CH-003: CH 8 + 9
1/8
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
What is Avogadro’s Number?
1 mol = 6.02 × 10²³ atoms/particles/etc
What does mole ratio convert?
From mole A to mole B
What does formula weight convert?
From grams A to mol A
What does Avogadro’s Number convert?
From mol A to atoms A
What does molar mass represent?
From grams A to one mol A (g/mol)
How do you find the mass percent of a given element within a compound?
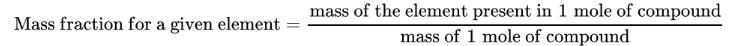
What does the empirical formula or simplest formula represent?
A formula that expresses the smallest whole number ratio of atoms present in a compound
What does the molecular formula represent?
The composition of # molecules present in a compound
What is the equation for percent yield?
Percent yield = (actual yield (g) / theoretical yield (g) ) * 100