enriched chem midterm terms
1/112
Earn XP
Description and Tags
most if not all of the terms from the terms sheet!!
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
113 Terms
Chemistry
the study of matter and the changes it undergoes
atom
a collection of protons and neutrons in the center surrounded by a cloud of electrons
Compound
2 or more elements that are chemically bound
molecule
a combination of 2 or more pure substances that are not chemically bound
element
a pure substance with one type of atom
solid
A state of matter characterized by structural rigidity and resistance to changes in shape or volume, where particles are closely packed together.
liquid
A state of matter that has a definite volume but takes the shape of its container, characterized by the ability of particles to flow past one another.
gas
A state of matter with no fixed shape or volume, where particles are far apart and move freely.
change of state
The process in which a substance transitions from one state of matter to another, such as solid to liquid or liquid to gas, often due to changes in temperature or pressure.
physical change
no new substances formed/new molecules, no bonds broken or formed, still the same substance just a different arrangement
Ex: boiling, melting, freezing, condensation, evaporation
chemical change
new substances are formed, bonds are either broken or formed or both
Ex: color/odor change, explosion, transparent to opaque, precipitate formed, bubbles, heat increased or decreased,
group
Other names: family, column
18 columns
Columns (1A-8A: main group elements) correspond to the number of valence electrons in the periodic table, with similar chemical properties.
Three major divisions: metals, metalloids, and nonmetals.
period
Other name: row
7 rows
Corresponds to the number of energy levels contains electrons in their group state in the periodic table, with each period representing elements that have the same number of electron shells.
metals
(lower, left)
Typically solid at room temp.
Ductile = can be made into wire
Malleable = bendable
Metallic luster (shiny)
Good conductors (heat + energy)
Metals tend to lose electrons easily (from cations)
nonmetals
(higher, right)
Can be solid, liquid, and gas at room temp.
Dull (not shiny)
Brittle (not malleable)
Insulators (not a good conductor)
Tend to gain electrons (from anions)
metalloids
(zig-zag down from 13th column corner)
Properties of both metals + nonmetals (in between them)
Metallic luster (shiny)
Brittle (not malleable)
semi-conductors
filtration
particle size based separation
quantity
The amount or number of a substance involved in a chemical reaction or present in a solution.
SI base units
Base Units:
Length = meter
Mass = kilogram
Time = seconds
Temperature = kelvin
Amount of substance = mol
scientific notation
#’s are wrote in the form m x 10^n for the factor m ≥ 1 and m ≤ 10, n = whole
#Determine n by counting # places that moved decimal
Move left → n = positive
Move right → n = negative
volume
The amount of space occupied by an object.
L x W x H
m3 or cm3
density
mass/volume
accuracy
The closeness of measurements to the correct or accepted value of the quantity measured.
close to the accepted value
precision
The closeness of a set of measurements of the same quantity made in the same way.
closeness to one another (not necessarily the accepted value)
sig figs
rules:
estimate one place beyond the smallest division
the last digit contains the uncertainty
counting zeros: # of digits = # of sig figs
zeros are counted when:
they are between non zeros or sig figs
they are after a non zero and a decimal is present
dimensional analysis
symbol | name | conversion |
k | kilo | 1 kilo - 103 base |
h | hecto | 1 hecto = 102 base |
da | deca | 1 deca = 10 base |
d | deci | 1 base = 10 deci |
c | centi | 1 base = 102 centi |
m | milli | 1 base = 10³ milli |
atomic mass unit
AMU
a measure of an atoms atomic mass
atomic number
the number of protons
average atomic mass
the average mass of an atom of a given element — abundance a x mass a + abundance b x mass b
avogadro’s number
6.02× 10²³
isotopes
different atoms of the same element with a different number of neutrons
mass number
the number of protons and neutrons combined
the mole
the amount of substance that contains as many particles as there are in 12.0 g of C-12
molar mass
the mass in grams of 1 mole of a substance
mole conversions
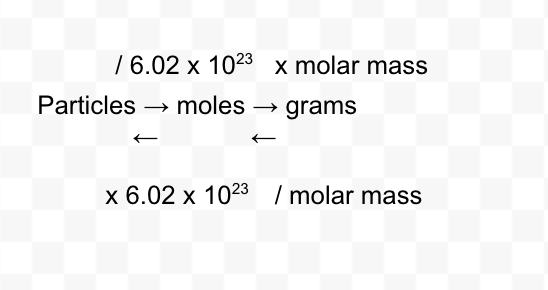
bohr
simple model of the atom based on understanding of the sharp line emission spectra of excited atoms
continuos spectrum
A continuous spectrum includes all wavelengths of light without gaps, representing a smooth transition of colors, typically produced by heated solids or liquids.
