Elimination reactions
1/52
Earn XP
Description and Tags
- Hydroxide ion acting as a base - Mechanism - Substitution vs elimination - Chlorofluorocarbons
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
How do halogenoalkanes typically react? When would they react by elimination?
Halogenoalkanes typically react by nucleophilic substitution but under different conditions they react by elimination
Why are elimination reactions called elimination? What is lost from the molecule and what forms?
Because a large molecule loses an atom or groups of atoms.
A hydrogen halide is eliminated, leaving a double bond in its place therefore an alkene is formed.
Finish the equation: C2H5Br + KOH (or NaOH) →
C2H4 + KBr + H2O
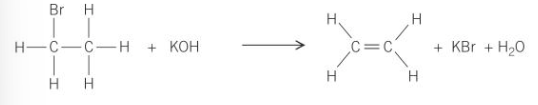
What does this -:OH act as in this instance?
A base
What is a base? What does a base do to halogenoalkanes?
A proton acceptor - removes a proton (H+) ion from the halogeonalkane
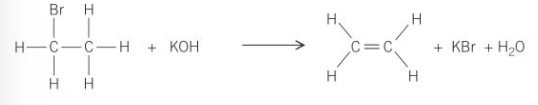
What are the two conditions for this reaction?
The KOH (or NaOH) is dissolved in ethanol - no water is present
The mixture is heated rather than just warmed
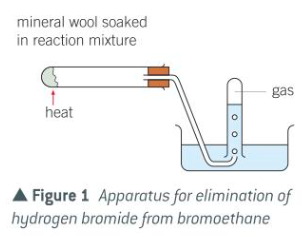
Apparatus used for the elimination of hydrogen bromide from bromoethane.
What is the gas product? How can you prove this product has been formed?
Ethene is the gas product. You can add the gas to bromine water and it will decolourise it if ethene is present, showing it contains a double bond
Is ethene able to burn?
Yes
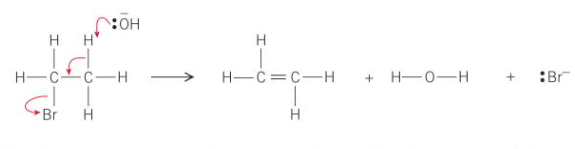
Mechanism for elimination of hydrogen bromide
The lone pair of electrons from the :OH- ion attracts to the delta positive H as it can be easily removed - acts as a base to form HBr
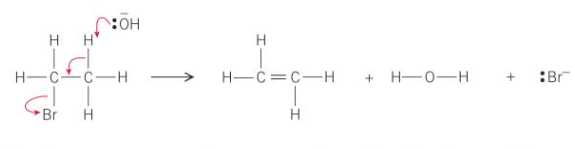
What do the curly arrows represent?
The movement of electrons
What does the hydroxide ion, OH-, do to the halogeonoalkane?
It acts as a base and uses its lone pair of electrons to form a bond with one of the hydrogen atoms on the carbon that is next to the C—X bond which removes it from the halogenoalkane
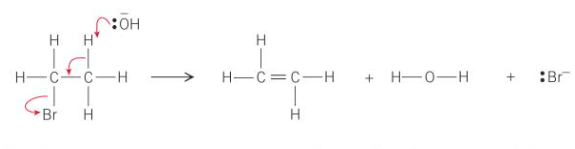
Where would the C—H bond that is broken by the -:OH be located in the halogenoalkane?
Next to the C—Br bond
How is the -:OH able to form a bond with the hydrogen atom?
The hydrogen atoms are slightly partically charged - delta positive
What happens to the electron pair from the C—H bond that has just broken?
It moves to be between the two carbon atoms to form the double carbon-carbon bond
What is the final part of the mechanism?
The C—Br bond breaks
How does the C—Br bond break?
The bromine takes the pair of electrons in the C—Br bond and leaves as a bromide ion
What is the bromide ion that is formed considered as within the mechanism?
The leaving group
What is the leaving group?
An atom or group of atoms that is ejected from the starting material - in this case the starting material is the halogenoalkane - normally taking with it an electron pair and forming a negative ion
What are elimination reactions useful for?
Making molecules with carbon-carbon double bonds
What is particular about the elimination reaction of longer-chained halogenoalkanes?
You can make different products because a variety of different hydrogen atoms can be removed
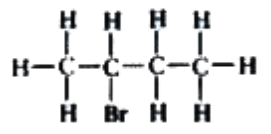
How many alkenes can form from the elimination reaction of 2-bromobutane? What hydrogen atoms could be removed from 2-bromobutane?
Two possible alkenes could be formed. Hydrogens connected to the carbons next to the C—X bond are removed.
It cannot be the hydrogen connected to the second carbon as this is where the halogen is positioned.
The hydrogens connected to the fourth carbon are too far away from the C—X bond
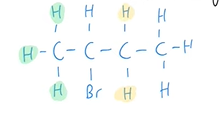
Why are both products able to be formed from the same reaction?
In mixtures of reactants there are billions of molecules so all the possibilities will occur
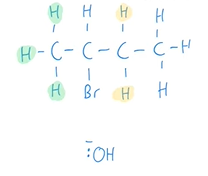
What two products are formed in the elimination reaction of 2-bromobutane with an hydroxide ion?
