Atomic Structure
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
What are the Historical Ideas of the Atom?
1661 - Robert Boyle proposed that there were some substances that could not be made simpler (Elements)
1803 - John Dalton suggested that elements were composed of indivisible atoms and that atoms of the same element had the same masses and they could not be broken down.
1896 - Henri Becquerel discovered Radioactivity which showed particles could come from inside the atom.
1897 - J J Thomson discovered the electron and proposed the “plum pudding” model.
1911 - Ernest Rutherford and his team found that most of the mass and positive charge was in a tiny central nucleus
1913 - Niels Bohr introduced the idea that the atom consisted of a tiny positive nucleus orbited by electrons in shells which was later used for quantum theory.
1926 - Erwin Schrödinger worked out an equation that used the idea that electrons had some of the properties of waves as well as those particles. This was called quantum mechanics which can be used to predict the behaviour of sub-atomic particles.
1932 - James Chadwick discovered the neutron.
What are the relative masses and charges of the Sub-Atomic Particles?
Electron - M:1/1840 - C:-1
Proton - M:1 - C:+1
Neutron - M:1 - C:0
Protons and Neutrons are often referred as nucleons and are held together in the central atom by the strong nuclear force and is stronger than intermolecular forces so it overcomes the repulsion between protons.
What is the Atomic Number, Mass Number and Isotopes?
Atomic Number (Z) = Number of Protons
Mass Number (A) = Number of Protons + Neutrons
Isotopes - Atoms with the same number of protons and a different number of neutrons. They react chemically in the same way.
What is Relative Atomic Mass/Relative Molecular Mass?
The mean mass of an atom of an element divided by 1/12 the mass of one atom of carbon-12
Ar = Average mass of 1 atom / 1/12 mass of 1 atom of 12C
Mr = Average mass of molecule / 1/12 mass of 1 atom of 12C
What is Carbon Dating?
A method used to determine the age of carbon-based materials using the radioactive isotope carbon-14 which has a half life of 5730 years. This is used to date materials up to 60,000 years old (Which would only contain 0.07% carbon-14 now) but is best for around 2000 years.
What is Mass Spectrometry?
A powerful instrumental method of analysis that is used to: find the abundance and mass of each isotope in an element allowing us to determine its relative atomic mass, find the relative molecular mass of substances made of molecules.
What is Time of Flight (ToF) Mass Spectrometry?
A common form of mass spectrometry where particles of the substance are ionised and accelerated, where their time taken to travel a fixed distance is used to find the mass.
What is the Stage 1 of ToF?
Stage 1 - Ionisation
The sample is ionised using methods like electron impact/ionisation and electrospray ionisation.
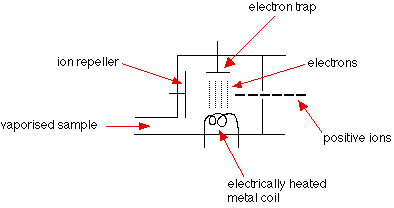
What is Electron Impact/Ionisation?
The sample being analysed is vaporised and then high energy electrons are fired at it. The high energy electrons come from an electron gun (A hot wire filament with a current running through it). This usually knocks off one electron from each particle forming a X+ (Molecular ion)
It is used for elements with low formula mass (Organic or Inorganic molecules). The molecular ion often breaks down into smaller fragments which are also detected in the mass spectrum.
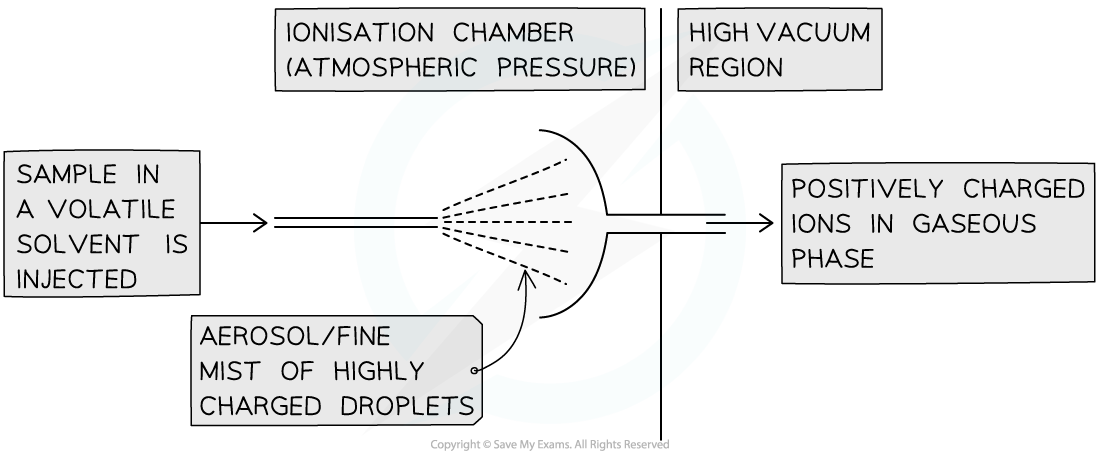
What is Electrospray Ionisation?
