Chemistry - Principles of Chemistry
1/149
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
150 Terms
Arrangements of solid particles
in neat rows and packed closely
movement of solid particles
vibrate only
energy of solid particles
less kinetic energy
arrangement of liquid particles
mostly touching but random arrangement
movement of liquid particles
nove and slide over each other
energy of liquid particles
more/greater kinetic energy
arrangement of gas particles
far apart and random
movement of gas particles
move randomly at high speeds
energy of gas particles
high kinetic energy
solid to liquid
melting
liquid to gas
evaporation
gas to solid
deposition
solid to gas
sublimation
gas to liquid
condensation
liquid to solid
freezing
diffusion
the net movement of particles from an area of high concentration to an area of low concentration until a mixture is formed; it takes place in gases and liquids where the particles are free to move.
Diffusion experiment 1
potassium permanganate crystal placed in a beaker will over time naturally dissolve and then diffuse to make a uniform purple solution
diffusion experiment 2
bromine is placed in a gas jar and a second gas jar is placed above - once released the bromine diffuses into the top gas jar and a mixture is formed
Gravity stops/affects diffusion. True or False
False
Diffusion experiment 3
concentrated ammonia (NH3) and hydrochloric acid (HCl) are soaking in cotton wool and placed at opposite ends of a long glass tube. NH3 and HCl gas molecules diffuse towards each other reacting to make a white ring of solid ammonium chloride (NH4Cl) [ NH3(g) + HCl(g) → NH4Cl(s) ] The white ring always forms nearer the HCl end because NH3 molecules are lighter tha HCl therefore diffuse faster and travel further than the HCl molecules.
Dilution experiment (diffusion experiment 4)
take a small amount of a coloured solution and add water several times, the solution gets paler and paler as there are less coloured particles in the same volume of liquid.
Why does diffusion take minutes when the gas particles are travelling very fast?
Diffusion takes time because the gas particles are colliding with each other, the walls of the container and air particles.
solute
the solid that dissolves
solvent
the liquids that the solid dissolve in
solution
the mixture formed when a solid dissolves in a liquid
saturated solution
a solution which contains as much dissolved solid as possible at a particular temperature.
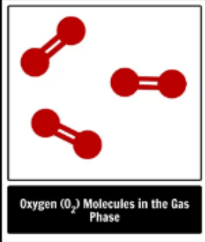
element, compound or mixture
element
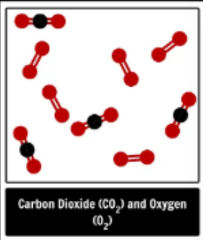
element, compound or mixture?
mixture
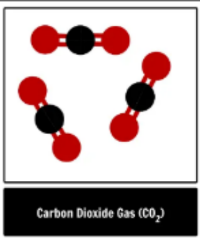
element, compound or mixture?
compound
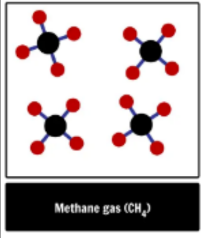
element, compound or mixture?
compound

