chem lab final
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
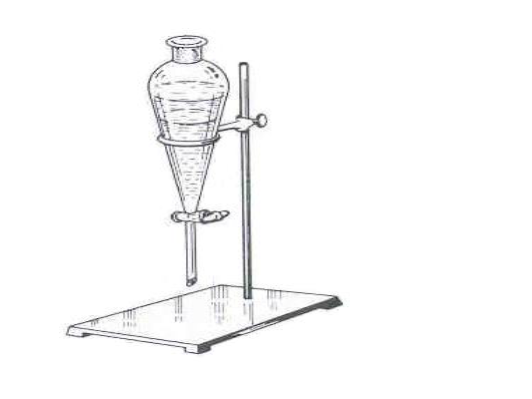
What is this?
Separatory funnel
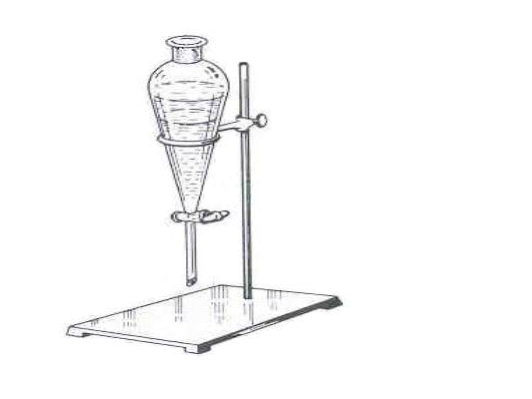
What is this objects function?
liquid-liquid extractions
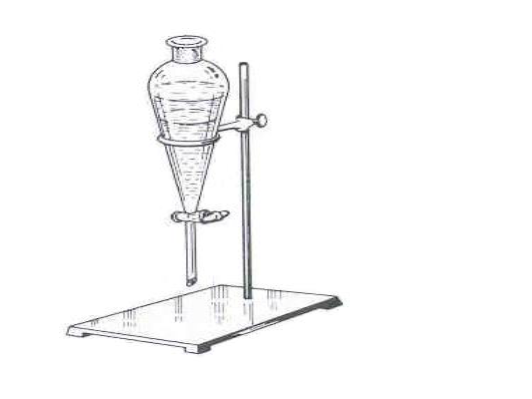
Are there any special considerations for the assembly or usage of this object?
Before adding anything through the funnel, ensure it is closed. Before draining, glass plug needs to be taken out, and while mixing gas needs to be let out.
What is this?
Condenser
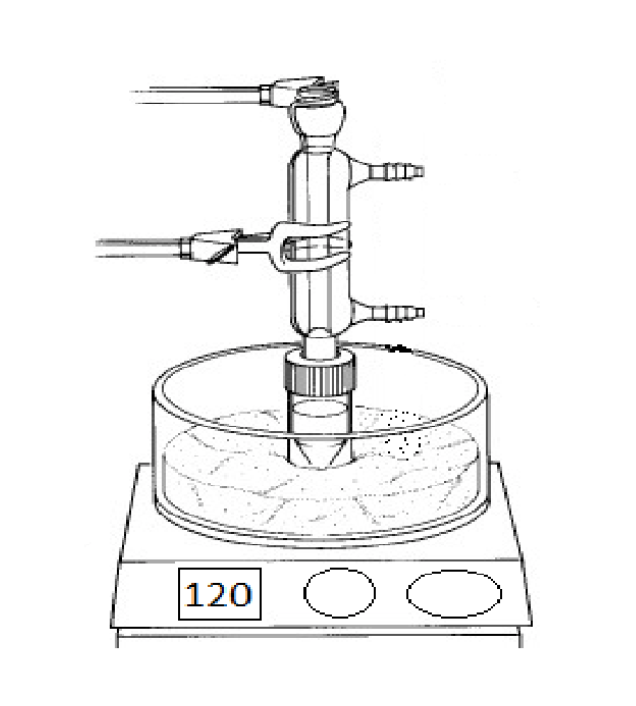
What is the purpose of this object?
