does a catalyst affect the rate of reaction?
A catalyst increases the rate of reaction.
This is because it provides an alternate reaction pathway with a lower activation energy.
More particles will have sufficient energy to overcome the activation energy and react so more successful collisions occur in the same time.
How does concentration affect the rate of reaction?
Increasing concentration increases the rate of reaction.
This is because there are more reacting particles in the same volume so there are more frequent successful collisions.
How does pressure affect the rate of a gaseous reaction?
Increasing the pressure of a gaseous reaction increases the rate of reaction.
This is because there are more reacting particles in the same volume of gas (or the same number of particles in a smaller volume) so more frequent successful collisions occur.
Marble chips react with hydrochloric acid to produce calcium chloride, water and carbon dioxide. How could you measure the rate of reaction?
Since gaseous carbon dioxide is released, the rate can be measured by using a digital balance to measure the change in mass over a period of time.
Marble chips react with hydrochloric acid to produce calcium chloride, water and carbon dioxide. How could the rate of this reaction be increased?
Increase the surface area of the marble chips by turning them into a powder.
Increase concentration of acid.
Increase temperature.
After completing an experiment, how can you find the rate of reaction using a graph?
Plot: X axis: time, Y axis: amount of reactant used or product formed.
Draw a tangent to a point on the graph and find the gradient of this line to find the rate of reaction at that time.
A graph showing time and the amount of gas given off during an experiment has a steep gradient. What does this tell you about the rate of the reaction?
Fast rate of reaction.
A graph is plotted to show time and the amount of gas given off during a reaction. Describe the shape of the curve
Initially the curve is very steep as the rate of reaction is relatively fast at the start.
The curve becomes less steep as the reactants get used up because there are fewer successful collisions occurring.
At the end, the graph is a flat line because all the reactants have been turned into products.
What is a catalyst?
A substance which speeds up the rate of a reaction without being chemically changed at the end.
How does a catalyst speed up the rate of a reaction?
It provides an alternate reaction pathway with a lower activation energy.
What are enzymes?
Enzymes act as biological catalysts that increase the rate of reactions in living cells.
Which enzyme is used to produce ethanol from glucose?
Yeast
What do the terms exothermic and endothermic mean?
Endothermic - a reaction that takes in heat energy from the surroundings.
Exothermic - a reaction that gives out energy to the surroundings.
Are neutralisation reactions endothermic or exothermic?
Exothermic
Are displacement reactions endothermic or exothermic?
Either exothermic or endothermic.
Is a salt dissolving in water endothermic or exothermic?
Either exothermic or endothermic.
Are precipitation reactions endothermic or exothermic?
Exothermic
How could you measure the temperature change of a neutralisation reaction?
Measure initial temperature of the solutions.
Mix both reactants in a polystyrene cup.
Record the highest temperature reached.
Calculate the temperature change.
When measuring the temperature change of a reaction, heat loss means that measurements may be inaccurate. How could heat loss be minimised during an experiment?
Use polystyrene cup.
- Place reaction cup in a beaker full with cotton wool for extra insulation.
- Lid on the reaction cup.
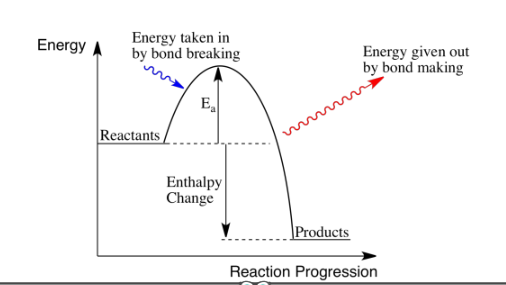
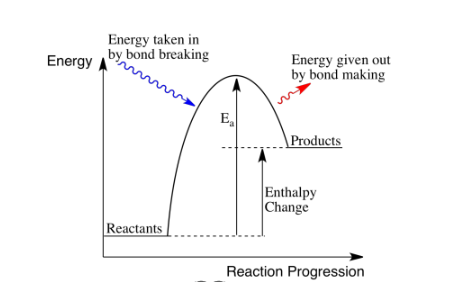
In terms of bond energies, what happens during a chemical reaction? Describe exothermic and endothermic reactions in terms of bonds breaking/forming.
Energy is needed to break bonds and is released when bonds are made.
Exothermic: Energy released from breaking bonds is greater than the energy used to make bonds.
Endothermic: Energy released in forming new bond is greater than the energy used to break old bonds.
What two types of reaction are exothermic?
Combustion, Neutralisation
Give an example of an endothermic reaction
Thermal decomposition, Photosynthesis
How can the energy change of a reaction be calculated from bond energies? (higher)
Energy change (kJ mol-1) = Total energy of bonds broken - total energy of bonds made.
If the energy change of a reaction is negative, is the reaction exothermic or endothermic? (higher)
Exothermic, energy has been lost to the surroundings.
What is a reaction profile?
A graph showing the relative energies of reactants and products, as well as the activation energy of a reaction.
Draw a reaction profile for an endothermic reaction
Draw a reaction profile for an exothermic reaction