MICROBIO ENTIRE COURSE
1/421
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
422 Terms
pathogen classifications (from most infectious to least)
bacteria
fungi
worms
viruses
parasites (protozoa)
outbreak, endemic, epidemic, and pandemic
outbreak
the occurrence of disease cases in a community, region, or specific group (ex. school/hospital) at a frequency clearly in excess of normal expectancy
endemic
disease is always present in a population within a region
epidemic
a rapid increase in cases higher than expected in a population within a region
ex. SARS, ebola
pandemic
an epidemic that becomes widespread over several countries or continents
ex. COVID
6 steps of the chain of infection
infectious agent
bacteria, virus, fungi, protozoa, parasite
reservoir
people, equipment, water
portal of exit
excretions, secretions, droplets
mode of transmission
contact, droplet, airbourne, or vectorbourne
portal of entry
resp tract, GI tract, mucous membranes, skin
susceptible host
people with chronic disease, immunocompromised, babies, elderly
5 opportunities to break the chain of infection
the infectious agent is eliminated, inactivated, or cannot survive in the reservoir
disinfecting the environment
the portal of exit is managed through good infection prevention and control practices
hand hygiene, PPE
transmission does not occur due to good infection prevention and control practices
hand hygiene, isolation of infected pts, air flow where necessary
the portal of entry is protected
aseptic non-touch technique, safe catheter care, wound care
reducing the susceptibility of pts receiving healthcare
treating underlying diseases, recognizing high risk pts
what is infection
infection: the invasion of the host by microorganisms, which then multiple in host tissues - NOT disease
dependent on 3 main factors
number of organisms in or on the host
virulence of the organism
host defences or degree of resistance
what is virulence
the ability of an organism to cause infectious disease
the reason why some pathogens cause disease, and others done
some infectious agents are easily transmitted, but they are not very likely to cause disease (contagious, but not virulent)
ex. ebola vs. polio (COVID is both)
5 factors that contribute to host susceptibility
dependent on physiologic and immunologic conditions of the host
immune system
age - infants and elderly!
nutrition
genetic defecits
medications (chemo)
what is host resistance
the ability of the host to prevent infection from occuring and infectious diseases from developing
non-intact barriers (skin, mucous membranes, Gi and resp tract)
chemical (gastric juices, saliva)
immunity against a particular agent (natural and artificial)
chicken pox
what are antigens
antigens: chemical substances that identify as foreign that continually challenge the immune system
can be bacteria, viruses, etc
OR from the environment like blood transfusions, vaccines, transplant tissue, or bee venom
OR pre-cancerous cells
innate vs adaptive immune system mechanisms
has 2 systems to protect against pathogens
innate immune system
barriers - 1st line of defence
innate cells - 2nd line of defence
adaptive immune system
B lymphocytes (humoral)
T lymphocytes (cell-mediated)
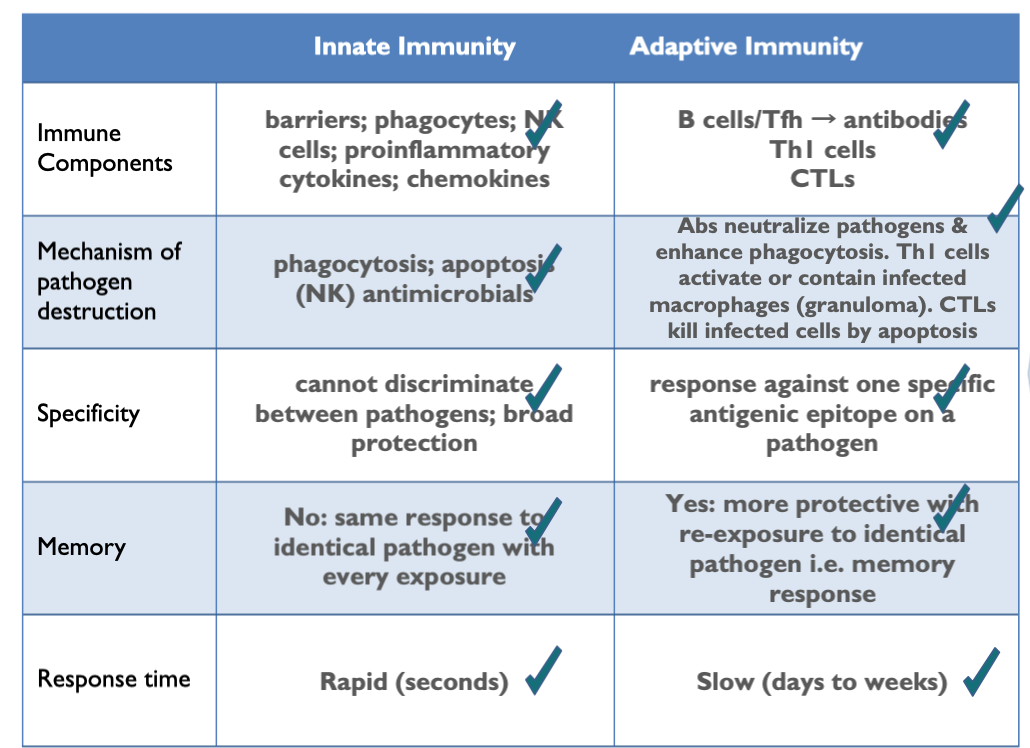
what is the function of the immune system
main function is to protect body from pathogens that can make you sick
destroy abnormal cells (cancerous)
has 2 systems to protect against pathogens
innate immune system (barrier and innate cells)
adaptive immune system (B and T lymphocytes)
properties of the innate immune system
innate = in body since birth
does not require memory and does not have memory (non-adaptive)
responds immediately and the same strength every time
is always initiated when pathogen contacts body, is non-specific
uses Pattern Recognition Receptors (PRRs) that recognize microbial structures
