Hydroboration-Oxidation Orgo Lab Final
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
electrophilic aromatic substitution
a reaction where an electrophile replaces a hydrogen atom on an aromatic ring, preserving the aromaticity
nitration of methyl benzoate
electrophilic aromatic substitution where a nitro group (NO2) is introduced onto the aromatic ring of methyl benzoate
activating group
a substituent that donates electron density to the aromatic ring, increasing its reactivity toward electrophilic substitution
deactivating group
a substituent that withdraws electron density form the aromatic ring, decreasing its reactivity toward electrophile substitution
resonance stabilization
the delocalization of electrons across a molecule that stabilizes intermediates formed during a reaction
rate-determining step
the slowest step of a reaction that determines the overall reaction rate; in EAS, it's the attack of the electrophile on the ring
TLC (Thin Layer Chromatography)
a technique used to identify and verify the product by comparing the movement of spots on a TLC plate
aromaticity
electrophilic aromatic substitution preserves the ______ of the benzene ring
nitronium (NO2)
in the nitration of methyl benzoate, the electrophile in the ____ ion
activating
electron-donating groups are called ______ groups because they enhance the rate of substitution
deactivating
electron-withdrawing groups are called ____ groups because they decrease the rate of substitution
meta
the _____ position is where the nitro group attached in the major product of methyl benzoate nitration
the ______ bath is used during the reaction to control temperature and prevent side reactions
attack
the rate determining step in electrophilic aromatic substitution is the ____ of the electrophile by the aromatic ring
delocalized
in resonance structures of benzene, electrons are _____ across the ring
catalyst
