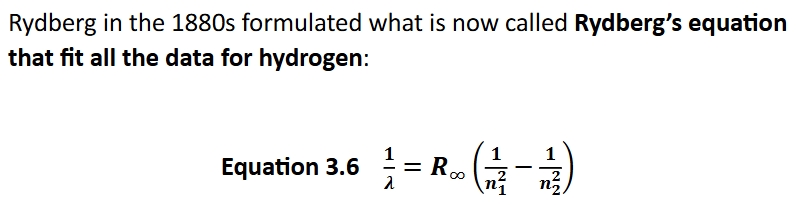CHEM1400 Chapters 1-3
1/130
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
131 Terms
What is chemistry
Often called the central science, the study of matter and the changes it undergoes with direct applications to many other fields of study
Biomolecules
DNA, RNA, proteins
Scientific laws
a concise verbal and or mathematical statement that describes a reliable relationship between phenomena
ex) ideal gas law
Scientific theory
explains a body of experiential observations and laws
ex) the kinetic theory of gases
SI base units
systems international; common modern form of the metric system
Weight
a force exerted by gravity on an object
Mass
a measure of the amount of matter regardless of the gravitational field
Atomic mass unit
AMU or Dalton Da
1.6605378×10^-24g=1amu=1Da
Accuracy
tells how close a series of measurement is to the true value
Percision
tells how close a series of measurements are to one another
Pure substances
form of matter that has a specific chemical composition and distinct properties, state, and solubility (water, iron, salt, oxygen)
mixtures of substances
ex) salt water and brass(combo of copper and zinc)
States of matter
solids, liquids, gases, plasma (more common)
Plasma
most common source of matter, ionized(charged) matter, doesn’t have well-defined boundary/shape but highly electrically conductive (the sun)
Homogenous mixture
uniform composition
Heterogeneous mixture
not uniform compesition
Physical state and properties of matter
melting point, color, observed
chemical properties of matter
observed when a chemical change of chemical processes happens to matter and pure substances/mixture no longer exists
extensive properties
ex) volume, mass, length
depends on amount of matter
These properties change as the quantity of matter changes, meaning they are additive; the total value is the sum of the values of the individual parts
Intensive properties
don’t depend on the amount of matter there
ex) temperature, density, color, solubility
It is a bulk property, meaning it is not dependent on the size or mass of a sample.
When dividing 2 extensive properties together
you get an intensive property
Atoms
they are the building blocks of matter, smallest units of matter that still retain the properties of that matter
Elements
term related to atoms; the substance can’t be broken down into 2 or more substances by any means
Carbon
C
14
Backbone of organic and biological molecules
Hydrogen
H
1
Most abundant element in universe; organic and biological chemistry
Oxygen
O
16
Basis of aerobic respiration, organic and biological chemistry
Nitrogen
N
15
Constituent of amino acids, proteins, nucleic acids
Sulfur
S
16
Important in metabolism including extremophiles
Sodium
Na
1
Important for nerve impulses, muscular function
Potassium
K
1
Similar to Na, important for nerve impulses and muscular function
Calcium
Ca
2
Bone formation; geology
Fluorine
F
17
One of the most reactive elements
Chlorine
Cl
17
Physiology, water balance in cells, digestion
Bromine
Br
17
animal life, collagen form
Iodine
I
17
metabolism, thyroid hormones
Silicone
Si
14
Semiconductor, important in modern electronics
Iron
Fe
8
steel, hemoglobin
Copper
Cu
11
cofactor for enzymes
helium
He
18
2nd most abundant; nuclear fusion
Thompson 1899
discovered the electron, first discovered subatomic particle
Millikin 1909
determined the charge of an electron by suspending charged oil drops in an electric field
Rutherford 1917
proposed the nucleus was a structure confined to a small region of space. Positive charged alpha particles → shot them at a thin foil like gold. Sometimes alpha particles went through sometimes not
Most atoms space is empty, there is a nucleus, nucleus is positively charged
Chadwick 1920
discovered neutrons
Atomic number
denoted by Z, number of protons
#of electrons=#of protons
Ion
When number of protons doesn’t equal the amount of electrons
Mass number
denoted by A, number of protons(Z) + neutrons (N)
Mass # A is superscript
Atomic # Z is subscript
Are the number of protons (Z) fixed for a given element?
Yes, the number of protons for an element are fixed
Isotopes
atoms with the same Z (Atomic #) but different A (Mass #)
Do the number of protons and electrons play a bigger role in chemical properties?
Yes the number of protons and electrons play a big role in chemical properties
Coulombic force
an electromagnetic force and same charged particles repel each other
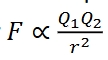
Strong nuclear force
extremely short-range force at lengths on the scale of the size of the diameter of the proton it’s about 100x stronger than the coulombic force
Weak nuclear force/interaction
force inside the nucleus, interconversion of protons and neutrons and important for nuclear fusion
When are isotopes reactive?
All isotopes arising from elements with Z greater than or equal to 82 are radioactive
Magic numbers
associated with stable nuclei, all even numbers
radioactive decay
involves emission of particles and or electromagnetic radiation from the nucleus
Alpha particles
the nucleus of helium without electrons
Beta particles
an electron that arises from the nucleus when neutrons convert to protons by the weak interaction. Often leaves the nucleus at relativistic speeds
Gamma rays
true electromagnetic radiation, release for many nuclear processes
Columns of periodic table
groups/families, share similar chemical and physical properties
Horizontal rows of the periodic table
the first period just has hydrogen, H and helium, He
Most elements are what?
Most elements are metals and good conductors of heat and electricity
Are nonmetals good conductors?
