Lecture 9 - Minerals & Trace Elements
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
What are the major minerals needed for our bodies?
*All form solid crystals
Na
Mg
P
S
Cl
K
Ca
What is the importance of minerals in the body?
Elements needed by the body in small amounts for health and maintenance
Major elements are needed in the diet in amounts greater than 100mg/day or are present in the body in amounts greater than 0.01%
Trace minerals are available in unprocessed foods, some processed foods, and dietary supplements
Not in pure form, unless in supplement
Bioavailability is very important
Upper limits have been established because certain minerals can be toxic if taken in too high an amount
Iron
How do minerals act as cofactors for particular enzymes?
The mineral cofactor combines with the incomplete enzyme to form the active enzyme
The active enzyme binds to the molecules involved in the chemical reaction and accelerates their transformation into the final product
The final products are released, while the enzyme remains unchanged
What is calcium as a mineral?
The most abundant mineral in the body
Accounts for 1-2% of adult body weight
99% of calcium in the body is found in bones and teeth
1% found in fluids
Nerve transmission, muscle contractions, blood pressure regulation, and hormone release
What is the calcium absorption pathway?
Calcium bioavailability decreases in the presence of tannins, fibre, phytates, and oxalates
In the small intestine, Ca can bind & inhibit; in the large intestine, it binds to bacteria, breaking down SCFAs
Ca is reactive to fibre, allowing binding
Levels of Ca in the blood are tightly regulated by parathyroid hormone and calcitonin
Vitamin D turns on the synthesis of calcium transport proteins
Calcium transport proteins shuttle calcium across the mucosal cell
A calcium pump that requires energy moves calcium from the mucosal cells to the bloodstream
What is the connection between Ca and inulin/acid
Increased Ca absorption by inulin/acid
Inulin (a prebiotic fibre promoting colonic bacterial growth) and acid (specifically short-chain fatty acids produced by fermentation) enhance absorption in the large intestine
Inulin increases the solubility and bioavailability of Ca, boosting bone health and density
What is the pathway of regulation of blood calcium levels?
Low blood Ca →
Parathyroid glands →
Parathyroid hormones →
Stimulates calcium release from bone →
Normal blood calcium
Ca reabsorbed by the kidney →
Normal blood calcium
Activates vitamin D →
Increases intestinal Ca absorption →
Normal blood calcium
High blood Ca →
Thyroid gland →
Calcitonin (osteoblast) →
Inhibits calcium release from bone →
Normal blood calcium
What is the pathway of bone remodelling?
Activation: Osteocytes detect damage and send signals to the bone surface, causing precursor cells to differentiate into active, mature osteoclasts.
Resorption: Osteoclasts attach to the bone matrix, creating a pit that degrades the inorganic and organic components, releasing calcium into the bloodstream.
Reversal: Osteoclasts undergo apoptosis. Mononuclear cells clean up the resorption site and signal osteoblasts.
Formation: Osteoblasts move into the resorption pit and deposit new, unmineralized matrix called osteoid.
Termination/Mineralization: The osteoid is mineralized with calcium and phosphate, becoming hard, new bone, lining the surface.
What is the calcium content of foods?
In a range of foods
Highest in milk and alternative products and fish with bones
Soft drink consumption is affecting the calcium intake of teenagers by replacing milk
Low calcium intake early in life increases the risk of osteoporosis and increases the number of overweight and obese individuals
What connection is there between sex and bone mass?
Bone mass in men and women is the same until puberty
Men achieve a higher peak and overall have higher bone density
After age 35, men and women lose bone
Women lose a lot during menopause since estrogen increased bioavailability & absorption of Ca
What is T score?
A numerical value of bone density
Osteoporosis < -2.5
Osteopenia < 1
Osteoporosis causes a hump in elderly people since the front edge of the vertebrae collapses
A broken hip is highly common in the elderly, causing a rapid decline
What is osteoporosis?
Major public health problem in Canada
2 million Canadians over 50 have osteoporosis, twice as many women as men
Estrogen increases calcium absorption
Osteoporosis is responsible for 80% of fractures in people over 60
The form of Ca is supplements is important
Calcium carbonate should be taken with a meal, Ca citrate can be taken any time
Antacids that contain aluminum and Mg may actually increase Ca loss
Some are just Ca carbonate, so it is okay
What are the risk factors for osteoporosis?
Gender
Age
Race
Genetics
Body Size
Smoking
Exercise
Alcohol abuse
Diet
What is phosphorus?
Makes up ~1% of an adult’s body
85% is found in the bones and teeth
More readily absorbed than Ca
In the extracellular environment, primarily in inorganic forms
HPO2-, H2PO4-
In the intracellular environment, primarily in organic molecules
ATP/ADP
DNA/RNA
Phospholipids
Phosphate sugars
Although not common because it can be efficiently absorbed, deficiency can lead to bone loss, weakness, and loss of appetite
What is magnesium?