To heat reactions without losing solvent
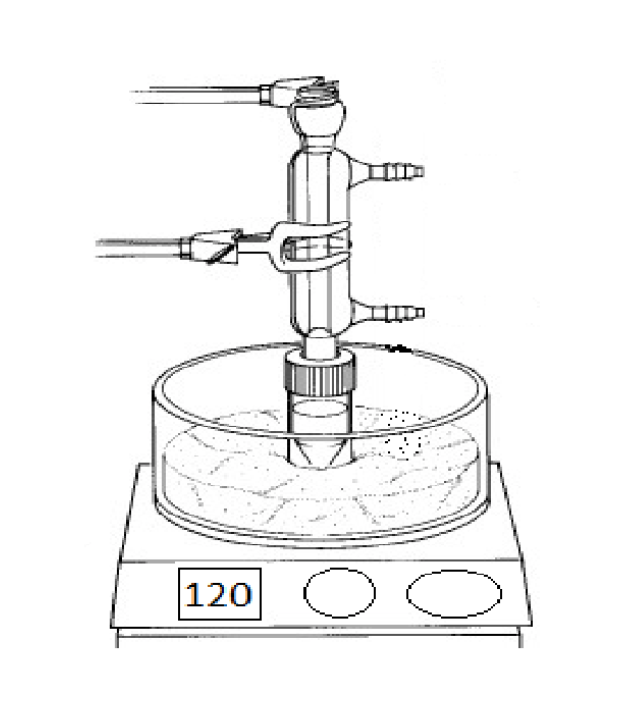
Are there any special considerations for the assembly or usage of this object?
Cold water enters from the bottom port and exits the top, and the top of the condenser is open.
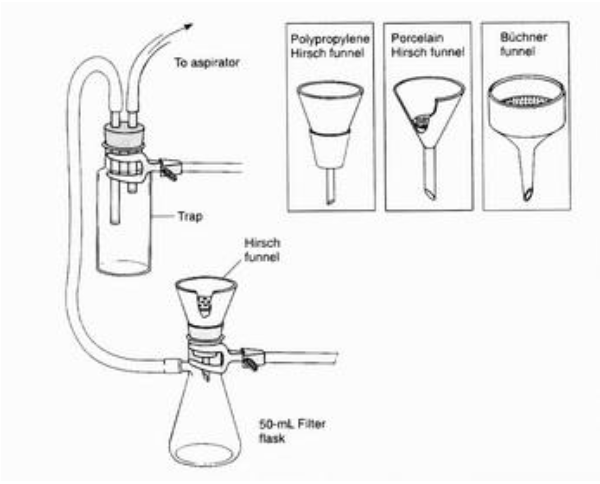
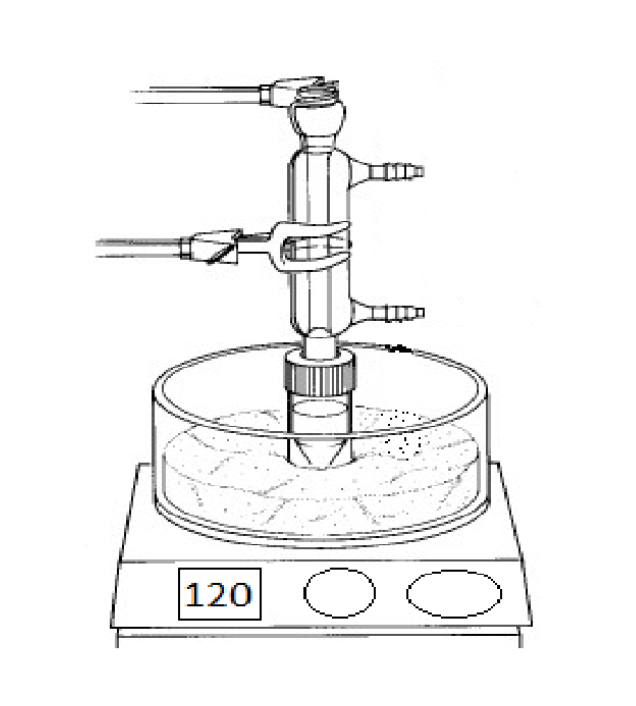
What is this?