this is to identify self and ensure the immune system does not destroy its own cells marked by PRRs
has first and second lines of defence
what are portals of entry + examples
microbes enter in body by various methods
most pathogens have specific portals of entry
skin (anything that breaks skin layers - trauma/burns)
GI tract (fecal-oral, or contaminated food)
resp tract (airbourne droplets, or contact)
urogenital (local invasion/STI or ascending infections/UTI)
7 main barriers in the body
skin
tightly knit epithelial cells that keep bacteria out
provides physical barrier to the entrance of microbes
acidic pH discourages the growth of organisms
fatty acids, sweat and oil gland secretions kill many bacteria
sloughing off of old skin
tears
wash away irritating substances and microbes
lysozyme kills many bacteria
saliva
washes microbes from the teeth and mucous membranes of the mouth
resp tract
mucus traps organisms
cilia sweep away trapped organisms
goblet cells that secrete mucous, that protect structures and trap microorganisms
stomach
acid kills organisms
antibacterial peptides (pepsin)
microflora and good bacteria to help keep foreign bacteria in check
large intestine
normal bacterial inhabitants keep invaders in check
bladder
urine washes bacteria out
NOTE: microflora associate with epithelial cells that line all pathogen entry points
the WBCs of the innate immune system
mainly leukocytes
neutrophils
monocytes
macrophages
eosinophils
basophils
dendritic cells - bone marrow derived WBCs
mast cells - found in connective tissues
NK cells - innate and adaptive immune system
what are phagocytes
defensive cells, white blood cells
eat unwanted invaders of the body and dead/damaged cells
non-specific (will eat any pathogenic organisms)
main function: destroy extracellular pathogens
the 3 main phagocytes
NOTE: these destroy extracellular pathogens by phagocytosis
neutrophils
first responders
live in blood
macrophage
arise from blood monocytes
can consume larger particles that neutrophils cannot handle
immature dendritic cells
an important intermediary between the innate and adaptive immune system
a mature dendritic cell is more involved in adaptive immunity
what is phagocytosis + 3 steps
ingesting and killing invading pathogens
recognition and adherence
engulfment
intracellular killing
phagocytosis - recognition and adherence
occurs when a pathogen-associated molecular pattern (PAMP) expressed by a microbe, binds will a PRR receptor on the membrane of a phagocyte
phagocytosis - engulfment
extension of the cytoplasm move around and eventually enclose the particle in a phagosome (membrane)
phagocytosis - intracellular killing
phagosome fuses with the cytoplasmic lysozome, forming a phagolysozome
contains antibacterial molicles and enzymes that can kill and digest microbes
accomplished through toxic reactive oxygen species (hydrogen peroxide) and nitrogen containing species (nitric oxide)
any indigestible material is then removed through exocytosis
what are eosinophils
type of leukocytes
defence against parasitic infections
able to digest much larger particles
also have role in allergic reactions
what are natural killer cells
goal is to survey the whole body and identify + destroy virally-infected cells and tumour cells
each NK cell has an activating receptor and inhibitory receptor
all healthy cells have an MHC1 complex sticking out of their cell, while infected cells do not - when NK cells connect with healthy cells, they are inhibited by MHC1 and do not kill this healthy cell
if not inhibited (with infected cells), the NK injects toxic cytoplasmic granules which perforates the cell resulting in viral DNA and RNA destruction, or apoptosis
kills INTRAcellular viruses
what are defence proteins
complement system (works with antibodies)
30 or more plasma and cell surface proteins that work in concert to fight invading microorganisms through OIL
opsonization → easier phagocytosis
