19: Amino Acid Oxidation and Urea Cycle
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Why does urea have to made?
Avoid circulating NH3, highly reactive and causes pH problems in blood + disturbs brain function
When are amino acids used as fuel
not needed for new protein synthesis
during starvation when carbohydrates unavailable
Alpha-keto acids
amino acids where amine group has been removed
key amino acids transported/delivered for deamination in the liver
glutamine
glutamate
alanine
enzymes that move amino groups between different carbon skeletons
aminotransferase
main molecule(s) responsible for transporting ammonia in the blood
glutamine, alanine
Alanine vs glutamine
Since alanine = 3C molecule, lose less carbon by transporting it from muscle compared to glutamine
amino acids that end up as TCA intermediates that can contribute Cs to synthesis of glucose
glucogenic amino acids
amino acids that end up as ketone bodies
ketogenic amino acids
Strictly ketogenic amino acids
Lysine
Leucine
urea structure benefits
2 amines and 1 carbon —> gets rid of more amines and not a lot of carbons
Well oxidized
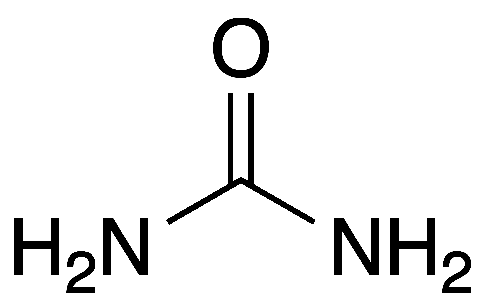
2 nitrogens on urea from…
aspartate
carbamoyl phosphate
3 sets of ketoacids
alpha-ketoglutarate to glutamate (5C)
Alanine to pyruvate (3C)
OAA to aspartate (4C)
ketoacids
Required to continue urea cycle
ornithine
non-protein amino acids in urea cycle
ornithine
citrulline
Arginosuccinate broken down to…
Fumarate (TCA)
Arginine (urea cycle)
Regulation of urea cycle
transcriptional regulation of genes for urea cycle enzyme (long-term changes responding to diet)
regulation of carbamoyl phosphate synthase I by N-acetylglutamate