A vacuum filtration system
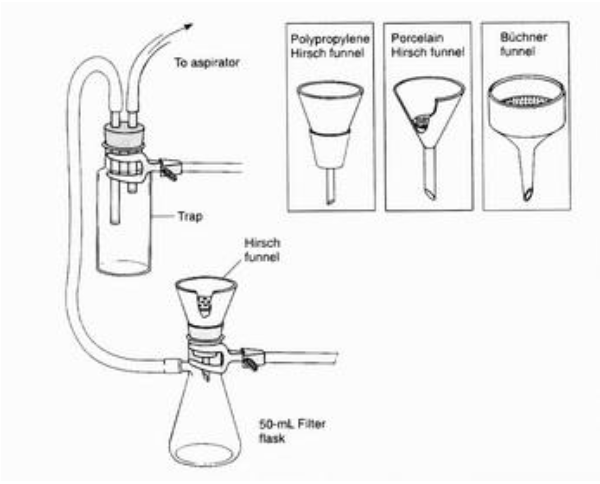
What is the purpose of this object?
Extract liquid solvent from the solid.
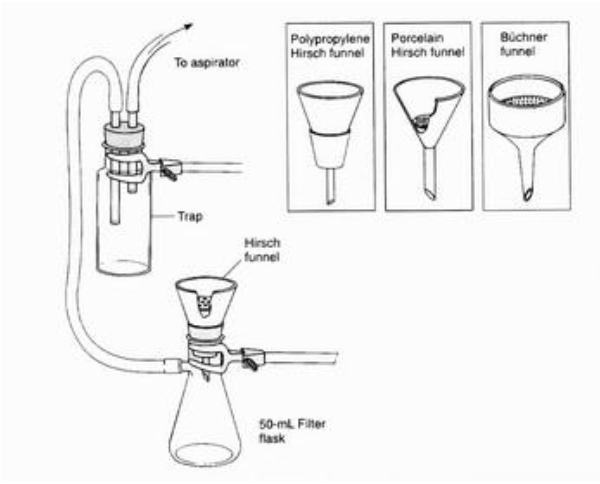
Are there any special considerations for the assembly or usage of this object?
Ensure that the little white thing is in the funnel, and have a second flask between the vacuum and the filter to prevent backflow or water entering the vacuum.
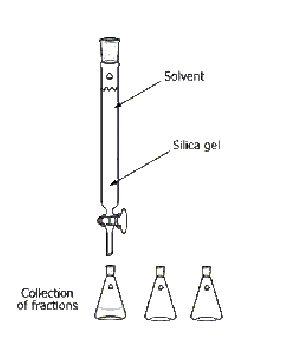
What is this?
Column Chromatography
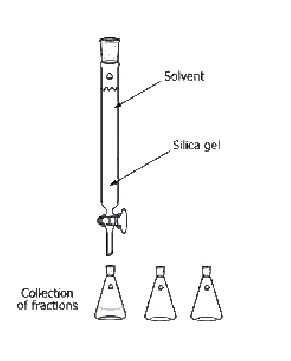
What is the purpose of this?
separate two compounds from a mixture
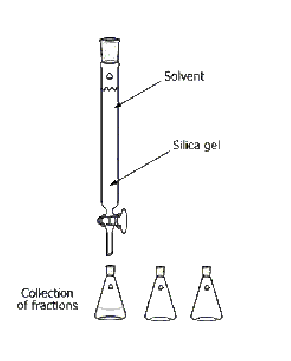
Are there any special considerations for the assembly or usage of this object?
Mixture placed on the top of the silica gel, the solvent must be continuously added, and the solvent must be changed when the polarity changes.