Non, nonmetals are not good conductors
Metalloids
between nonmetals and metals in terms of properties and include elements like silicone
Group 1
alkali metals
Group 2
Alkali earth metals- alkaline
Group 3 to 11, sometimes 12
Transition metals/ transition elements D block
Group 17
the halogens
Group 18
The Noble gases, don’t react much
Other groups on the periodic table are called what?
Other groups are simply called by the name of the uppermost element of the group
ex) 13 is called Boron group/family
2 rows underneath the main periodic table
F-block elements
1 mole particle=
6.0221413×10²³ of those particles (Avogadro’s number)
Molar mass
mass of grams of a substance in 1 mole
S block
Left side of the periodic table
D block
Middle of the periodic table
P block
Right side of the periodic table past the diagonal line
metals
nonmetals
metalloids
Group 1 and 2 throw off what easier?
They throw off electrons easier
Energy
The ability to do work/transfer of heat
Kinetic energy
Ek energy of motion, associated with the mass of the moving object(m) and its speed(u)
Can have the units joules, kilojoules, J/mol, KJ/mol, electronvolts(eV), calories(cal), or nutritional calories(Cal)
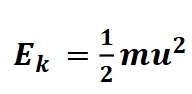
Potential energy
associated with the position of an object in a field
Electrostatic(potential) energy
Eel the potential energy and object (like an ion) experiences in an electric field. Charged particles of the opposite sign attract and the same sign repulse each other
Q is charge
d is the distance between the charged particle when Eel is positive is repulsive, Eel is negative is attractive
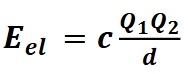
Law of conservation of energy
Energy is not created nor destroyed, but different forms of energy can interconvert
Joules, Kilojoules, KJ/mol, J/mol
don’t forget to convert g ←→kg
1000g=1kg
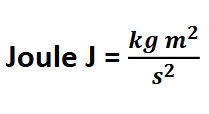
Energy (J) is directly related to?
Force(N-newtons) J=NxM work =forcexdistance
Electromagnetic spectrum
EM light is just a region of spectrum
The energy of EM radiation is transferred as waves(wavelength, frequency, and amplitude)
Visible light
400nm (violet) to 700nm(red) based on average human perception of light, ROYGBIV
Wavelength
Distance between identical points meters(m), nanometers (nm), picometers(pm)
10^-9m=1m
1pm=0.001nm
Frequency
number of waves that pass through a defined point per unit time (1sec)
1Hz=1/s
short wavelength-high frequency
long wavelength- low frequency
Amplitude
the vertical line from the midline to the top of the crest/bottom of the trough
The frequency and wavelength of EM waves is related by the following:
C= speed of light in a vacuum 2.9979×10^8m/s
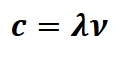
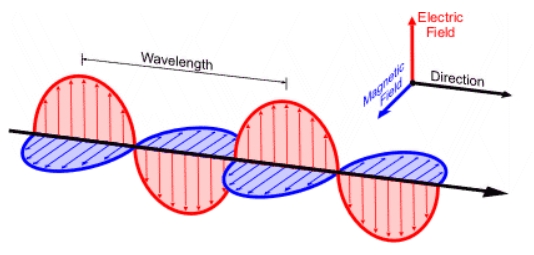
Electric field
EM waves have up/down waves
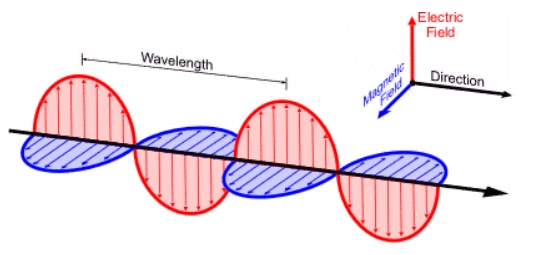
Magnetic field
EM waves have perpendicular out/in waves
Constructive interference
EM waves can have same 2 frequencies/wavelength/amplitude that leads to one larger wave
Destructive interference
EM waves can have 2 different frequencies/wavelengths/amplitudes that leads to one flat line
Quantum theory
how matter and energy behaves at the smallest scale vs bigger scale
Quantization of energy
Problem in science that was trying to explain blackbody radiation- EM radiation emitted from heated objects (sun, glowing swords)
Blackbody radiation
when an object is cooler, it gives off most of its energy as longer wavelengths (infrared). When an object is hotter, the max of the curve shifts to shorter wavelengths toward visible (light of even UV)
hotter=bluer
A hotter object doesn’t just change color it gives off more total energy
What is a blackbody?
An ideal object that absorbs all light that hits it and then re-emits energy as light when its hot. The light it gives off depends only on its temperature (T) intensity of radiation vs wavelength
Planck distribution law
He was able to model the blackbody radiation behavior incorporating a parameter h
h=6.626×10^-35 Js
Oscillations inside the heated material had discrete modes, not a continuum of values—and the EM radiation coming off the blackbody was emitted in discrete amounts. This is one of the earliest proposals of quantization of energy in a material
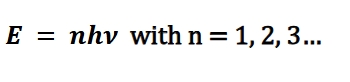
Photoelectric effect
metal exposed to light of under a certain threshold wavelength or frequency did not eject electrons form the surface (ex photoelectrons)
above that threshold, photoelectrons were ejected and increased in numbers in direct proportion to the intensity of light
photoelectron energy=Light energy in - binding energy
W is work
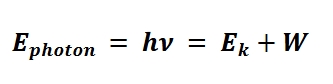
Bohr’s theory of the hydrogen atom
sun and other objects that emit white light that forms its emission spectrum, this white light is a combo of colored light. Pure elements like H show the atomic line spectra- the EM radiation emitted is specific wavelengths and is characteristic of that element
Rydberg constant
1.09737316×10^7 m^-1