Electron models
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
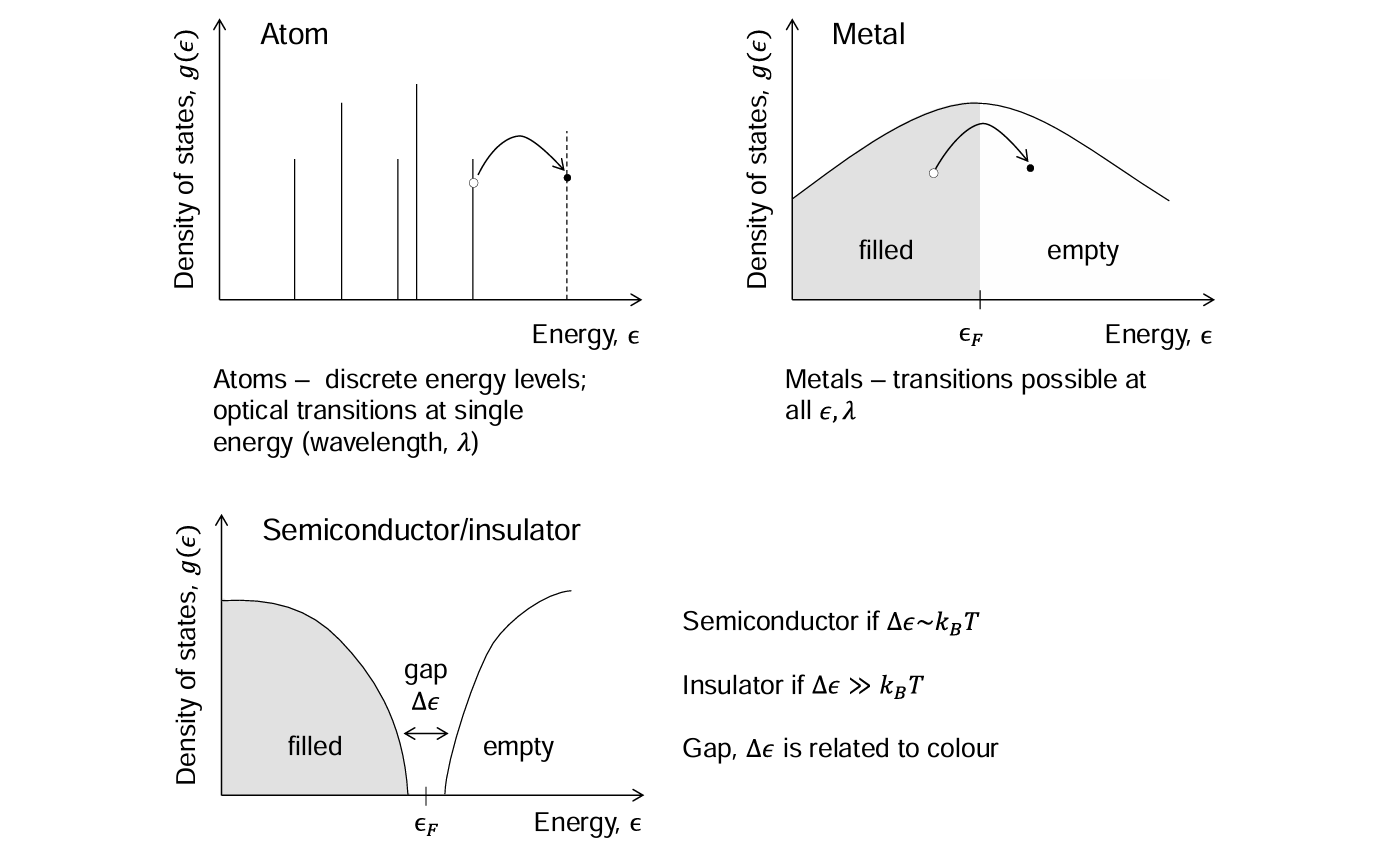
Density of states for insulators vs metals
For a metal, there is no gap between the highest occupied and lowest empty state, and so the density of states forms a continuous spectrum.
For a semiconductor, there is an energy gap of order kBT between occupied and unoccupied states and therefore thermal energy is required for conductivity.
For insulators, the gap is much larger than kBT.
Assumptions for free-electron theory of metals
This model is built on the idea that electrons occupy electronic states in accordance with the Pauli Exclusion Principle, i.e. obeying Fermi-Dirac statistics, and assumes:
A fixed background of static positive charges due to the nuclei (with core electrons), modelled as constant potential. (This is the adiabatic approximation.)
The valence electrons are completely delocalised and propagate freely without interacting with the ion cores or other electrons.
That the crystalline lattice can be ignored.
Total number of electrons in a crystal at T=0
At 𝑇=0, all the states up to the Fermi energy 𝜖=𝜖F are filled and the total number of electrons in the crystal is Ne=∫0ϵFg(ϵ)dϵ.
where g(ϵ)dϵ=2g(k)dk as there are two electron states (up or down) for each value of k.