The sample is dissolved in a volatile solvent (eg water/methanol) and injected through a fine needle into the chamber at a high voltage. The particles are ionised by gaining a proton from the solvent as they leave the needle forming XH+ ions. The solvent evaporates away while the XH+ ions are attracted towards a negative plate where they are accelerated.
This technique is used for substances with higher molecular mass including biological molecules such as proteins. This is known as soft ionisation as fragmentation rarely occurs.
What is Stage 2 of ToF?
Stage 2 - Acceleration
The positive ions are accelerated using an electric field so that they all have the same kinetic energy.
KE = ½ M V²
Therefore, the velocity is: V = Root (2KE/m)
Given that all the particles have the same kinetic energy, the velocity of each particle depends on its mass. Lighter particles have a faster velocity and heavier particles have a slower velocity.
What is Stage 3 of ToF?
Stage 3 - Flight Tube
The positive ions travel through a hole in the negatively charged plate into a tube. The time of flight of each particle through this flight tube depends on its velocity which in turn depends on its mass.
The time of flight along the flight tube is given by the equations:
t = d/v where d = Length of tube
Therefore t = d root (m/2KE)
What is Stage 4 of ToF?
Stage 4 - Detection
The positive ions hit a negatively charged electric plate. When they hit the detector plate, the positive ions gain an electron from the plate. This creates a current in the detector. The size of the current is proportional to the abundance of the ions.
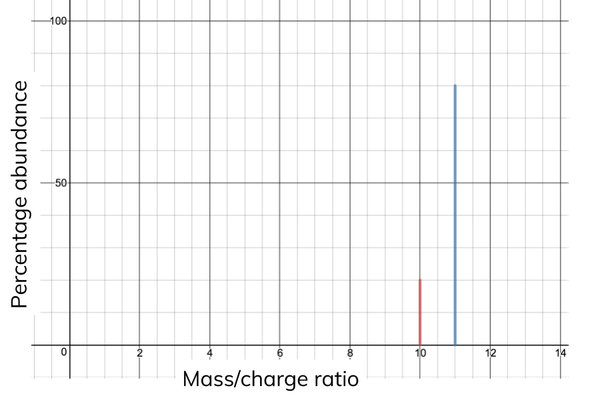
What is m/z and how is it used in Mass Spectra?
It is Mass to Charge ratio.
It is plotted against the relative abundance of ions in mass spectrometry graphs to find out the relative atomic mass.
The mass spectra can also be used to identify the different isotopes that make up an element. It can measure relative atomic masses to five decimal places (High resolution) but most work is done to 1 decimal place (Low resolution).
Sometimes more than one electron could have been removed meaning that it will have half the m/z value due to the extra charge, even though it may have a very similar mass.
What is the electronic arrangement of atoms?
Each shell can hold an increasing amount of electrons, following the rule 2n² where n is the shell number, resulting in the arrangement 2,8,18,32.