sulfuric acid acts as a ______ in the nitration reaction, helping to generate the nitronium ion
ortho
- describes the substituents on a benzene ring that are attached to adjacent carbon atoms (positions 1 and 2)
meta
- describes the substituents on a benzene ring that are separated by one carbon atom (positions 1 and 3)
para
- describes the substituents on a benzene ring that are opposite each other (positions 1 and 4)
ortho/para directors
- substituents that are usually activating groups (strong or moderate) or weak deactivating groups
- they donate electron density and favor electrophilic attack at positions 1 and 2 or 1 and 4
meta directors
- substituents that are strong deactivating groups
- they withdraw electron density and favor electrophilic attack at positions 1 and 3
para, ortho
bulky substituents will attach at the ____ positions rather than the ___ positions due to steric hindrance
the OH substituent is a _______ that will attach at the ortho or para positions
activating
generally substituents possessing a lone pair of electrons that can be donated are _______ groups
deactivating
generally electron withdrawing substituents are ____ groups
meta
carbonyl (C=O) groups next to the benzene ring are moderately deactivating, thus direct other substituents to the ____ position
meta
-NO2, -NR3+, and -CN are all strongly deactivating and are therefore ___ directors
nitration of methyl benzoate
mixed melting point, melting point, and TLC were used to determine the identity of the product in the _______ (EAS) lab
formation of nitronium ion
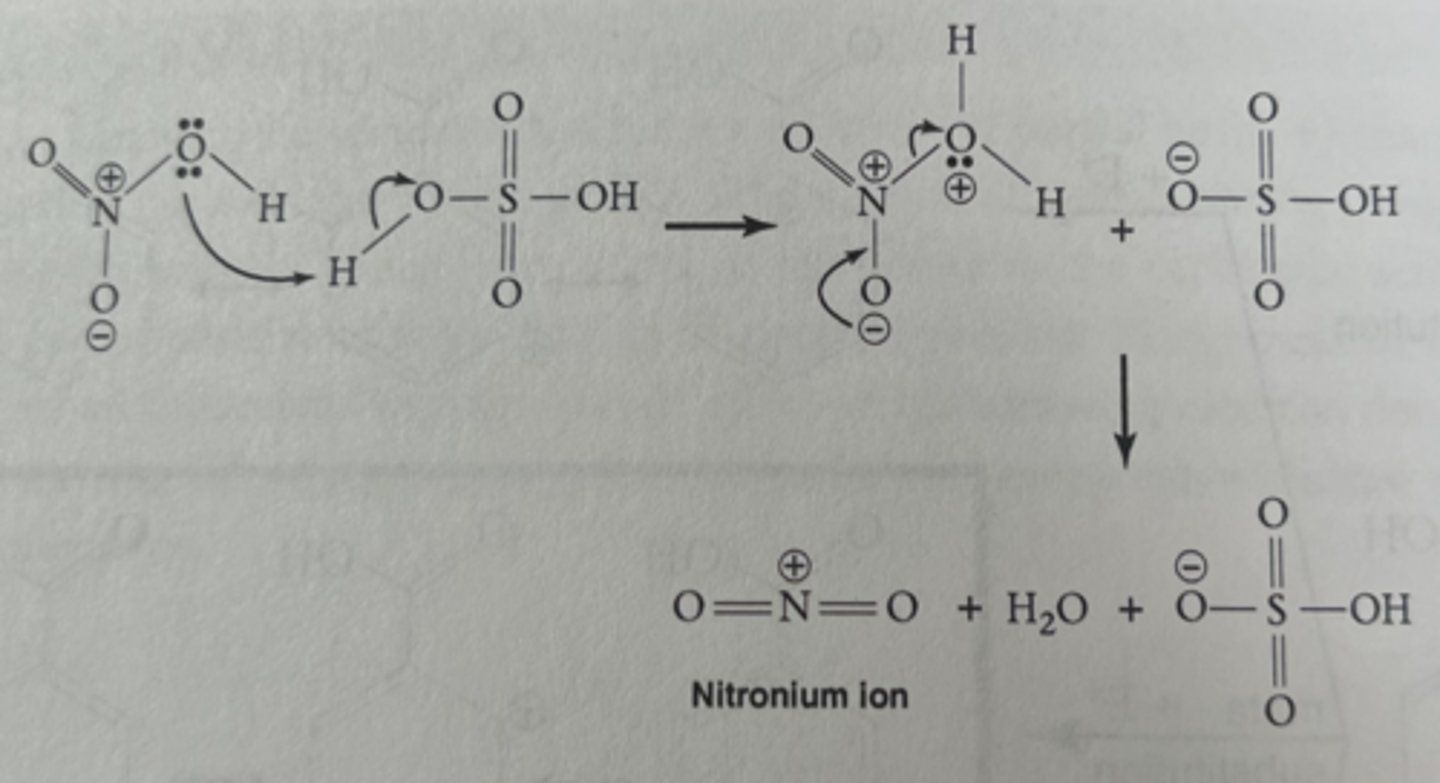
step 1 of nitration
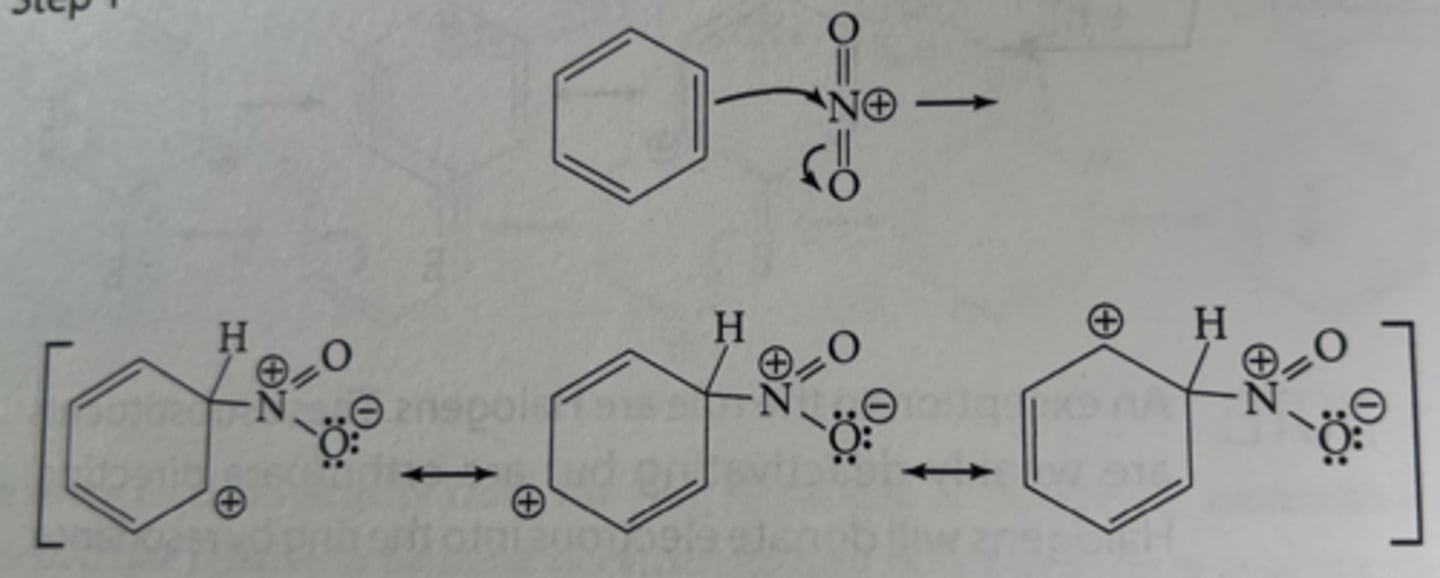
step 2 of nitration
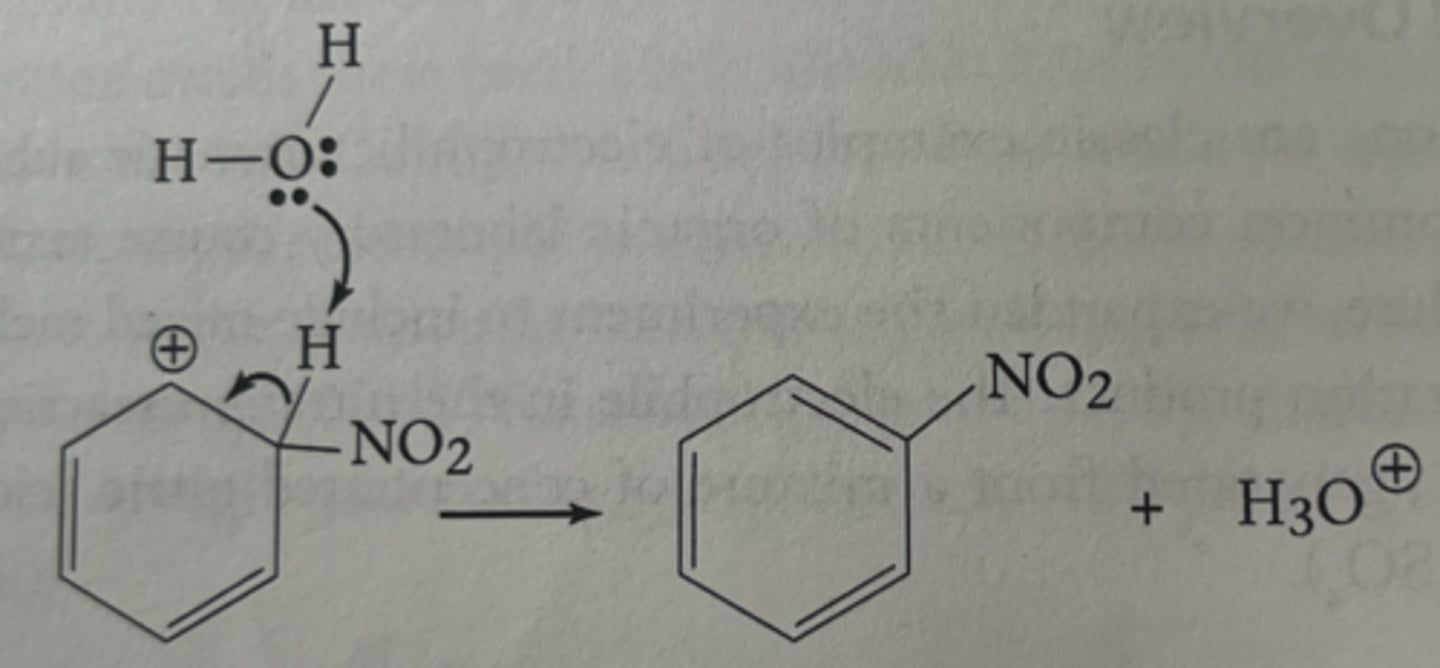
nitronium ion
nitric acid was combined with sulfuric acid to make the ______ that became the electrophile for the nitration of methyl benzoate
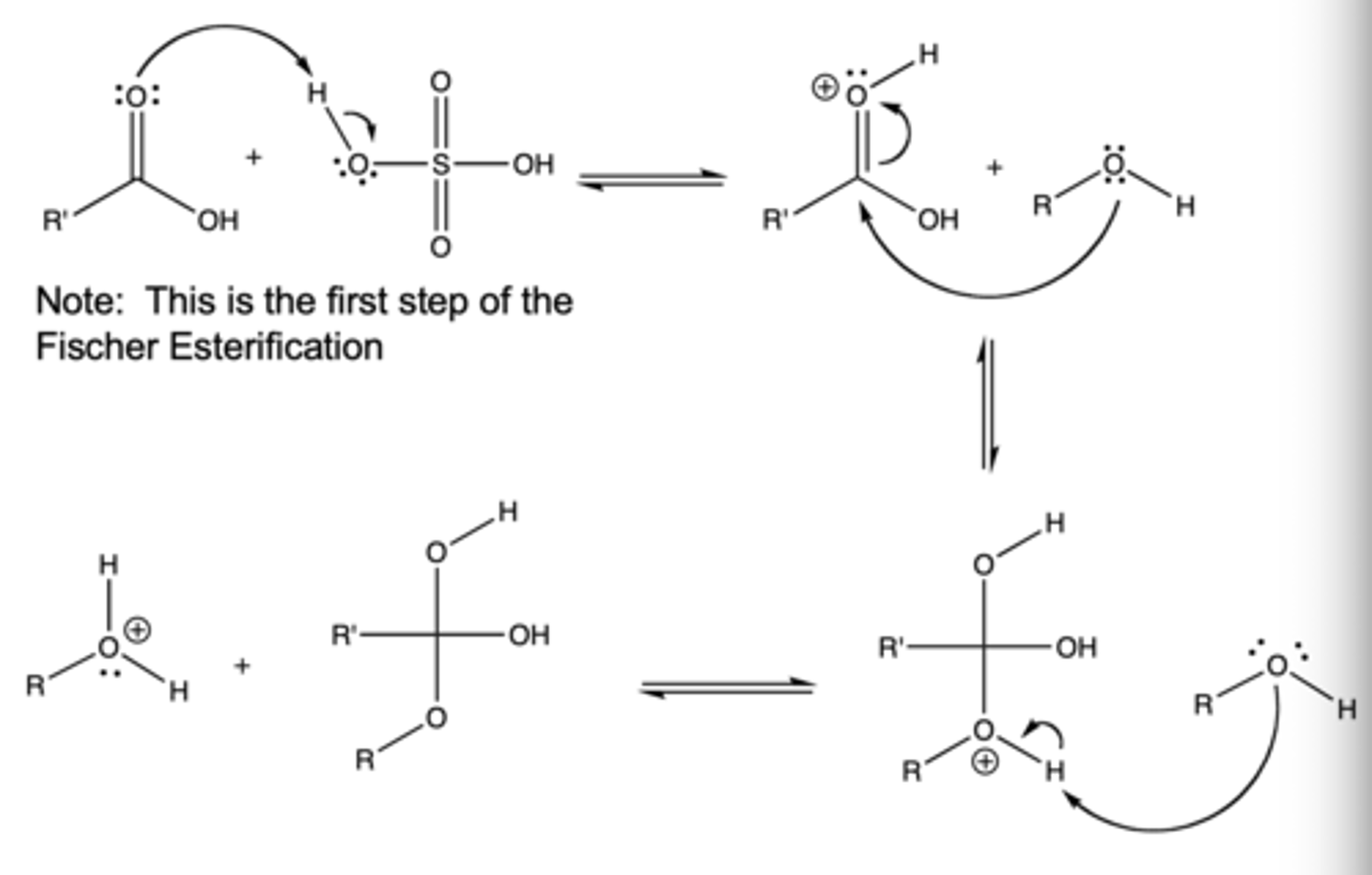
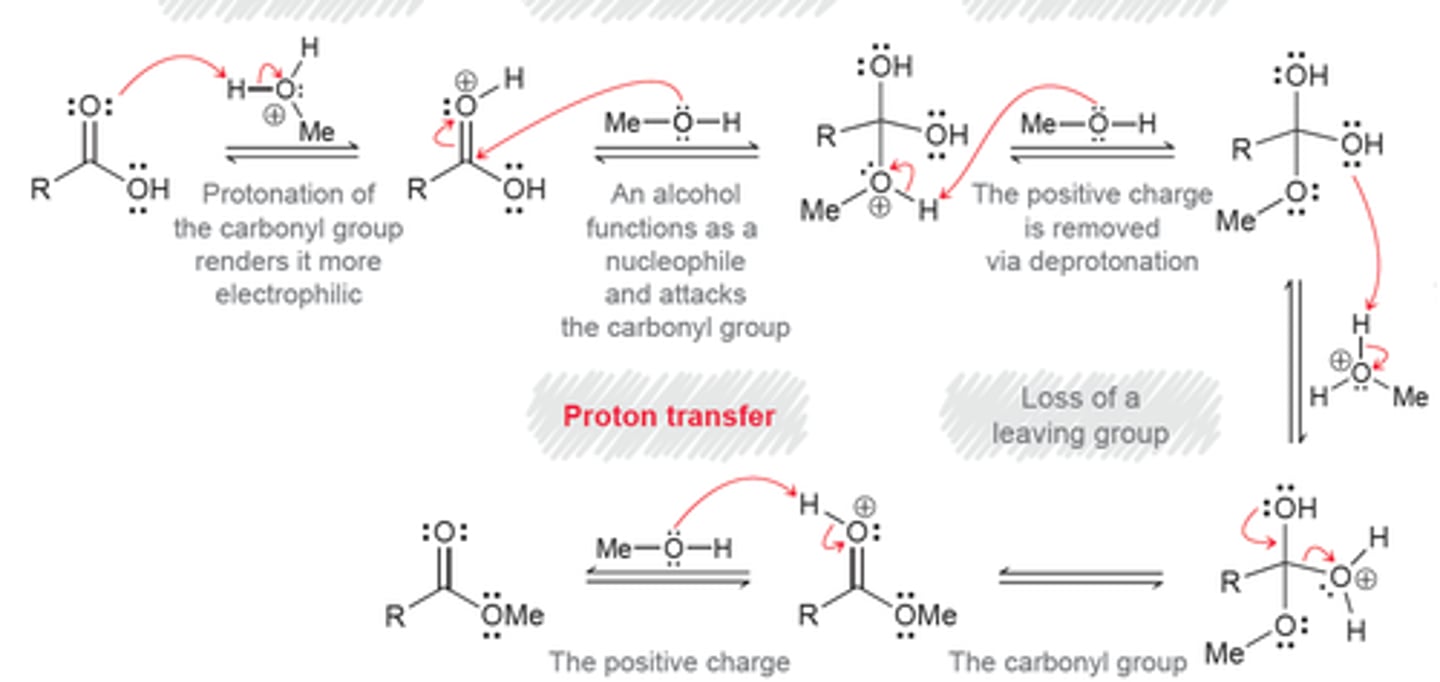
fischer esterification
acid-catalyzed reaction where a carboxylic acid and an alcohol combine to form an ester and water
reversible
each step of the acid-catalyzed esterification is _____
equilibrium
appreciable concentrations of the carboxylic acid and alcohol remain at ______ in the fischer esterification reaction
fischer esterification steps
1. protonation of carbonyl oxygen
2. weakly basic ROH attacks the carbonyl carbon of the carboxylic acid
3. deprotonation
4. left with newly formed ester
reactant, product
to favor the formation of ester, one must either add an excess of one ____ (acid or alcohol) or remove a ____ (typically H2O) as it is formed
leaving group
in fischer esterification, the acid catalyst also functions to protonate the _____
odor, IR
the ester products of the fischer esterification reaction were identified by _____ and _____
alcohol, ester
because every step of the esterification is reversible, an excess of ____ will favor the formation of an ____
fischer esterification mechanism