complement system acts as a glue to help attach antigens to macrophils/neutrophils for easier phagocytosis
inflammation
histamine release by mast cells/basophils to dilate blood vessels and making more them leaky
lysis
complement system induces bacterial lysis/burst due to water entering
pro-inflammatory cytokines
type of defensive protein
part of an integrative signalling network to encourage inflammation and innate immunity via chemotaxis of leukocytes
chemotaxis: the movement of an organism in response to a chemical stimulus
main types of cytokines
chemokines
interferons (IFN)
interleukins (IL)
tumor necrosis factor (TNF)
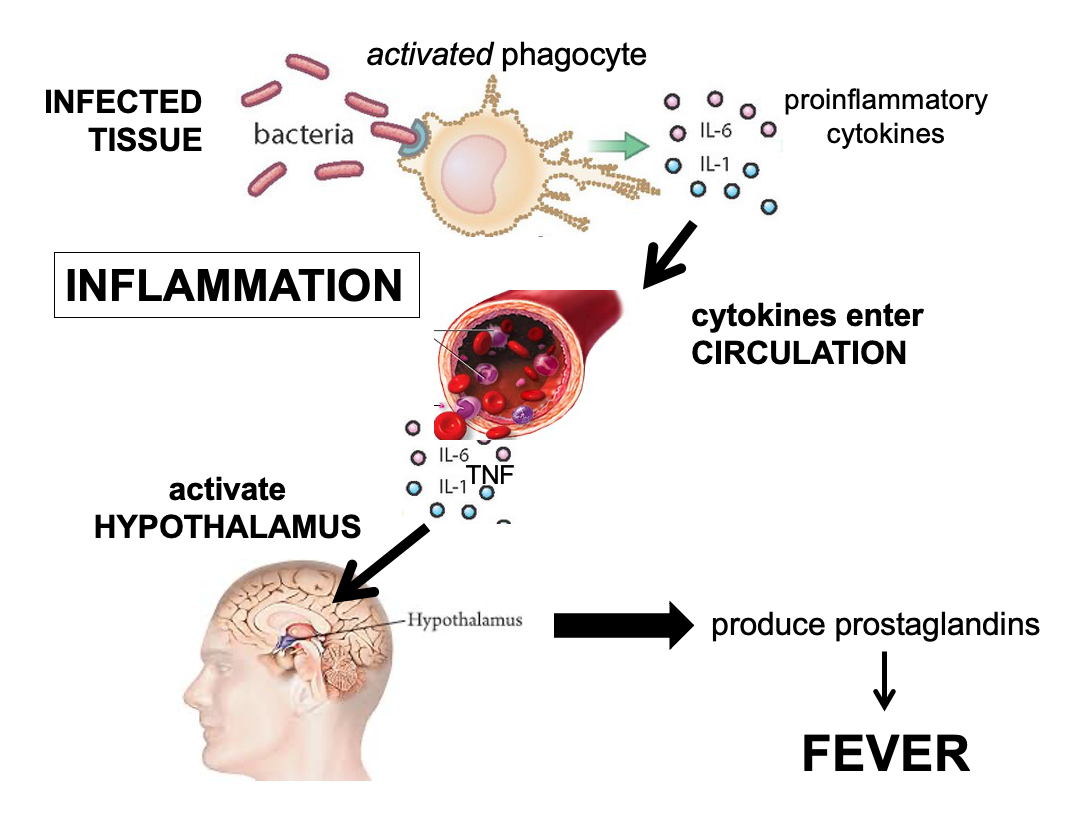
the process of inflammation
recruits circulating immune cells and plasma proteins into infected tissue to encourage immune defences
occurs in 3 main steps
activated phagocytes secrete chemokines and pro-inflammatory cytokines
vasodilation and increased vascular permeability cause redness, heat, and swelling
inflammatory cells migrate into tissue, releasing inflammatory mediators that cause pain
fever = higher temp to kill off bacteria, helps slow bacteria growth, and speed up body defences
properties of adaptive immunity
can distinguish one specific microbe from another
develops memory for subsequent exposures of that same microbe
two types:
humoral immunity (B lymphocytes)
cell-mediate immunity (T lymphocytes)
autoimmune (AI) disorders → cannot distinguish microbial antigens from self antigens
what is humoral immunity
involves the production of antibodies by plasma cells in response to a pathogen
main function is to produce specific antibodies to fight against the particular EXTRAcellular pathogens
arise in the fetal liver and from stem cells in the bone marrow where they mature
once the mature, they enter the bloodstream where they migrate and are stored in the lymphoid tissue (spleen + lymph nodes)
the process of B cell activation
they must be activated before maturation - exposing them to an antigen
antigens binds to the B cell surface receptor
activates the B cell
B cell proliferate to make identical cell copies
these copies then differentiate to become either antibody-producing (plasma) cells or memory cells
plasma cells secrete antibodies; non-dividing B cells and only last about a few weeks + die by apoptosis
memory cells live for many years and recognize + respond to multiple exposures of the same microorganism that initially stimulated their formation (primary and secondary responses)
the 5 antibodies
IgG
most abundant, crosses placenta for 3 months of neonatal immunity, neutralizes and opsonizes pathogens
IgA
dominant mucosal antibody in bodily secretions, blocks pathogen entry at GI and resp tract, provides passive protection to breastfed infants
IgM
prominent in the primary antibody response, main function is to neutralize pathogens
IgE
triggers mast cell-mediated type i rxns and some antiparasitic responses, does not neutralize pathogens or promote phagocytosis
IgD
mostly remain bound to naive B cells - main function is to bind antigens (b cell receptor)
2 ways antibodies eliminate pathogens
neutralization
blocks pathogen receptors that are used to gain entry into a cell or tissue
phagocytosis
express surface receptors that bind Ig - Fc repectors → triggers phagocytosis
**only EXTRACELLULAR pathogens
the process of antibody production
primary response
the rxns of the immune system when it contacts an antigen for the very first time
5-7days (IgM) to 14 days (IgG/A/E)
low number of mainly IgM → IgGAE
secondary response
the run of the immune system when it contacts an antigen for the second or subsequent times
1-3 days of high IgGAE
IgM and IgG lab differential
IgM vs. IgG
its got me vs its gone
acute infection (many symptoms) vs. previous infection (almost gone)
IgM resonds first, and then IgG secondarily
this helps us understand the level of infection for certain patients