Fermi energy and wavevector
Found via energy eigenvalues ϵF=2mℏ2kF2
Fermi surface
In k-space, the surface separating the filled states from the empty states, which corresponds to a sphere of radius kF, which only depends on the electron density Ne/V.
The Fermi surface determines the electrical and thermal properties of metals.
Electrons near the Fermi surface have a wavelength comparable to the interatomic spacing and a kinetic energy comparable to the binding energy.
Fermi-Dirac distribution function (quantitively)
At T=0, all states up to the Fermi energy are filled up, (no states are at \epsilon>\epsilon_F), and μ=ϵF.
At a finite temperature, the function is smooth and the occupation number drops more gradually, with a transition region of width ∽kBT .
At ϵ=μ, the distribution function is f(ϵ)=21.
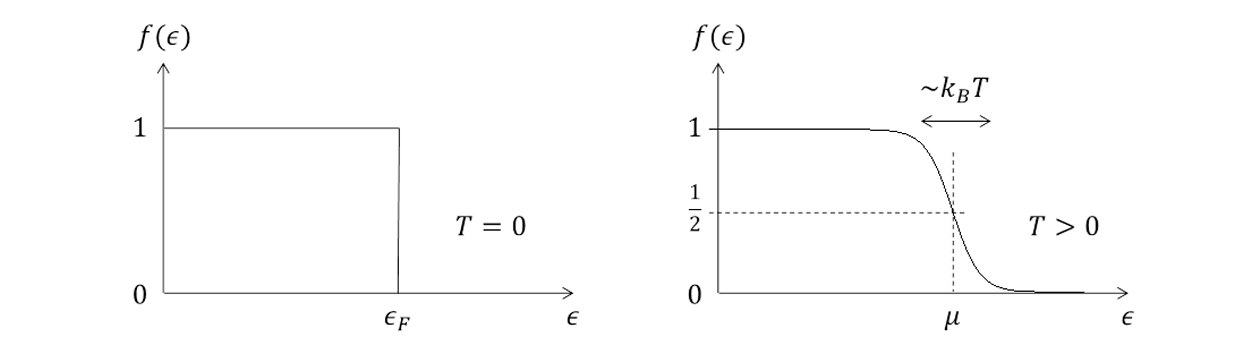
f(𝜖)=e(𝜖−μ)/kBT+11
Number of electrons per unit energy at a given temperature
The number of electrons per unit energy at a given temperature (i.e., at thermal equilibrium) is N(ϵ,T)=g(ϵ)f(ϵ,T).
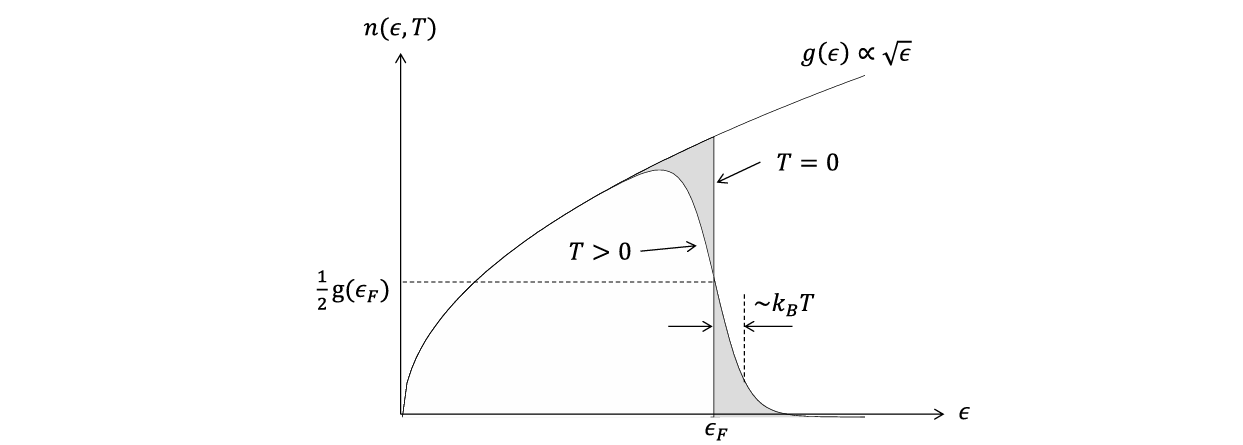
Shaded areas represent areas which would/would not be occupied if T=0.
Electronic heat capacity
Given by CVel=2π2NekBTFT.
If TF is large, at TF∽10000K, we see CVel≪3NekB as we would find classically, and CVel≪CVphonon at room temperature.
At very low T, we find that CVphonon→0 as T3 from the Debye model, whereas CVel∝T.