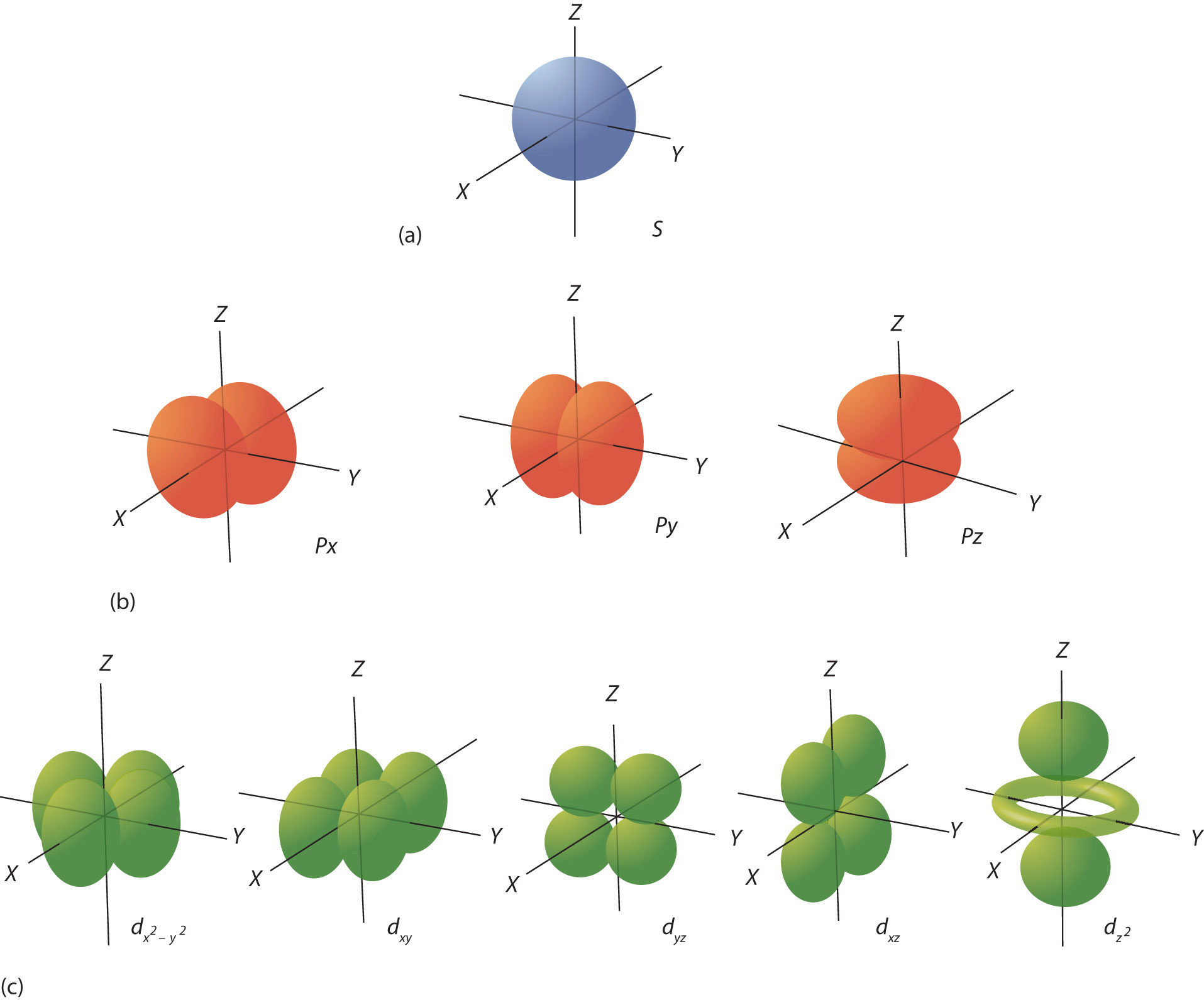
What are the four different orbitals?
The four different orbitals are s, p, d, and f. Each type of orbital has a distinct shape and energy level, influencing the distribution of electrons in an atom.
Each orbital has has an electron density where there is approximately a 95% chance to find an electron in that region.
S shell - 1 Orbital (2 Electrons)
P shell - 3 Orbitals (6 Electrons)
D shell - 5 Orbitals (10 Electrons)
F shell - 7 Orbitals (14 Electrons)
The different orbitals are in the image where you need to know the approximate shapes of s, p and d.
What are the energy levels like for Atomic Orbitals and What is Spin?
Energy levels increase with distance from the nucleus and is shown in the image where orbitals like 3d have more energy than 4s.
Spin is an electron property where two electrons in the same orbitals have opposite spins and is represented using arrows pointing in different directions (They don’t actually spin). By having two opposite spins it makes the atom more stable.