IgM in ABO blood compatibility
IgM works as an anti-A and anti-B antibody
types A, B, and O all have antibodies (anti-…) without having prior contact
AB don’t have any antigens so are the universal recipient
O has both antigens, so are the universal donor
what is cell-mediated immunity
deals with INTRAcellular bacteria, viruses, and any bacteria that are out of reach of antibodies and have escaped phagocytosis
T cells arise in the bone marrow → migrate to the thymus to undergo several steps of maturation
TCR (T cell receptors), CD4, and CD8 co-receptors
destroys invading microorganisms and helps B cells produce antibodies
two types:
helper T cells (CD4+)
cytotoxic T cells (CD8+)
what are the main defences in T cell immunity
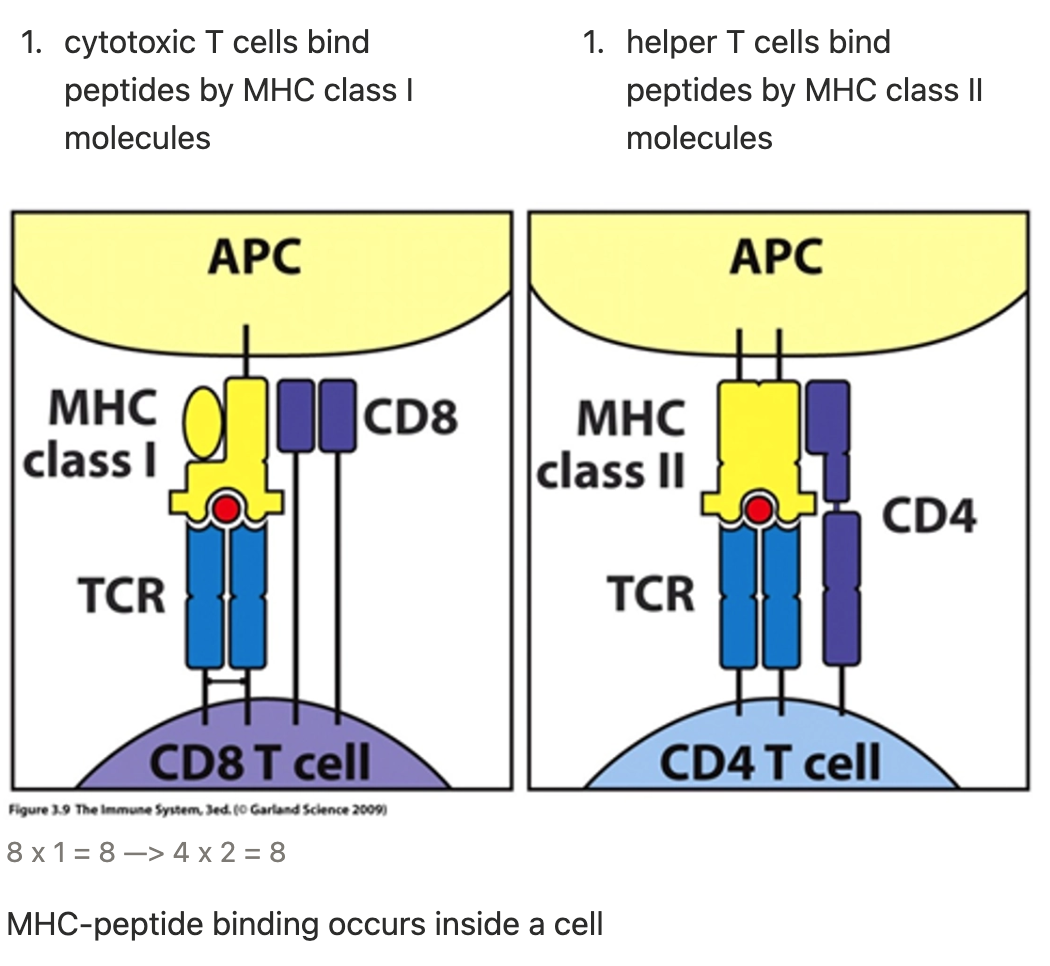
helper T cells (CD4+) - MHC2
cytotoxic T cells (CD8+) - MHC1
MHC class 1 and class 2 - proteins that play a pivotal role in the adaptive branch of the immune system
both share the task of presenting peptides on the antigen cell surface for T cells to recognize
T cell receptors can only bind to protein antigens (peptides) - all microbes present these peptides
for a T cell receptor to be activated it must be presented with a peptide by an MHC
each T cell expresses a variable region and antigen binding site, specific for one unique peptide or antigen
2 classes of MHC molecules
the process of T cell activation
APC = antigen presenting cells
prior to activation, T cells are known as naive T cells
activation of T cells begins with recognition and binding of that T cell receptor to the MHC antigen complex (costimulation)
proliferation and differentiation requires interleukin 2 (IL2) which is produced by naive T cells and send signals for the T cells to duplicate
differentiation into either effector or memory cells
how do helper T cells work
CD4+
remember: bind with MHC2
the “alarm bell” of the immune system
once activated, Th cells release cytokine
this stimulates other parts of the immune system to react
NK cells, macrophages, cytotoxic cells to proliferate, B cell proliferation
how do cytotoxic T cells work
CD8+
remember: bind with MHC1
once binding occurs, proliferation into effector and memory cells
the effector cytotoxic T cells release toxic enzymes that degrade nucleic acids in the cells
latching onto the abnormal cell to release molecules, forming pores and forcing apoptosis
very similar to NK cells!!!!!
NOTE: some viruses block the presentation of MHC1 molecules to evade cytotoxic T cell response
ex. herpes and HPV
what is a hypersensitivity rxn
refers to sensitization of the immune system by repeated exposure to an allergen (when the immune system goes wrong)
hypersensitivity disorders refer to excessive or inappropriate activation of the immune system
4 types of hypersensitivites
type I
IgE-mediated response
within 1 hr
ex. anaphylaxis
type II
antibody-mediated response
specifically IgG and IgM cytotoxic
hours-days
ex. hemolytic anemia or blood incompatibility
type III
complement-mediated immune responses
1-3 wks
ex. lupus or glomer nephro
type IV
T-cell-mediated responses
days-weeks
metal sensitivity or TB tests
atopic conditions
atopy refers to the genetic tendency to develop allergic diseases such as allergic rhinitis (hay fever), atopic asthma, and atopic dermatitis (eczema)
most common with IgE responses
people with one atopic condition are more likely to develop another atopic condition
type 1 hypersensitivity rxns
begin rapidly, often within minutes of an antigen challenge through inhalation, ingestion, injection, or skin contact
allergen is met by phagocytic cell that ingests it, chews it up, and spits it out
an APC then creates a cascade where B cells are created → secreting IgE
IgE then binds to mast cells (called first exposure/sensitization)
re-exposure to the same allergen = memory response occurs immediately
allergen-IgE binding activates the mast cell to degranulate
