Final Cell Bio
1/103
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
104 Terms
Two microscopes with the same lenses are using different light sources. Microscope A is using visible light. Microscope B is using ultraviolet light that has a wavelength of 320 nm. Which of the following is true?
The resolution of the microscope B is better than microscope A
Which of the following polymers are created by dehydration or condensation reaction?
All of the above
Which of the following chemical bonds is due to the random, ever changing accumulation of electrons in one part of the molecules?
Van der Waals Interaction
Name 4 things that all living things do?
Maintain homeostasis, Reproduce, Respond to stimuli, and Grow/develop
What living things are the universal building blocks of living things?
Cells
Seeing things clearly using the microscope depends on all the following expect______
Microscope working distance
The cell theory was proposed by ____
Schleiden and Schwann
The majority of amino acids have side chains that are ____.
Nonpolar
An electron microscope would be needed to view which of the following?
Ribosomes
A cell with the endoplasmic reticulum most likely belongs to the domain___.
Eukaryotes
The simplest amino acid is_____, and its R group is ____.
Glycine….. a hydrogen atom
There are ____ common amino acids.
20
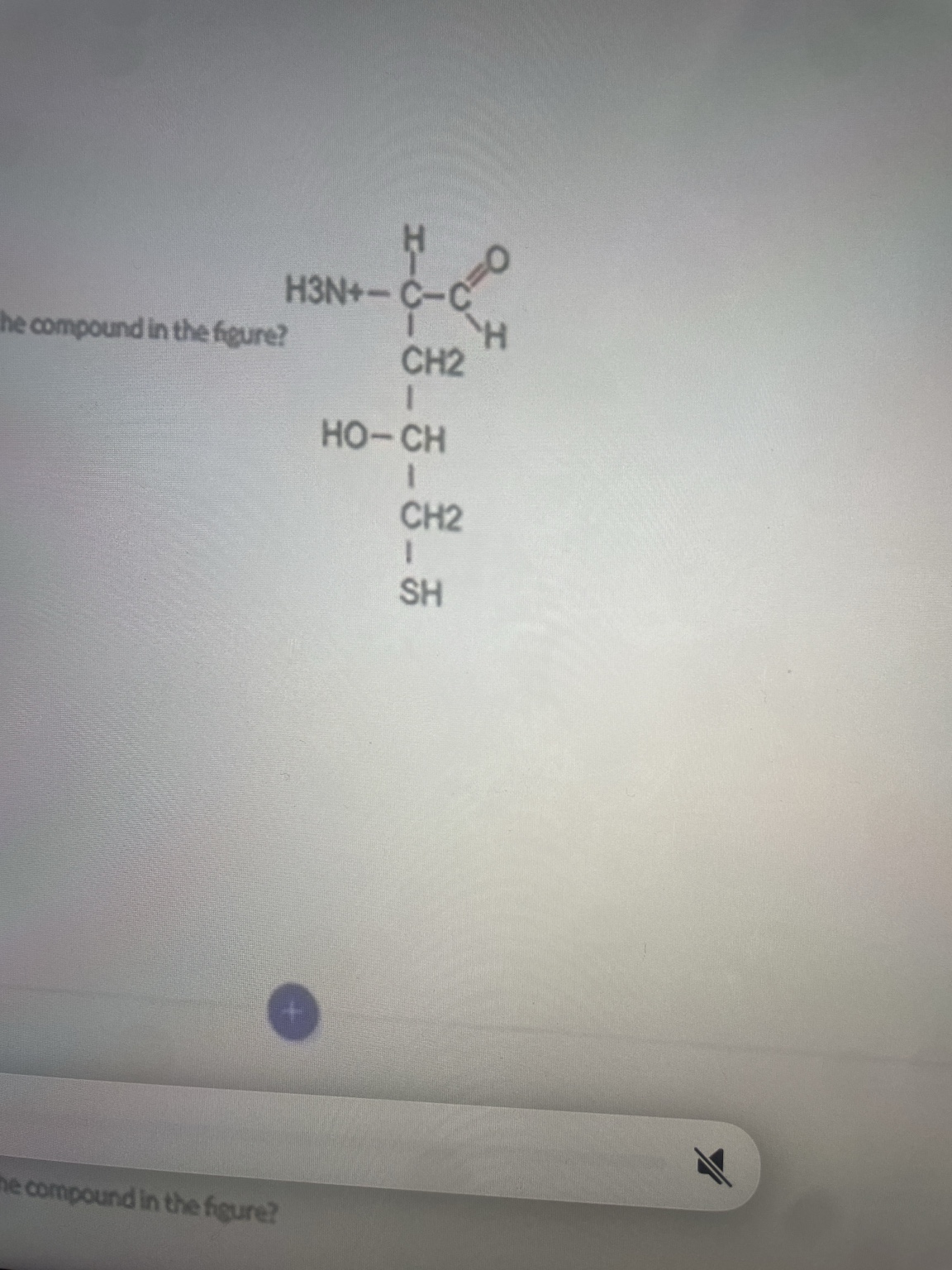
Which of the following chemical groups is NOT present in the compound in the figure?
