CHAPTER 1&2- EXAM 1- Burns
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
What is the supreme law of the land? Does it describe the structure of the state or federal government?
U.S. Constitution—> federal government
What are the 3 branches of government and their role?
legislative- make laws
executive- enforce laws/provide leadership
judicial- interpret/explain laws
Match the branch of government to their title:
Congress (House and Senate) | |
President | |
Judges (Supreme Court and lower courts) |
Legislative | Congress (House and Senate) |
Executive | President |
Judicial | Judges (Supreme Court and lower courts) |
What is the main idea of each of the following Articles of the Constitution?
Article I
Article II
Article III
Article I- grants legislative branch, legislative power
Article II- grants executive branch (president) executive power
Article III- grants judicial branch (supreme court, etc.) judicial power
What does Article I, section 8 of the Constitution explain?
outlines the powers given to congress—> including the “interstate commerce clause” which allows congress to “regulate commerce w/ foreign nations, and among several states, and w/ indian tribes”
What does Article VI of the U.S. Constitution explain? What is it known as?
that the Constitution is the supreme law of the land—> known as “Supremacy Clause”
The Bill of Rights consists of the Constitution’s first 10 amendments… What does the Amendment X explain?
States have the power to legislate in all areas—> except those prohibited/given to congress by the Constitution
(think: X = gives states power)
What does Amendment XIV explain?
right to DUE PROCESS!!!!!! —> States cannot deprive any person of life, liberty, or property without due process of law
equal protection—> States must provide equal protection under the law to all people
What prevents branches of government from becoming too powerful?
check and balances/separation of powers
What are judicial opinions?
Are they enforceable as law?
What is this known as?
opinions are rules ENFORCEABLE as law
they are binding on lower courts within the SAME jurisdiction
common law (courts make law by issuing judicial opinions)
Explain Stare Decisis/Precedent:
decisions made in prior cases are followed in current cases and will be followed in future cases with the same facts
basically—> principle that courts should follow what happened before
Which of the following issues licenses?
a. state government
b. federal government
a
What is the hierarchy of laws?
Constitution
Federal statutes/laws
state constitutions
state legislation
ordinances
The _______ government has general police powers to protect public health, safety, and welfare.
a. state
b. federal
a
The 4th branch of the government are ___________________. What do they do? Examples?
The 4th branch of the government are administrative agencies
pass RULES AND REGULATIONS!!!! NOT LAWS!!!!!!!!!
but they have the “force of law”/ aka must be obeyed
examples:
fed: DOJ, DEA, FDA
state: state board of pharmacy
Where do federal agencies publish their regulations and other info chronologically where? Where do they publish final regulations?
(from book)
federal register
code of federal regulations
Answer the following about EMPLOYER policies:
how does it relate to state/federal law?
MAY be stricter than state/federal laws BUT should NOT be less strict than state/federal laws (“can be tighter, but not looser”)
Describe “civil” consequences for pharmacy providers:
(describe, example, results/goal?)
private party (plaintiff) sues another party alleging harm/injury of some type
goal/results: compensate the plaintiff/ have the defendant do/stop something
ex: malpractice pt. sues for harm, employee sues employer over employment matter, breach of contract, defamation
Describe “administrative” consequences for pharmacy providers:
(describe, example, results/goal?)
action taken by administrative agency to enforce its regulations
ex: state board of pharmacy and licenses, DEA action against a registrant
results: public reprimand, fine, license probation/suspension /revoked
Describe “criminal” consequences for pharmacy providers:
(describe, results/goal?)
involves criminal charges by the government against the pharmacy/pharmacist for violating a LAW w/ criminal penalties
results: if convicted= fine, probation, jail
The practice of pharmacy and distribution of drug products are subject to what kind of law? (from book)
a. state
b. federal
c. both
c
PRACTICE:
Federal authority to regulate drug distribution comes primarily from what?
(from book)
INTERSTATE COMMERCE CLAUSE (article 1, section 8)
PRACTICE:
State authority to regulate pharmacy practice and drug distribution comes primarily from what? (from book)
TENTH AMENDMENT and inherent authority of a state’s police powers
What is the FDCA? What does it state?
What agency overlooks/enforces it?
What is the goal of this law?
FDCA—> “Food Drug Cosmetic Act”
states- NO new drug may be market/sold unless proven safe and effective for its intended use AND approved by the FDA
FDA
goal—> protect consumers from ADULTERATED and MISBRANDED products
What is the definition of a “drug”? (focus on B and C)
basically—> any substance intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease, or intended to affect the structure or function of the body
full def in picture