electromagnetic radiation
A kind of radiation including visible light, radio waves, gamma rays, and X-rays, in which electric and magnetic fields vary simultaneously.
electromagnetic spectrum
higher frequency = high energy
long wavelength = low energy
red (700 μm) → violet (400 μm
radio → micro → infrared → visible (red → violet) → x-ray → gamma
low energy/frequency/wavelengths → high energy/frequency/wavelengths
excited state
emit light of certain wavelengths — this determines its color and depends on the element
frequence
cycles/sec
ground state
lowest possible energy level
speed of light
3.00 × 108 m/sec
planck
Solids emit radiation as heated; study the relationship between wavelength and intensity of radiation emitted and temperature.
planck’s constant
6.625 × 10-34 j x s
line emission spectrum
when atoms absorb energy, that energy is often released as light energy — when that energy is passed through a prism, a pattern is seen that is unique to that type of atom (this is the emission spectrum)
falls = emits ⬇
jumps = absorbs ⬆
to 1st = UV light
to 2nd = visible
to 3rd = IR
photon
electromagnetic energy with the properties of waves and particles
energy of a photon = hc/λ
quantum
a discrete amount of energy released when an electron rests
wavelength
the space between adjacent waves — λ
photoelectric effect
When light shines on a metal, electrons are ejected from it. (WHY metals appear shiny).
The ejected electrons have a certain amount of KE
Depends on wavelength, not intensity
Proves light not only behaves as waves but also as photons (particles)
heisenberg uncertainty principle
light starts to fan out when it reaches a certain point
orbital
A mathematically described region around a nucleus in an atom or molecule that many contain zero, one, or two electrons.
quantum theory
Quantum objects have both particle-like properties (such as mass, charge, and energy) and wave-like properties (such as wavelength and frequency).
aufbau principle
electrons will occupy the lowest potential energy orbital possible
hund’s rule
aka the roommate rule — all orbitals of equal energy must have one electron before any have 2 electrons
noble gases
full valence shell (8)
rare gases
stable
noble gase configuration
[closest noble gas] rest of electron configuration
pauli exclusion principle
electrons in the same orbital must have opposite spins (beds facing opposite ways)
quantized
electrons could only have very specific amounts of energy and traveled in orbits that were at fixed distances from the nucleus
mendeleyev
made the periodic table — predicted there would be new elements in the spots he had left blank, predicted densities based on properties of other atoms around blank ones
moseley
established the concept of atomic numbers — essentially discovering protons, ordering of the wavelengths of the x-ray emissions of the elements coincided with the ordering of elements based on atomic numbers
actinide
Inner transition metal: A series of metallic elements (atomic number 89 - 103).
Radioactive
Soft, shiny, silver-colored
All radioactive
First four are naturally found, the rest are lab-made
lanthanide
Inner transitional metal: A series of metallic elements( atomic number 58-71).