Carboxylic acid
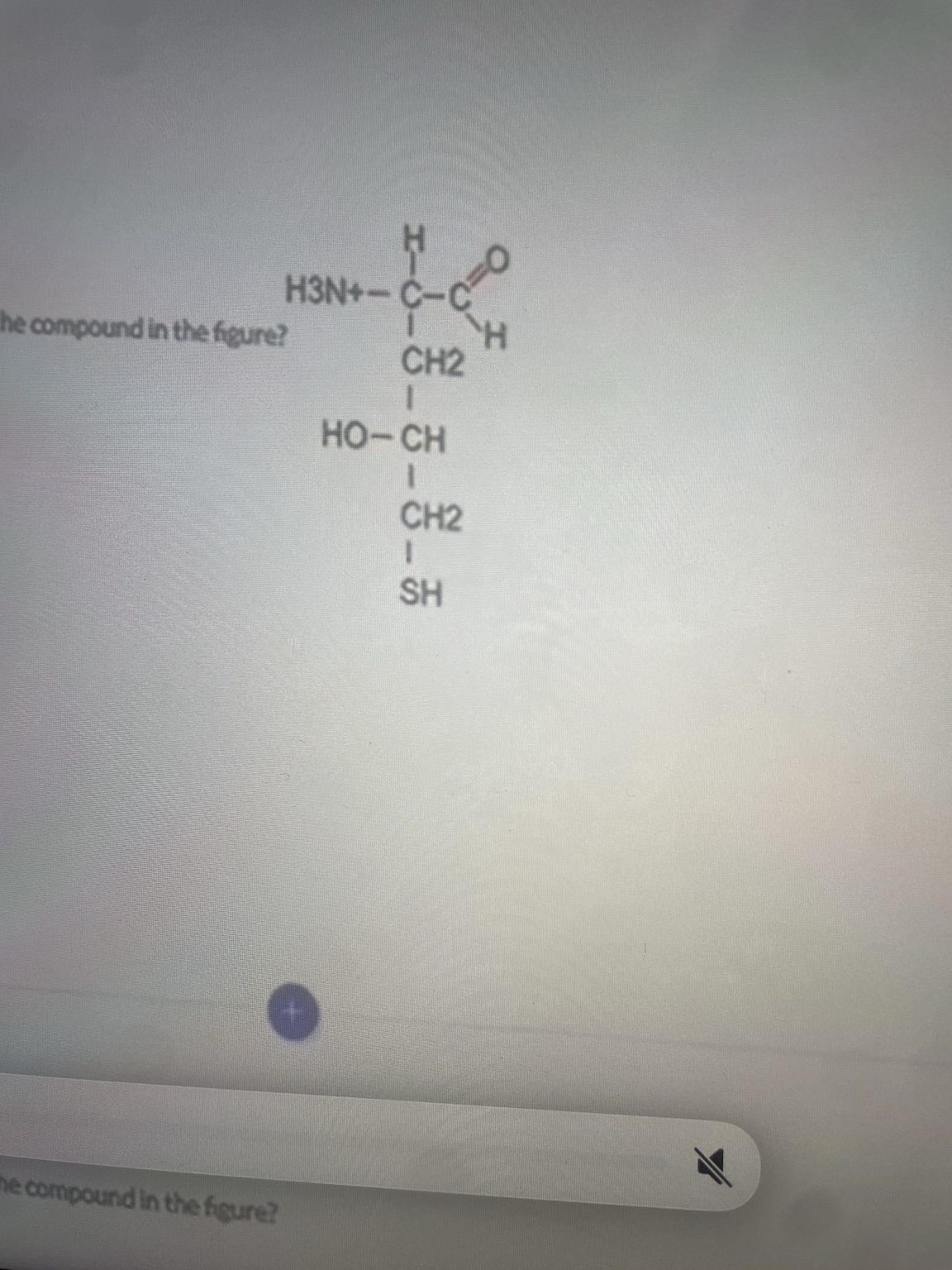
Which of the following chemical groups is NOT present in the compound in the figure?
Carboxylic acid
At least some of the illumination source passes through specimens in all of the following types of microscope, expect?
Scanning electron
The lenses in an electron microscope are actually____
Magnets
The earliest light microscope were noted in the____.
1600s

Which of the following atoms is most likely to form an anion in a dissociated ionic bond?
Fluorine (F)
In a polar covalent bond consisting of hydrogen and oxygen, the _____.
Oxygen has a partial negative charge because it is more electronegative than hydrogen
Which of the following elements make up 96% of all living matter?
Carbon,Hydrogen, Nitrogen and Oxygen
How many double bonds are in docosahexaenoic acid (DHA)?
6
Which of the following chemical bonds is strongest in cells?
Covalent
Which of the following are true of ALL protein?
They have ligands
How many valence electrons are in carbon___?
4
Which of the molecules below demonstrates the ability of the chains of carbons to form ring structures?