What’s the definition of a “device”?
i don’t think important
basically—> instrument, apparatus, implement, machine, or contraption intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease, or to affect the structure or function of the body, without relying on chemical action. Essentially, a device functions primarily through physical means rather than chemical or biological means.
full def in the picture

What’s the definition of a “cosmetic”?
basically—> intended to be applied to the body for cleansing, beautifying, promoting attractiveness, or altering appearance
full def in the picture
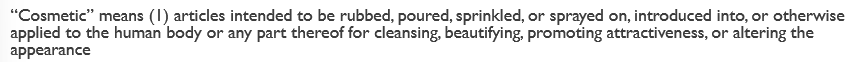
Can a product be a drug and cosmetic at the same time?
yes
What is the main determinant of whether a product is a drug?
whether the supplier makes a therapeutic/health claim or a structure/function claim!!!
Does the FDA regulate tobacco and related products?
yes
What’s the difference between food and a drug? What are the exceptions?
basically food is food, until it starts making health/disease prevention claims or any claim that would classify it as a drug
some exceptions—> 2 special categories:
“special dietary foods”
“medical foods”
Describe special dietary foods and medical foods?
idk how important
special dietary foods: foods made to meet dietary needs of people with specific conditions like pregnancy, hypersensitivities, overweigh/underweight
ex: infant formulas, artificial sweeteners, caloric sup.
medical foods: specially formulated and intended for the dietary management of a disease or condition that has distinctive nutritional requirements. These foods must be used under the supervision of a healthcare provider and are not meant for general consumption or as a substitute for a regular diet
ex: enteral nutrition formulas
remember: THESE ARE NOT DRUGS!!!!!
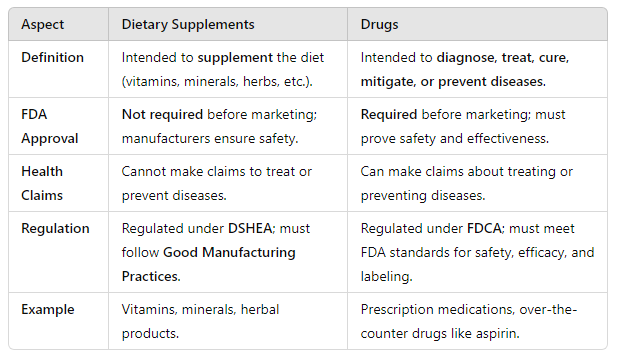
DSHEA amended the FDCA and created the category of dietary supplements. What are dietary supplements? How are they regulated compared to drugs like FDA approval? What’s allowed to be claimed?
dietary supplements: product intended for ingestion and to supplement the diet, contains 1+ vitamins, minerals, etc.
LESS regulated compared to drugs, regulated more like food—> does NOT require FDA approval
cannot make claims like drugs—>
HOWEVER allows 4 types of nutritional statements with a disclaimer on the label
DSHEA also allows limited health/disease claims that describe relationship between product/disease
Manufacturers can do voluntary recalls or FDAs can mandate them. What are the 3 classes of recalls?
Class I- MOST SERIOUS reasonable probability the product WILL cause serious ADRs
Class II- temporary/reversible health consequences
Class III- LEAST SERIOUS- not likely to cause ADRs
What’s a pharmacist’s responsibility in drug recalls? What could happen if a pharmacist dispenses a recalled product?
keep up to date
DO NOT dispense recalled products—> violates FDCA
could face civil actions if pt. is injured
The FDCA prohibits 2 major offenses called MISBRANDING and ADULTERATION!!!!!! WHAT IS the ADULTERATION?
think: “contamination”—> by bad ingredients, unsanitary conditions, the container, unsafe color additives, etc.
pay attention to how it says “may” contaminate
full definition in picture