About 50-60% of Mg in the body is found in bone, where it is essential for the maintenance of structure
Most remaining Mg is present inside cells, where it is the second most abundant intracellular ion after K
Cofactor for over 300 enzymes → necessary for generating energy from carbs, lipids and proteins
Required for Na+/K+ ATPase
Variable Mg2+ reabsorption in nephron → homeostasis
Deficiency is rare but can occur with alcoholism, malnutrition, Kidney and GI disease
How is Mg absorbed?
Paracellular
Through a tight junction driven by electrochemical gradients
Transcellular
Carrier-mediated mechanism becomes active to transport magnesium through the intestinal cells
Mg2+ stabilizes the negative charge on phosphate-containing molecules
What is sulfur?
Dietary sulphur is found in protein foods and sulphur-containing amino acids in vitamins (B1 & B7)
Sulphur-containing amino acids (methionine, cysteine) needed for protein synthesis
Found in nonfood additives
Preservative
Typically considered bad because it is in “unhealthy” foods
No recommended daily intake for sulphur
No known deficiencies
How is iron in the body?
Essential for the delivery of oxygen to cells
2 oxygen-containing proteins, hemoglobin and myoglobin, contain iron
Most of the iron in the body is part of hemoglobin
What is iron?
Hemoglobin: iron-containing component of the blood
Iron from animal products is heme iron and from plants is nonheme iron
Heme iron is more efficiently absorbed than nonheme iron
Bioavailability
Ferritin is the major iron storage protein
Intracellular
Transferrin is an iron transport protein in the blood
Hemosiderin is an insoluble iron storage compound produced by the body when iron exceeds the storage capacity of ferritin
What is the pathway of heme iron absorption?
Heme iron is absorbed as part of the heme group; nonheme is absorbed in the ferrous form (Fe2+)
Once inside the mucosal cells, some iron may be bound to ferritin for storage
When mucosal cells die, iron that remains bound to ferritin is excreted in the feces
Iron that enters the blood is converted to ferric iron (Fe3+) by a copper-containing protein in the cell membrane. The Fe3+ binds to transferrin for transport
Transferring transports iron to the liver, bone, and other body cells,
When red blood cells die, they are broken down by cells in the liver, spleen, or bone marrow and the iron is released for reuse
Excess iron is stored primarily in the liver, bound to ferritin
Most iron loss is due to blood loss
What are the DRI for iron?
Low in 0-6 months because in the third trimester, babies steal iron from mom and store it
Females have an increased DRI than males due to menstruation
If taking the pill need less since it lessens blood loss
Vegetarians need higher amounts due to the bioavailability of plant sources
What is iron deficiency?
When there is insufficient hemoglobin, red blood cells are microcytic and hypochromic and unable to deliver sufficient oxygen to the tissues
~80% of the world’s population may be iron-deficient, and 30% suffer from iron deficiency anemia
If hematocrit, hemoglobin, serum iron, or ferritin levels are low, one should be conscious of iron intake
What are the stages of iron deficiceny?
Adequate Iron Status
Normal iron storage
Normal iron in plasma
Normal iron in RBCs
Low Iron Storage
Low iron storage
Normal iron in plasma
Normal iron in RBCs
Depleted Iron Storage
No iron storage
Normal iron in plasma
Normal iron in RBCs
Low Levels of Circulating Iron
No iron storage
Low iron in plasma
Normal iron in RBCs
Iron Deficiency Anemia
No iron storage
Very low iron in plasma