releases histamine and cytokines → atopic symptoms
allergen route of entry rxns
inhale
resp tract
mucus buildup, sneezing, nasal congestion, bronchial smooth muscle constriction
asthma/rhnitis
inject
skin & bloodstream
local and systemic inflammation
hives or anaphylaxis
ingest
GI tract
intestinal smooth muscle constriction, systemic inflammation
NVD or anaphylaxis
what is anaphylaxis
provoked by any allergen that enters the bloodstream - injected directly into blood or rapid absorption across the epithelial surface of the skin/gut
a severe, potentially life-threatening allergic reaction requiring immediate attention characterized by widespread edema, vascular shock secondary to vasodilation, and dyspnea
level of severity depends on level of previous sensitization
anaphylactic shock
anaphylactic shock: the catastrophic loss of blood pressure due to extensive fluid loss from blood vessels
3 ways to diagnose allergies
serum bloodwork to reveal increased IgE levels
biopsies during endoscopies and colonoscopies (for GI intolerances)
skin prick/patch tests
local inflammation at injection site with 10-15 mins
allergy desensitization immunotherapy
aka allergy shots
principle: divert immune response from IgE to IgG
a form of long-term treatment that decreases symptoms for those with rhinitis, asthma, pink eye, or stinging insect allergies
works like a vaccine - gradually increase dose to develop immunity
risks: local rxns, systemic rxns, anaphylaxis (within 30 mins usually)
what are type ii hypersensitivities
mediated by IgG or IgM antibodies directed against target antigens on cell surfaces
can be endogenous on cell membranes or exogenous absorbed on membrane surface
the rxn destroys the antigen in 3 different ways
3 steps of type ii hypersensitivity
opsonization and phagocytosis
cells are coated (opsonized) with molecules that make them attractive to phagocytes
antibody dependent cellular cytotoxicity
IgG binds to surface antigen on infected cell and NK cell kills the infected cell
complement activation
the binding of a IgG can activate the complement system and cause lysis of the antigen by membrane attack complex or by phagocytosis
complement activation recruits neutrophils and monocytes which release specific enzymes → inflammation and tissue damage
Rh incompatibility
example of type ii hypersensitvity
a condition that occurs during pregnancy if a woman has Rh- blood and her baby has Rh+
Rh factor is a protein on RBCs that is inherited
the baby’s blood can pass through to the mother through the bloodstream/placenta and mom’s body will react and begin producing antibodies against baby → hemolytic anemia
type iii hypersensitivities
mediated by the formation of insoluble antigen-antibody complexes which activate the complement system and localized edema
the immune complexes formed damage vessel linings as they are delivered and deposited into desired tissues
once deposited, inflammatory response is activated→ complement system → neutrophils recruited → inflammation
the neutrophils try to damage complexes, but destroy target tissues with them
responsible for vasculitis
seen in AI disorders like lupus or acute glomerulonephritis/kidney damage (due to post-strep infections)
post-strep glomerulonephritis (PSGN)
example of type iii hypersensitivity
characterized by rapid deterioration of kidney function due to inflammatory response (type III rxn) following a strep infection
affects the glomeruli and small blood vessels of the kidneys
most frequently in children 1-2 wks after a sore throat or 6wks after a skin infection
the body responds to nephrogenic streptococcal infection by forming immune complexes containing the strep antigen with human antibody → complement pathway
complement pathway causes infiltration of leukocytes and proliferation of other cells in glomerulus → reduction in GFR and perfusion → renal failure, acid-base imbalance, electrolyte abnormalities, volume overload, edema, and HTN