element, compound or mixture?
mixture

element, compound or mixture?
Element
element, compound or mixture?
compound

element, compound or mixture?
element

element, compound or mixture?
mixture
elements
elements are made up of just one type of atom
compounds
compounds are made up of two or more types of atom chemically bonded together in fixed proportions
mixture
a mixture is a number of elements and/or compounds mixed but not chemically joined
example of an element
carbon (C), hydrogen (H2), iron (Fe)
example of a compound
carbon dioxide (CO2), water (H20), calcium chloride (CaCl2)
example of mixture
eg air, sea water, crude oil
pure substances
melt or boil at a single fixed temperature
impure substances
tend to melt or boil over a range of temperatures
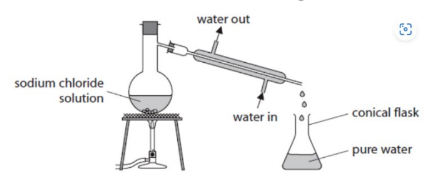
simple distillation
the separation of water and a soluble salt
simple distillation apparatus name
condenser
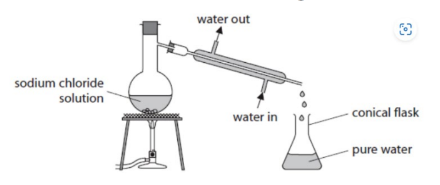
simple distillation apparatus process
condenser is heated, the water boils first and water vapour then travels down the cold condenser turning it back into liquid water for collection. The salt (NaCl) remains behind as it never reaches its boiling point. (water goes in the bottom and out of the top of the condenser so that it remains filled with water for better cooling.
fractional distillation
the separation of mixtures of liquids
examples of mixtures that could be fractionally distilled
ethanol, alchohol, water, crude oil
what helps with the separation of substances with similar boiling points?
fractionating column
how does the fractionating column help with the separation of substances with similar boiling points?
one substance is collected first at one temperature, the flask is switched as the temperature rises and the next substance is collected.
filtration
the method to separate an insoluble solid from a liquid
an example where filtration would be needed
removing sand from water
residue
the insoluble solid left in the filter paper eg sand
filtrate
the liquid collected e.g water
crystallisation
the method used o separate a solute (soluble solid) from a solution
an example where crystallisation would be used
sodium chloride crystals from a sodium chloride solution
crystallisation process (4 points)
solution is heated in an evaporating dish to remove some water
the solution is cooled and crystals form
he mixture is filtered to collect the crystals
crystals are dried on filter paper
chromatography
the method used to separate inks, dyes and food colourings
In chromatography the more ______ inks travel further up the paper, separating them out.
soluble
chromatography process (5 points)
a pencil line is drawn across the chromatography paper 1cm from the bottom
spots of the mixture of dyes are put on the pencil line and allowed to dry
the paper is lowered into a beaker with a little solvent (usually water), the paper must be touching the solvent but the spots must be above
when the solvent nearly reaches the top, the paper is taken out and where the solvent has got to, you mark the ‘solvent front’
the paper is left to dry
why must the inks not be in the solvent at the start
they will just dissolve in the solvent and not travel up the paper
why do we use a pencil line
the pencil is insoluble in most solvents (water) and will not move up
Rf value equation
Rf = distance moved by dye / distance moved by solvent

how to find Rf value (chromatography - 3 points)
measure the distance from the pencil line to the centre of the spot to nearest mm
measure the distance from pencil line to solvent front
use equation
atom
the tiny particles that make up all matter
molecules
a group of chemically bonded atoms (can be an element or a compound)
relative mass of a proton
1
relative mass of a neutron
1
relative mass of an electron
1/1836
relative charge of a proton
+1
relative charge of a neutron
0
relative charge of an electron
-1
atomic number
number of protons in an atom
mass number
number of protons and neutrons in an atom
what kind of atoms always have the same number of protons and electrons
neutral
isotopes
atoms of the same element that have the same number of protons but a different number of neutrons
relative atomic mass (Ar)
the average mass of the isotopes of the element relative to the mass of 1/12th of a 12C atom
relative atomic mass equation
((mass number x %) + (mass number x %)) / 100

are the groups vertical or horizontal columns?
vertical columns
True or False? The periods are the horizontal columns of the periodic table.
True
how many electrons can fit into the first shell?
2
True or False? the second, third and fourth shells all fit up to 8 electrons
False, only the second and third fit up to 8
how many electrons can the fourth shell fit?
18
are metals good or bad conductors of electricity?
good
are metal oxides acids, alkalis or bases?
bases
are non-metal oxides acidic, alkaline or bases?
acidic
what does the group number represent
the number of electrons in the outer shell of the atom
what does the period number represent?
the number of shells
why do elements in the same group all react in a similar way?
because they have the same number of outer shell electrons
what is the name for the group 0 elements
noble gases
what does inert mean? which group is inert?
inert means undergoing almost no chemical reactions. Group 0 (the noble gases) is inert
why are the noble gases (group 0) inert
because their atoms have full outer shells of electrons which is a very stable electronic configuration
why is argon used in light bulbs
to stop the metal filament reacting with oxygen that would be present in the air
what must be kept the same in balancing equations
there must be the same number and types of atoms on both sides of a chemical equation as atoms are not created or destroyed in chemical reactions
relative formula mass (Mr)
the mass of a compound or molecule compared to Carbon-12
a mole
a unit of the amount of a substance
what is the equation to find Moles
Moles = mass/mr
actual yield
the amount of product you actually get
theoretical yield
the amount of product that your calculations say you should get if the reaction was perfect
yield equation
% yield = (actual yield / theoretical yield) x 100