Total heat capacity of metal at a low T
The general form of the total heat capacity of a (non-magnetic) metal at low temperatures is CVtotal=CVphonon+CVel=βT3+γT
Experimentally, plotting CV/T vs T2 gives a straight line with slope 𝛽 and intercept 𝛾.
The theoretical value for γ tends to be smaller than the measured value, where this difference is counted for by defining an effective mass of electrons.
Effective mass by γ
Defined by mem∗=𝛾(free electron)𝛾(observed)
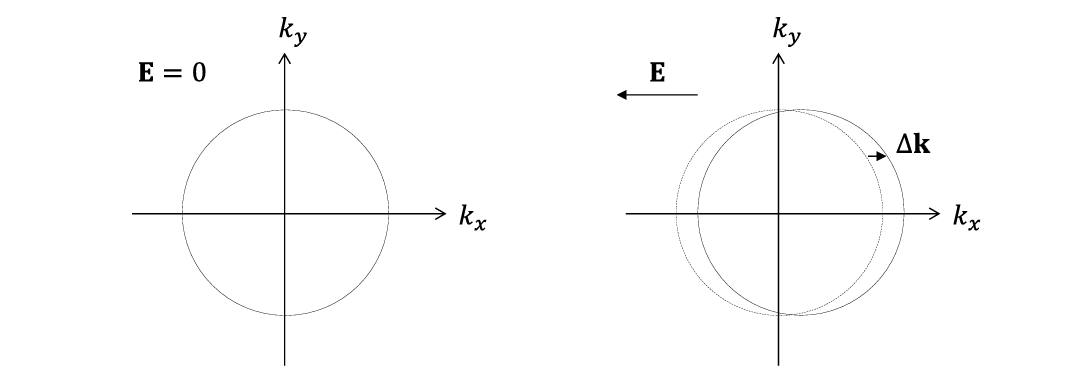
Result of a EM field on the Fermi surface
In the absence of electric fields or thermal gradients, there is no net flow of electrons in any direction, hence the Fermi surface is symmetric around k=0.
In an E field, electrons are accelerated, and the Fermi surface shifts by Δk in the direction opposite to the field.
Why do electrons not accelerate indefinitely in the presence of an electric field?
Electrons are scattered off impurities/defects, phonons, and other electrons. The effects of these collisions can be described by adding a damping term to the equation of motion.
Matthiessen’s rule
The scattering times from different processes add up.
Residual resistivity ratio
The ratio of the total resistivity at room temperature over that at low temperature 𝜌(300K)/𝜌0, where 𝜌0 is the residual resistivity determined by the purity and structural perfection of the sample.
What does the free electron model not explain?
why some materials are not metals (i.e. insulators, semiconductors);
why the mean free path of electrons is much larger than the interatomic distances;
the positive Hall coefficients of some metals;
the large effective masses obtained from heat capacity measurements.
Heat transport in insulators vs conductors
In insulators, heat transport is only due to phonons.
In metals, conduction electrons also contribute, and so metals usually have higher thermal conductivities.
Thermal conductivity with electronic contribution
Electronic contribution due to thermal conductivity is given by κ=31C~VvFl where vF is the mean velocity here, since only electrons within kBT of 𝜖F (i.e., near the Fermi energy) can change their occupation (i.e., be scattered) as 𝑇 varies.
Mean free path is given by l=vFτ, where τ is the relaxation time, which is the characteristic (average) time between collisions of electrons with impurities, defects, or phonons.
Wiedemann-Franz Law
For many simple metals, the ratio of thermal to electrical conductivities is proportional to temperature with a constant of proportionality that is independent of the material.
𝜎Tκ=L
where L is the Lorenz number, which can be calculated from free electron theory, but is not always universal.
The Wiedemann-Franz law breaks down because of our incorrect assumption that electrical and thermal transport are limited by the same scattering processes, i.e., the assumption that 𝜏thermal=𝜏electrical.
Improved Wiedemann-Franz Law
𝜎Tκ=Lτelectricalτthermal
Electrical conductivity limiting processes
Electrical conductivity is due to a uniform shift in k of the Fermi sphere, therefore the conductivity will be limited by processes that restore the Fermi surface to the original position (i.e., its position at zero electric field).
This typically requires a large change in momentum, phonons with a high ∣k∣, which are only available at high temperatures.
Thermal conductivity limiting processes
The thermal conductivity 𝜅 is limited by processes that even out the difference in distributions.
- In thermal transport, there is no net flow of electrons, but electrons travelling in one direction have a broader distribution of wavevectors around kF than those going in the other direction.
- This does not necessarily require large ∣k∣ phonons, and so small momentum scattering events are important.
How to find number density of atoms/electrons n ?
n=VN where N is the number of lattice points per conventional cell, and V=a3.
For FCC, N=4, for BCC, N=2.
Some structures will have multiple atoms per lattice point, for example, NaCl is FCC structure with 2 atoms per lattice point, giving N=4×2=8.
For electrons, pay attention to how many free electrons there are per atom.