Medium iron in RBCs
What is iron toxicity?
Hemochromatosis → iron deposits in liver, skin, etc.
Primary
Some genetic mutation influencing the likelihood
Secondary
Too much iron in the diet or other dietary factors
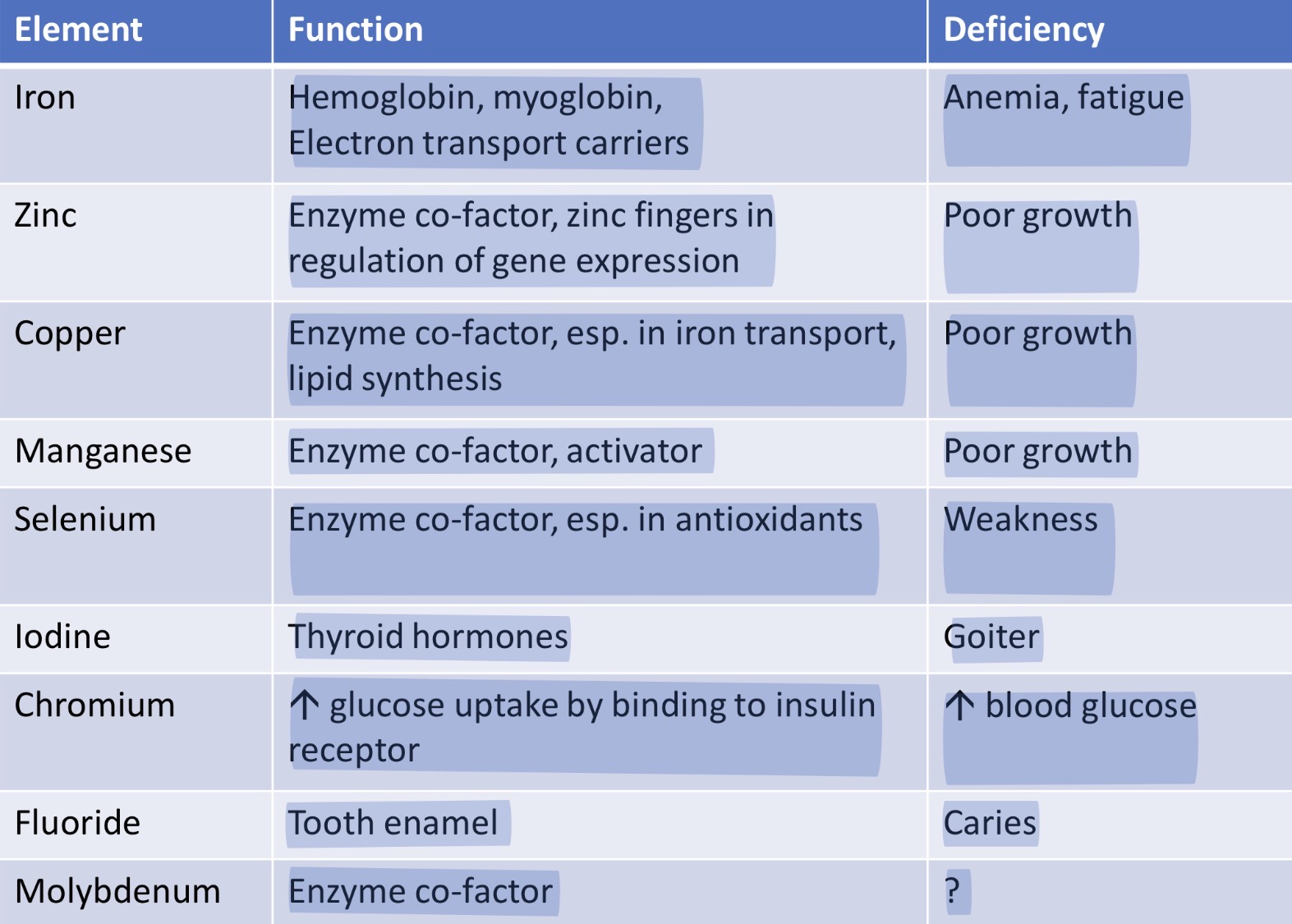
What is zinc?
Most abundant intracellular trace element
Essential in the diet for growth and development
Better absorbed from animal sources than plant sources
Vegans ~50% more vulnerable due to the lower bioavailability of zinc from vegan sources
Can be bound by phytates, affecting bioavailability
Involved in the functioning of over 300 different enzymes, including superoxide dismutase, important for protecting cells from free radical damage
Zinc homeostasis within cells is maintained by the ZIP (zinc transporter) and ZnT (zinc transporter protein)
What is the regulation of zinc absorption?
When zinc is low, more zinc moves from the lumen into the mucosal cells and from vesicles into the cytosol, and little metallothionein is synthesized
When zinc intake is high, little zinc is transported from the lumen into the mucosal cells, and more zinc moves out of the mucosal cells into the lumen and from the cytosol into vesicles
The synthesis of metallothionein, which binds zinc and limits its uptake into the blood, increases
What are zinc fingers?
Small, functional protein domains that use zinc ions to stabilize their structure, enabling them to bind DNA, RNA, or proteins
Binds into the spiral of DNA like fingers
What are zinc lozenges?
Started with an agar plate, and when zinc was added, growth stopped
Created the conclusion that zinc helps with colds
Studies using zinc with COVID infection was there was no significant difference between zinc and common care
How is copper absorbed?
High levels of dietary zinc can inhibit copper absorption by stimulating the synthesis of metallothionein, which then preferentially binds copper and limits its absorption
Zinc and copper are in competition for binding to metallothionein for storage
Since Zinc is more common, there is an association with copper deficiency
What are thyroid hormones?
Tyrosine
Thyroxine (T4)
Most common
Not biological active form
Can convert into T3
Triiodothyronine (T3)
Biologically active form
*Every cell has receptor for thyroid hormones
What is the pathway for thyroid hormones?
T4 & T3 circulate in the blood bound to plasma proteins
T4 and T3 enter the cell, where a selenium-containing enzyme converts T4 to T3
T3 enters the nucleus and binds to a nuclear protein receptor
The T3-protein receptor complex then binds to a regulatory region of a target gene
Transcription of the gene is turned on, increasing the amount of mRNA made
mRNA directs translation, increasing the synthesis of the protein coded by this gene
There is an increase in the amount of protein, and hence the cellular functions and body processes affected by this protein
What is chromium?
Least understood trace mineral
Beneficial for blood glucose regulation
When chromium is present, a small peptide inside cells becomes active and enhances the action of insulin by binding to the insulin receptor, which increases glucose uptake
When chromium is deficient, the active peptide is not formed and thus cannot bind the insulin receptor
The result is that insulin is less effective and less glucose can enter the cell
What is fluoride?
Naturally occurring mineral
Rocks with fluoride will leech into water
Helps strengthen enamel
What is fluoride toxicity?
High doses can cause staining and pitting of the teeth
Exhibits why fluoride is good
Fluoride attaches itself to the teeth