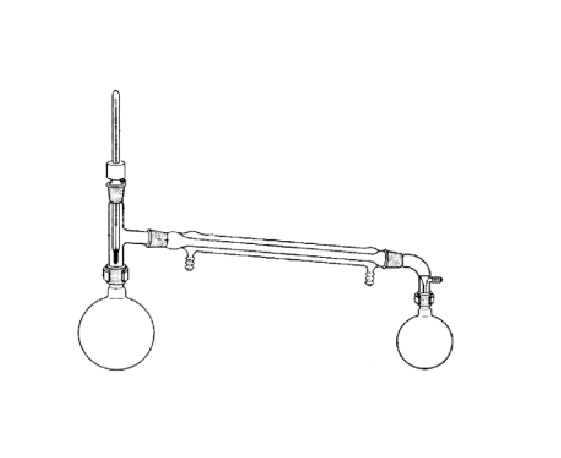
What is this?
A simple distillation apparatus.
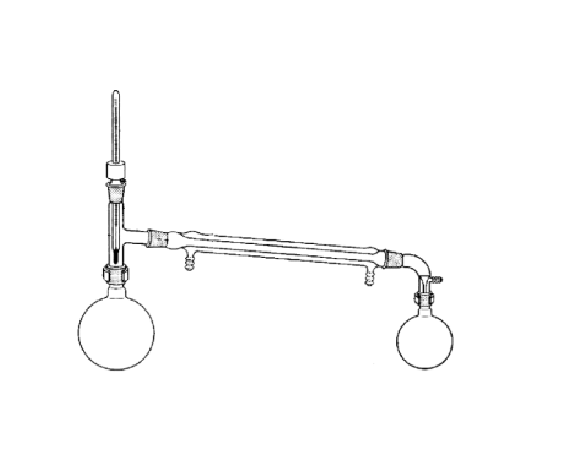
What is the purpose of this?
Collect the component with the lower boiling point evaporates first, and the condenser cools the vapor into liquid so it can be collected.
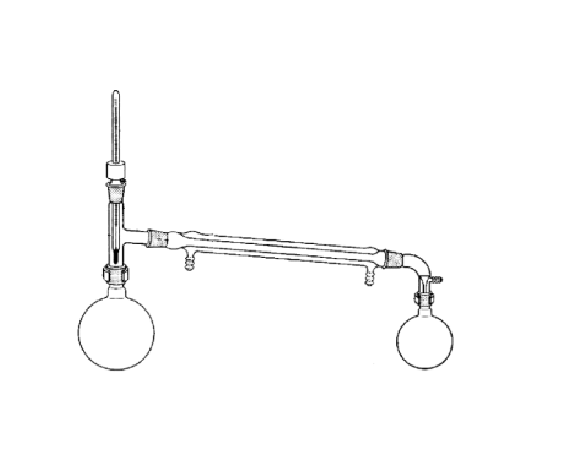
Are there any special considerations for the assembly or usage of this object?
Bulb of the thermometer must be below the side arm of the three-way adapter, water must flow so it enters the bottom of the condenser and leaves through the top.
How are simple and fractional condensers different?
Fractional condensers have a fractioning column that is placed between the boiling flask and the distillation head.
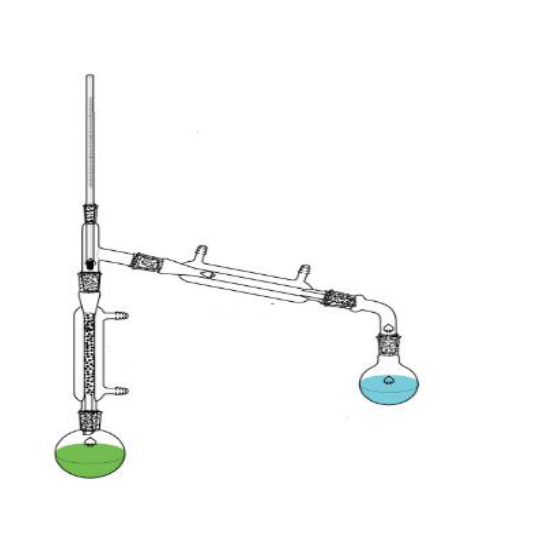
What is this?
Fractional distillation apparatus.