PSGN triad: hematuria, edema, and HTN
type iv hypersensitivities
aka delated-type hypersensitivity (DTH)
cell-mediated NOT antibody-mediated
immune rxn in which T cells are activated by antigens
there must be a previous exposure to mount an immune response
subsequent exposures on SQ tissue = rash
local APC deem these cells normal, but T effector cells are activated and release cytokines → phagocytes, fluid, protein are recruited to the site → physical lesion
ex. TB test, poison ivy, nickel, latex
immune self tolerance
the immune system being able to differentiate foreign antigens from self-antigens
results from central and peripheral mechanisms that delete self-reactive immune cells that cause autoimmunity
two types: central and peripheral tolerance
immune self-tolerance: central
derived from the deletion/inactivation of self-reactive T cells (eliminated in thymus) and B cells (eliminated in bone marrow)
immune self tolerance: periphipheral + what is anergy
maintenance of tolerance in secondary lymphoid tissue (spleen or lymph nodes)
these processes work through deletion/apoptosis of autoreactive cells or anergy that escaped central tolerance
anergy = a state where cells become unresponsive to antigen stimulation and suppression of effector T cell activation
what is autoimmunity
results from a loss of self-tolerance
reason is unknown, however gender and genetics play a role, as well as self-reactive lymphocytes (HLA)
more women have AI disorders (5:1)
classification of autoimmunity
**a spectrum
organ-specific AI diseases
AI response targets an antigen present in a specific organ
ex. DM1 (pancreas-beta cells), Graves Disease (thyroid-TSH receptors), MS (neurons/brain)
systemic AI diseases
AI response targets an antigen present at many different sites (can involve multiple organs)
ex. Systemic Lupus Erythematosis (many cell types)
what is graves disease
organ specific autoimmune disorder
immune system attacks thyroid → overactive thyroid/increased T3 and T4
immune system makes thyroid-stimulating immuniglobulin (TSI) that attaches to thyroid cells - acts like TSH, but makes the thyroid produce even more TSH and TSI
DM1
organ-specific autoimmune disorder resulting from a chronic AI destruction of the beta cells
autoreactive Cytotoxic T Lymphocytes (T cells) kill Islets of Langerhans → reduced insulin production → hyperglycemia + unk. inflammatory response
MS
organ-specific autoimmune disorder
immune system attacks myelin sheath that covers nerve fibers → communication problems between brain and rest of body
inflammatory process that causes T cells and their mediators triggering injury of axons and their myelin sheath
the T cell-mediated inflammation is driven by the AI process, which triggers a degenerative phase that is immune dependent
5 main treatments for autoimmune disorders
immunosuppression
plasmapheresis (remove antibodies) - temp
block MHC with similar peptide (mimic)
use antibodies that block B or T cells
disease-specific treatments (ex. insulin or joint replacements for RA)
based on effector mechanism, bodily tissues/organs involved, magnitude, and chronicity of effector processes
treatment should target underlying mechanisms
4 key goals of immunization
prevent
control
eliminate
eradicate
by directly protecting vaccine recipients and indirectly protecting vulnerable populations
what is vaccination + purpose
the intentional exposure to pathogens in a form that cannot cause an infectious disease
purpose: for the recipient to develop long-term immune protection against the pathogen
key time stamps of vaccination
1774 - Ben Jesty
innoculated family with pus from cowpox clister of a cow to protect from smallpox
1796 - Ed Jenner
demonstrated immunity to smallpox by innoculating an 8yr old with cowpox virus
1798
smallpox vaccine was developed - the first vaccine!