Pharmacies, not just manufacturers can violate adulteration provisions. What are some examples?
pharmacy counts tablets on dirty trays
drug is repackaged in contaminated container
drug is stored at an incorrect temperature
Can drugs be adulterated and misbranded at the same time?
YES
Drugs must be manufactured in accordance with _______, if not = adulteration.
CGMPs (current good manufacturing practices)
What is a CGMP? (from book)
set of regulations establishing minimum standards for manufacturing methods, facilities, and controls
The FDCA prohibits 2 major offenses called MISBRANDING and ADULTERATION!!!!!! WHAT IS the MISBRANDING?
false or misleading labeling or HCEI is not accurate and reliable
includes ANYTHING that comes with the drug—> container, label, insert, etc.
Does misbranding apply to rx, otc, or both?
both
“Labeling where product can be used safely by a lay person for purposes intended” describes…
a. OTC
b. RX
c. BOTH
a
“Labeling directed towards practitioner for safe use and prescribing” describes…
a. OTC
b. RX
c. BOTH
b
What are some other examples of misbranding?
when dispensing rx drugs without a ___________
not providing ____________
not meeting __________
products not meeting _______ requirements
when dispensing rx drugs without a prescription
not providing required pt. information
not meeting PPPA
products not meeting REMS requirements
What happens in a pharmacy if I repackage or relabel OTC products?
must now comply with manufacturing labeling requirements (aka… just don’t do it, its a lot of work)
What is the most IMPORTANT thing found on a prescription drug label NOT found on OTC labels?
a legend or “rx only” label

What are some of the components of an OTC drug label specifically the “drugs facts panel” of the label?
active ingredient
uses
warnings
inactive ingredients
purpose
directions
other info (ex: storage)
questions/contact #
A label is defined as the label affixed to the container, but labeling is defined as…
the label PLUS accompanying information (ex: package insert)
There are many requirements to an rx drug label in addition to the legend. One of those is an expiration date. What does the expiration date assume?
the date assumes in original container
Other requirements for rx drug label:
idk how important

What type of labeling is used in many hospitals?
Unit Dose label
What are some of the components of a package insert? (from book)
idk how important
highlight section (BBW, recent changes, indication, dosage, all that stuff)
table of contents
full prescribing info
patient counseling
pregnancy warnings (reminder: not A-X categories anymore)
What is a national drug code? What do each segment mean? Are they found on OTC or rx drug labels?
unique 10 digit, 3 segment code; 11 used for billing
segments:
first—> identifies manufacturer
second—> identifies drug (strength/dose form)
third—> package size
found on OTC AND RX
If a drug has an NDC, does that mean it is an approved NDA?
NO NO NO NO —> just bc drug has NDC does NOT mean it has an approved NDA
What does FAERS stand for?
FDA Adverse Event Reporting System
What helps manage known or potential serious risks of a product/class? Is it FDA approved?
REMS programs—> approved by the FDA (manufacture makes it)
The Department of Health and Human Services (HHS) can do what in situations where the nation is facing serious threats by making available the use of certain measures like vaccines? ex: covid
declare EUA or Emergency Use Authorization
There are drugs on the market that have not been approved? Explain 1 scenario how this is possible.
pre-1938 drugs—> before the laws were passed/created
ex: synthyroid
What is the name of the FDA’s safety information and adverse event reporting program?
MEDWATCH
Is Medwatch for rx, otc, or both?
both
In terms of MedWatch, what must the pharmacy do?
must provide pts. with MedWatch number
What are the 3 classes of medical devices? Which class is least/most regulated?
Class I- LEAST REGULATED- pose least potential harm
needles, scissors, toothbrushes
Class II- general controls are insufficient to ensure safety and effectiveness, must meet additional standards before marketing
ex: insulin syringes, infusion pumps
Class III- MOST REGULATED- life-supporting or substaining devices, MUST have premarket approval
ex: pacemakers
Answer the following about Cosmetics:
does it require pre-market approval?
registered with the FDA, and compliance of what is voluntary?
where is misbranding found?
where is adulteration found?
does NOT require pre-market approval
registered with the FDA, and compliance of CGMP voluntary
misbranding—> if false/misleading label, lacks required info, not clearly understood, packaging doesn’t comply with PPPA, or if doesn’t have necessary safety warnings
adulteration—> same as with drugs—> “contamination”
What does the Durham-Humphrey 1951 Amendment add to the FDCA?
distinguished rx from OTC drugs
allowed verbal transmission of prescriptions and refills if authorized