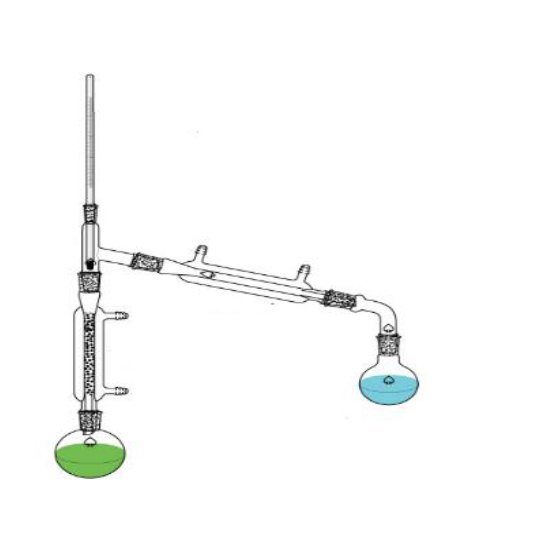
What is the purpose of this?
Separate miscible liquids.
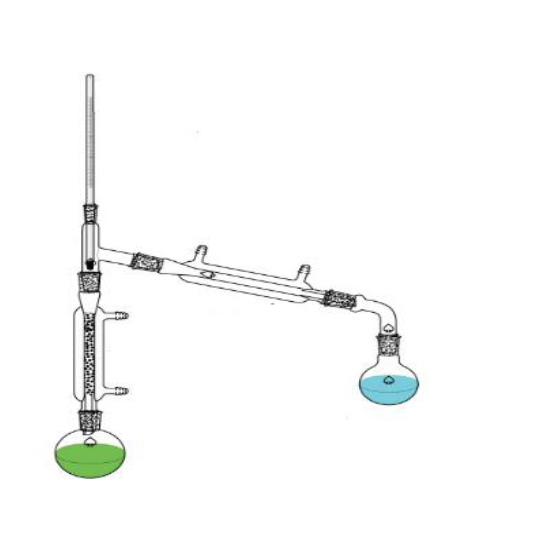
Are there any special considerations for the assembly or usage of this object?
Make sure that the flask is not empty before the distillation ends and the heat source must me carefully controlled so things dont boil over.
Equation for percent yield
actual yield/theoretical yield x100%
What happens if you add water to a reaction mixture
shifts equilibrium
Rf equation
distance of substance/distance of solvent x100%
Which compounds are more polar on TLC plates
The ones that travel less distance.
Percent composition equation
area of component 1/ total area of all components
how to determine dipole vector
towards more electronegative atoms, double bonds do not effect unless it is symmetrical.
Is the compound with higher or lower density on the top for separation
Lower density stays on top
Explain the difference between an extraction and a wash during a separation
Extraction moves a desired product from a mixture into a new solvent, while a wash removes undesired impurities from the desired product, which stays in the original solvent
What step is usually performed after an organic extraction and separation from an aqueous mixture?
What compounds did we use in lab for this step
Drying the organic layer with a drying agent like Mag sulfate
When using melting point to determine the purity of a solid, what indicates a pure sample and what
indicates an impure sample? Explain
A melting point range that includes the expected melting point indicates a pure sample, while an impure sample has a lower melting point
What would be the explanation for a broader melting point that is lower than the literature value?
an impurity
Theoretical yield equation
(Moles of Limiting Reactant) x (Mole Ratio of Product/Reactant)x (Molar Mass of Product)
How to determine the limiting reagent
divide the weight of your compound by the molecular mass, then compare that to the other compound
How to do the theoretical plate.
start at the composition of the greater compound, then follow the line up until it hits the curve for the lower boiling point compound. then follow the graph until ypu hit a point on the higher boiling point then follow down to see what percentage it is it
what is a possible solution to improving the separation of the spots
use a more polar developing solvent
What type of distillation work best for liquids with greater than 100 degree celcius difference in boiling points?
simple distillation
What type of distillation work best for liquids with less than 100 degree celcius difference in boiling points?
fractional distillation