All very similar in properties
Shiny metals
Similar reactivity of that of alkaline earth metals
periodic law
states that the properties of elements are periodic functions of their atomic number
alkali metals
Valence e- configuration = 1 val e-
Soft, silvery, shiny
Highly reactive (not free elements in nature, always found w/ another element)(lose e- easily)
alkaline metals
Valence e- configuration = 2 val e-, s2
Less reactive than 1A (still too reactive to be found free in nature)(lose e- easily)
Forms bases when it reacts with water
Harder, denser, higher melting point than 1A
Moderately metallic
halogens
salt formers (with alkali’s)
brinclhoff’s: gases found diatomically in nature: Br, I, N, Cl, H, O, F
most reactive nonmetals
transition metals
good conductors, harder, denser, higher melting point than s and p block elements, less reactive than s block elements
metals lose electrons to form positive
cations
nonmetals gain electrons to form negative
anions
shielding effect
the repulsion of outer electrons by inner electrons
F = (+ charge) (- charge) / r2
effective nuclear charge (z*)
net positive charge that is attracting a particular electron
protons - inner electrons
atomic radius
size of the atom
get smaller as you go from left to right
the more electrons you add, the more protons are attracted
the bigger they are, the weaker they hold their electrons
ionic radius
The distance between the nucleus of an ion and the outermost shell of the ion.
ionization energy
energy that an atom needs to absorb in order to lose an electron
successive ionization energies
each successive removal of electron requires more energy than the last with it having a sizeable energy cost for the inner shell electrons
valence electrons
electrons in the highest energy levels s and p subshells (lose or gain to complete shell — except for noble gases)
electronegativity
the strength an atom has when attracting electrons in a bond
increases as you go up and to the right
reactivity
metals have a low Ionization energy and lose electrons to become smaller cations, reactive, conductive, not brittle
nonmetals have high electron affinities and gain electrons to become larger anions, brittle not conductive
metallic character
The level of reactivity of a metal (increases going down + right).
Metals tend to lose electrons in chemical reactions (due to low ionization energies and low electronegativities)
chemical bond
Hold molecules together and create temporary connections that are essential to life.
ionic bonds
when one electron is transferred from one atom to another resulting in oppositely charged particles with an attraction to each other (between a metal and a nonmetal)
covalent bond
2 elements of similar strength and electronegativity, nonmetals (some metalloids) sharing electrons
nonpolar covalent
electrons shared equally
polar covalent
electrons shared unequally
bond polarity
Describes how the electric charge is distributed across a chemical bond between two atoms.
molecular compound
A chemical compound is made up of two or more nonmetal atoms that are held together by covalent bonds.
Shares electrons
Lowers melting points/boiling points because they’re easier to break
chemical formula
Tells us the number of atoms of each element in a compound.
formula unit
ratio of atoms
octet rule
atoms must have a complete valence shell (8 electrons)
lewis dot structures
A diagram that shows how electrons are arranged around atoms in a molecule.
single bond
longest, weakest, 2 electrons
double bond
in between strength and length, 4 electrons
triple bond
6 electrons, shortest, strongest
lattice energy
energy released when 1 mole of an ionic compound forms from its ions
VSEPR theory
Valence Shell Electron Pair Repulsion → molecule adopts the shape that minimizes the electron pair repulsions.
molecular shapes
Electron Pair Geometry: lone pair on central atom + atoms bonded to central atom
2 = Linear: 180º
3 = Trigonal-planar: 120º
Trigonal-planar (e- pair geometry) + bent (molecular geometry): <120º
4 = Tetrahedral: 109.5º
Tetrahedral (e- pair geometry) + Trigonal-planar (molecular geometry): <109.5º
Tetrahedral (e- pair geometry) + Bent (molecular geometry): <<109.5º
polarity
The polarity of each molecule has to do with the unevenness of electron distribution.
Polar = a positive side and a negative side
Nonpolar = symmetrical, no positive + negative charge (they cancel each other out)
dipole
integrates polarity/asymmetry (point towards negative)
Lone pair = negativity
Shown through an arrow pointing to the negative side
intermolecular forces
the attractive and repulsive forces that arise between the molecules of a substance