1870 - Louis Pasteur
created vaccines against chicken cholera, anthrax, rabies
principle: isolate, inactivate using heat and inject
the UK MMR controversy
Dr. Andrew Wakefield claims the MMR vaccine causes autism
“no causal link was established between MMR vaccine and autism as the data was insufficient”
Wakefield had been funded by lawyers who had been sued by vaccine manufactures - study was fraudulent in favour of his lawful argument
faking data
herd immunity
parents excuse as to why they will not vaccinate their child
protects susceptible individuals by stopping transmission
the risk of infection is reduced when the number of individuals who can spread that pathogen is reduced
**herd immunity is only effective when a sufficient number of people are immune
can vaccines cause disease (live and inactivated)
NEVER
inactivated vaccines
killed viruses or bacteria
cannot cause disease
live vaccines
may cause mild symptoms, but full blown-disease is extremely unlikely
why do children get multiple vaccine doses
inactivated vaccines
build immunity in phases
multiple doses
live vaccines
one dose - sufficient immunity
not all children respond to the first
common rxns to vaccines
minor rxns
redness/pain at injection site
fever
prolonged crying
vomiting
headache
moderate rxns
febrile seizures
severe rxns (very rare)
anaphylaxis
encephalitis/encephalopathy from measles vaccine
what are in vaccines
the live or non-live bacteria (or parts)
adjuvants
help boost immune response
*multi-dose vials contain preservatives
may include thimerosal, formaldehyde, aluminum
what is vaccine hesitancy
a delay in acceptance or refusal of vaccination despite availability of vaccine services
the WHO declared vaccine hesitancy as one of the top 10 threats to global health in 2019
9 reasons why people are vaccine hesitant
mistrust in government and the medical system
not believing that vaccine are safe
too many vaccine
philosophical or religious belief
thinking that alternate healthcare can replace vaccines
vaccine myths are prevalent
it is hard to understand vaccine information
worried about vaccine side effects
diseases that vaccines prevent aren’t a serious threat to their health
5 Cs of vaccine hesitancy
complacency
convenience
confidence
calculation
collective responsibility
what is immunization
immunization: the process by which a person becomes protected against a disease through exposure to immunizing agents
what is immunoprophylaxis
prevention of disease using immunizing agents
agents are classified as active or passive immunization
passive immunity
involves the natural transfer of pre-formed antibodies from one person to another (or from an animal product) to provide immediate, temporary protection from infection, or reduce severity of illness
most commonly can occur by transplacental transfer
an infant receives antibodies from the mother through the placenta in the last 1-2 months of pregnancy
the newborn will have the same antibodies as the mother for up to a year
artificial/systemic admin of immunoglobulins can also provide protection when vaccines aren’t available or exposure has already occurred
active immunity
the stimulation of immune system to produce antibody and cellular immunity
lasts for many years-lifetime
can occur through getting infected or getting vaccinated
the memory B cells remember the disease and already have the antibodies to protect to the best of their ability (immunologic memory)
vaccines give the same immunity without the individual having the infection + also provide immunologic memory
how do vaccines work
admin of a vaccine antigen triggers an inflammatory rxn that is initially mediated by thew innate immune system, then works its way to the adaptive immune system through B and T cells
most vaccines provide humoral immunity (through B cells), most live vaccines act through cell-mediated immunity (T cells) - or both
long-term immunity requires the persistent of antibodies, or creation + maintenance of antigen-specific memory cells (priming) that will produce an immune response when needed
guiding rule of vaccines
the more similar a vaccine is to the disease-causing form of the organisms, the more effective the vaccine
classification of vaccines
live attenuated
viral
bacterial
inactivated
whole
viruses, bacteria
fractional
protein-based (toxoid, subunit)
polysaccharide-based (pure, conjugate)
live attenuated vaccines
contain whole, weakened bacteria or viruses
because the vaccine is so similar to the agent, this vaccine will last longer and be more effective than non-live vaccines
to produce an immune response, the live vaccines must replicate/grow in the body
underattenuated → causes disease
overattentuated → does not provoke immune response
usually one dose, rarely 2 doses
require careful storage and handling to avoid inadvertent inactivation (heat, light, etc)
contraindicated for immunocompromised
examples of live vaccines