All of the above
The percentage of water in cells ranges from___ percent.
70% to 90%
Name of the four macromolecules of life.
Carbohydrates, lipids, proteins and nucleic acids
Sucrose is a disaccharide made of ____.
Fructose and glucose
Which of the following polysaccharides is made by humans?
Glycogen
A monosaccharide with 4 carbons will have ____ oxygens and ____ hydrogens.
4 and 8
Which fatty acid type contain NO double bonds?
Saturated
Which of the following is/are present in all cells?
All of the above
Name of the purine nucleotides.
Adenine and Guanine
A derivation of the nucleotide adenine forms the energy molecules____.
ATP and ADP
Name four types of proteins
Enzymatic, Structural, Transport and Hormonal
Which of the following is a part of all amino acids?
Amino group
Name two of the basic amino acids
Arginine and Lysine
Name two of the uncharged,polar amino acids
Serine and Theronine
If the pH of cells the normal range of approximately 7.2, the ____group in amino acids can donate a hydrogen to lower the pH.
Carboxyl group
What is the carbohydrate in DNA, and how many carbons are in it ?
5 carbons
The amino acid sequence of a polypeptides is its_____ structure
Primary
Beta-sheets in which the amino(N)-termini of the beta-strands are aligned are described as___.
Parallel
What would the resolution of a microscope using a light source with a wavelength of 400nm and a numerical aperture of 1 be?
200nm
How many nanometers are in 1.78 centimeters?
1.78×10^7
Who showed in the laboratory that spontaneous formation of organic compounds that could form the macromolecules of life could occur in conditions found on primitive Earth?
Stanley Miller
When was the cell theory proposed?
1839
Which of the following is correct about helices?
They are stabilized by hydrogen bonding
Which amino acid is incompatible with the formation of alpha helices?
Proline (Pro)
Which of the following types of bonds act in protein folding?
All of the above(Electrostatic attractions,Hydrogen bonds,Hydrophobic forces, and Van der Waals interactions)
Which type of microscopy requires coating the specimen with heavy metal?
Scanning electron
Scientist estimate the prokaryotes were first found on earth approximately ___billion years ago.
3.5
Name the three domain of life
Bacteria, Archaea and Eukaryotes
Define a protein domain
specific region of a protein that can fold independently into its own stable 3D structure and has a particular function.
Photosynthesis occurs in which of the listed organelles?
Chloroplasts
DNA can be found in which of the limited organelles?
Chloroplasts, Mitochondria and Nucleus
Which of the listed organelles has/have cisternae?
Golgi apparatus, Rough endoplasmic reticulum
Which of the listed organelles is/are where proteins are assembled, modified, and sent to other destinations?
Golgi apparatus
Which of the listed organelles make proteins that are secreted from the cell?
Rough endoplasmic reticulum
Which of the listed organelles has two phospholipid bilayers?
Chloroplast, Mitochondria and Nucleus
What happens to the volume to surface area ratio in eukaryotes compared to prokaryotes?
Increases
Cristae are formed by the ____.
Inner mitochondrial membrane
Regions of DNA where transcription is occuring would most likely be found in____.
Active euchromatin
Chromatin remodeling complexes____.
Use energy from ATP to change the position of nucleosomes
Histone H1____.
Shortens the linker regions between nucleosomes
Part of the mitotic spindle attaches at the ____of chromosomes.
Centromeres
A/an___ is a segment of DNA that directs the production of a particular polypeptides or functional RNA molecule.
Gene
How many hydrogen bonds are between guanine and its correct base-pairing partner in DNA?
3
Which of the following statements about the plasma membrane of the cells is correct?
None of the above
Who proposed the fluid-mosaic model of the plasma membrane?
Singer and Nicolson
____transfers phospholipids randomly from the cytosolic to the lumen side of its phospholipids bilayer and is found in the ___.
Scramblase… smooth ER
Which of the following chemical interactions is most important in formation of the phospholipid bilayer of around cells?
Hydrophobic forces
At normal body temperatures, cholesterol makes cells membranes____.
Less fluid
Name the three parts of a DNA nucleotides.
5-carbon sugar , phosphate group and nitrogenous base pairs
Which of the following movements is done by phospholipids in cellular membranes?
All of the above
Proteins make up___ of the mass of most plasma membrane
50%
Phospholipids most frequently have fatty acid tails with ___ carbon atoms.
18 to 20
Which parts of nucleotides are directly involved in the phosphodiester bond of the backbone of the nucleic acids? Be very specific: the ___bonds with the ____.
the 3′ hydroxyl group bonds with the 5′ phosphate group.

If only osmosis can occur in the pictured situation, what will move across the membrane and which way will it go?
The blue water will move to the left
Organisms in which of the following domains typically have linear DNA?
Eukaryotes
In nucleosomes, how many nucleotides pairs wrap around the histone core?
147
Which of the following is accurate according to the base-pairing rules?
None of above (C pairs with A, G pairs with A, C pairs with T, and G pairs with T)