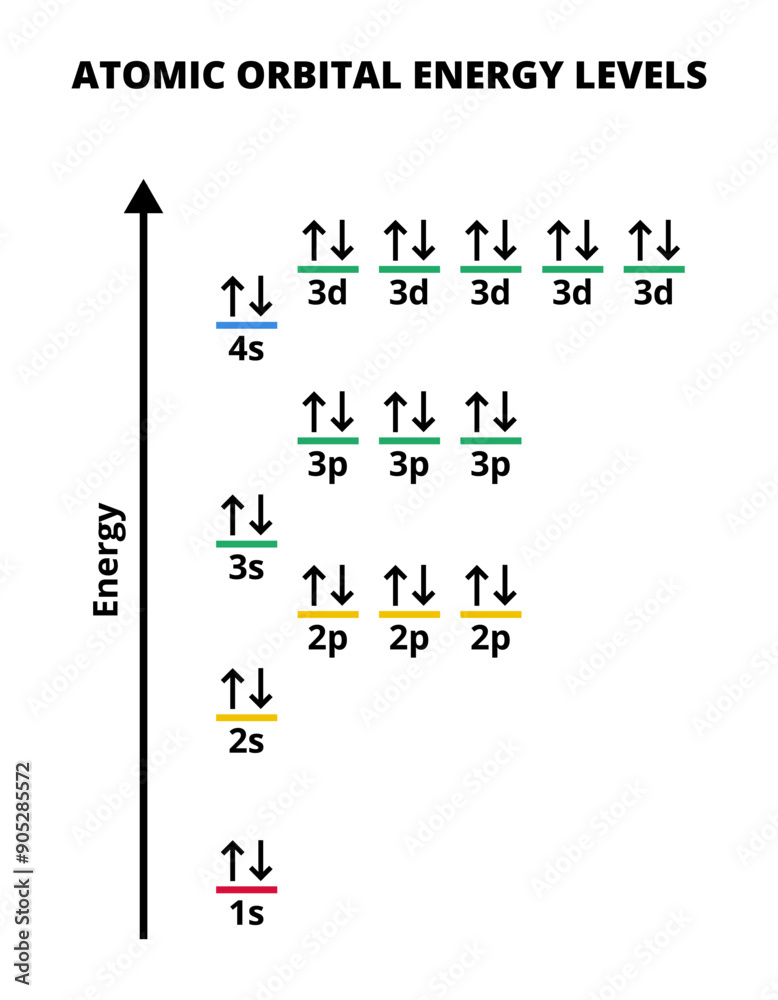
What are the rules for allocating electrons to Atomic Orbitals?
Atomic Orbitals of lower energy are filled first due to the strong electrostatic attraction from the nucleus, resulting in lower energy levels filling first.
Atomic Orbitals of the same sub-level fill singly before pairing starts due to electrons repelling each other.
No Atomic Orbitals can hold more than two electrons.
What is the Electron Arrangement of the following elements: Oxygen, Aluminium, Calcium and Vanadium?
Oxygen -1s² 2s² 2p⁴ , [He] 2s2 2p4
Aluminium - 1s² 2s² 2p⁶ 3s² 3p¹ , [Ne] 3s2 3p1
Calcium - 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² , [Ar] 4s2
Vanadium - 1s² 2s² 2p⁶ 3s² 3p⁶ 3d³ 4s² , [Ar] 3d3 4s2
What is Ionisation Energy?
The energy required to remove a mole of electrons from a mole of atoms in the gaseous state to form one mole of gaseous ions, measured in kJ mol-1. (IE)
The first ionisation energy would be to form one mole of gaseous 1+ ions and the second ionisation energy would for one mole of gaseous 2+ ions.
Successive Ionisation Energies of Sodium:
Na(g) → Na+(g) + e- First IE
Na+(g) → Na2+(g) + e- Second IE
Na2+(g) → Na3+(g) + e- Third IE
And so on.
What are the Ionisation Energy trends across a Period?
The trends of ionisation energy across a period generally increases due to increasing nuclear charge and decreasing atomic radius, resulting in stronger attraction between electrons and the nucleus.
However, there may be exceptions caused by electron-electron repulsion in filled or half-filled sub-shells, which can lower the ionisation energy. This leads to a decrease in ionisation energy between two elements across a period.
This is evidence that supports the atomic orbital theory
What are the Ionisation Energy Trends down a Group?
Ionisation energy generally decreases down a group due to increased atomic size and shielding effect. This leads to a weaker attraction between the nucleus and the outermost electrons, making them easier to remove.