viral: measles, mumps, rubella, vaccinia, varicella, zoster, yellow fever, rotavirus, intranasal influenza, oral polio
bacterial: BCG, oral typhoid , rotavirus
inactivated (non-live) vaccines
contain the whole inactivated (killed) bacteria or virus, their parts/products secreted that are modified to remove their toxoids (what makes them infectious) with heat or light
cannot replicate or cause disease (even in immunocompromised)
require adjuvants + primary series and booster doses
the addition of adjuvants enhances the immune response and extends the B and T cell activation
subunit vaccines include toxoid, recombinant, polysaccharide, and conjugate vaccines
these use pieces of the germ like its protein, sugar, or capsid (casing around the germ) to provide immunity
polysaccharide vaccines - pure
unique type of inactivated subunit vaccine composed of long sugar molecule chains that make up the casing of certain bacteria - making the vaccine more potent
available for pneumococcal, meningococcal, and Salmonella Typhi
usually T-cell independent - can stimulate B cells without Th cells
should not be given to children under 2
do not require booster shots due to predominantly IgM being produced, rather than IgG
this is why conjugate vaccines were made
polysaccharide vaccines - conjugate
pure polysaccharides are chemically combined with a protein molecule
conjugation changes the immune response from T-cell independent to T-cell dependent
this allows children under 2 to receive this vaccine + requires booster shots now
pneumococcal, meningococcal, and Hib
toxoid vaccines
use a protein-based toxin to create immunity strictly to the toxin in the germ - rather than the entire germ
the toxoid is absorbed to the Al or Ca salts
toxoid is harmless, cannot cause disease
do not actively multiply or spread + less susceptible to damages by light or heat
usually require several doses
ex. TDAP
nucleic acid-based vaccines
mRNA vaccines contain material from the virus that causes disease that gives our cells instructions for how to make a harmless protein that is unique to the virus
after our cells make copies of this protein, they destroy the genetic material from the vaccine
our bodies recognize that the protein should not be there and build T cells and B cells that will remember how to fight the virus if we are infected in the future.
4 main challenges with vaccines
not effective in immunocompromised ppl
high costs of vaccine development
inadequate access to vaccines
constant disease evolution will cause constant updating of vaccines
hx of mRNA vaccines
1960 mRNA discovered
1974 liposomes used for drug delivery
1978 first liposome mRNA delivery to cell
1993 first mRNA vaccine tested (influenza in mice)
2005 discovery of modified RNA that evades immune detection
2013 first clinical trial of mRNA vaccine (Rabies)
2018 first drug with lipid nanoparticles approved
2020 mRNA based COVID vaccines
how mRNA vaccines work
1⃣ What the Vaccine Contains
mRNA that codes for the spike protein of SARS-CoV-2
Lipid nanoparticle system to:
Protect the mRNA
Help it enter cells
2⃣ What Happens After Injection
Lipid nanoparticles help mRNA enter dendritic cells
mRNA stays in the cytoplasm (does NOT enter nucleus)
Ribosomes read the mRNA
Cells produce the spike protein
3⃣ Immune System Activation
Spike protein is displayed on the dendritic cell surface
Dendritic cell travels to nearby lymph node
In lymph node:
Helper T cells activate B cells
B cells produce large amounts of antibodies
Cytotoxic T cells learn to kill infected cells
pros of mRNA vaccines
easier and safer to produce than vaccines that require a weakened or inactivated pathogen
mRNA can be easily altered for different protein spikes/common viral mutations
cons of mRNA vaccines
mRNA can be a challenge to deliver to cells as the innate immune system can identify and destroy mRNA, hence the use of lipid nanoparticles to protect the mRNA
these lipid nanoparticles require uninterrupted refrigeration/freezing and are easily damaged.
stringent protocols are required for transportation and vaccine preparation for delivery
bacterial suface structures - capsule
only in some bacteria
extra outer covering
protects against phagocytosis
helps retain moisture
aids in adhesion to surfaces and nutrients
bacterial suface structures - cell wall
rigid outer layer
gives cell its shape
protects from the envirpnment
prevents dehydration
controls permeability
provides attachment site for flagella and pili
bacterial suface structures - plasma membrane
thin layer beneath the cell wall
semi-permeable
controls movement of substances in and out of the cell
bacterial internal structures - cytoplasm
gel-like substance (mostly water)
contains enzymes, salts, organic molecules, and cell components
house ribosomes and nucleoid
bacterial internal structures - ribosomes & nucleoid
ribosomes
responsible for protein synthesis
nucleoid
region containing the single circular bacterial DNA molecule (plasmids)