But-1-ene and but-2-ene
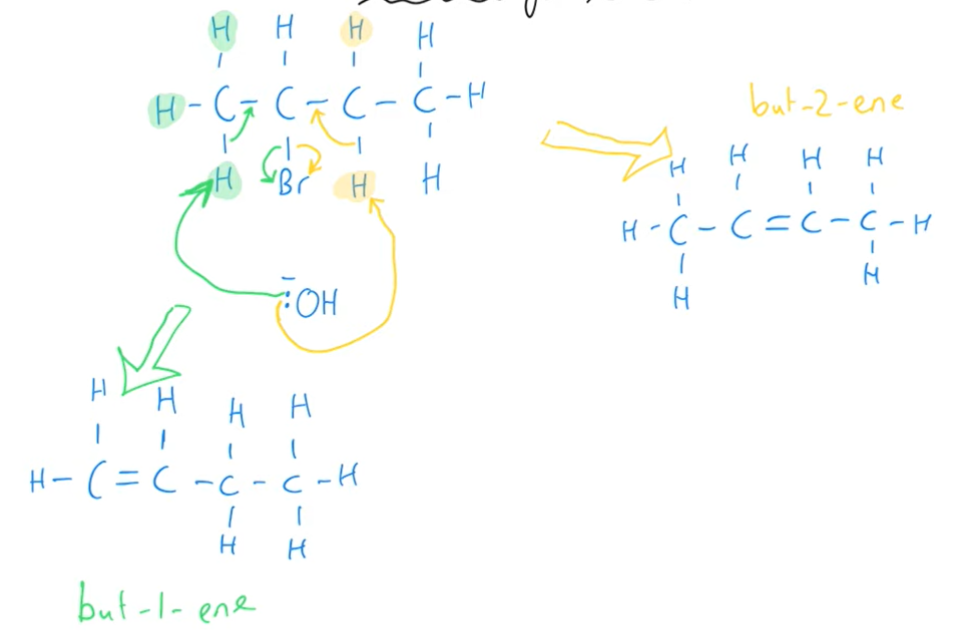
Making sure to draw the correct spacial arrangement around the alkene, what type of isomers does but-2-ene form?
E-Z isomers
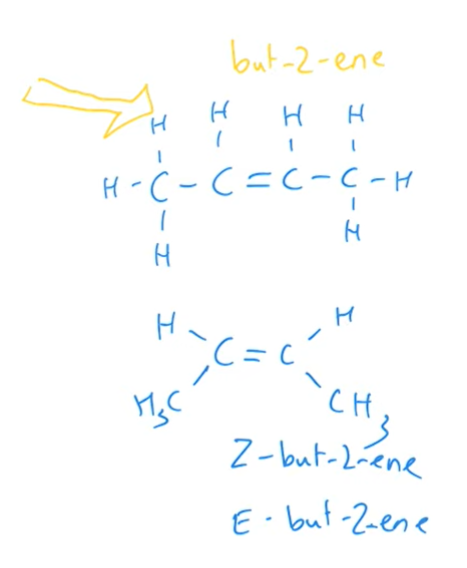
What results in a mixture of isomeric products?
When the halogenoalkane is not symmetrical
What are the two ways that the hydroxide ion can react with halogenoalkanes?
As a nucleophile or as a base
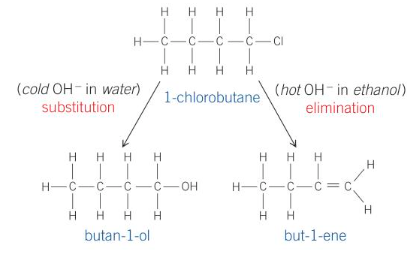
In the reaction between 1-chlorobutane and -:OH ions, both substitution and elimination reactions can occur. What products would be formed?
An alcohol from substitution and an alkene from elimination
butan-1-ol and but-1-ene
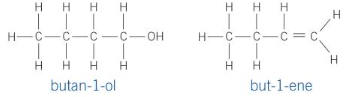
How would you seperate the mixture of products formed?
By fractional distillation
What conditions would favour the elimination reaction - producing more alkenes?
Hydroxide ions at high temperature, dissolved in ethanol
What conditions would favour the substitution reaction - producing more alcohol?
Hydroxide ions at room temperature dissolved in water (aqueous)
What other factor determines whether substitution or elimination will take place?
The type of halogenoalkane that reacts - whether it is a primary, seconday or tertiary halogenoalkane
How do primary halogenoalkanes tend to react?
By substitution
How to tertiary halogenoalkanes tend to react?
By elimination
How do secondary halogenoalkanes tend to react?
They do both substitution and elimination
What are chlorofluorocarbons?
Halogenoalkanes that contain chlorine and fluorine but no hydrogen
What is the abbreviation for chlorofluorocarbons?
CFCs
What physical state are short-chain CFCs? What is an example of a use for them?
They are gases and they are used as solvents and refrigerants
What are two uses for long-chain CFCs?
Dry cleaning and de-greasing solvents
What happens if CFCs manage to leak out of the products they are in?
They rise up to the ozone layer
What is the formula for ozone?
O3
What does ozone do in the upper atomosphere?
It decomposes to make oxygen gas:
2O3 → 3O2
Why is ozone beneficial?
It absorbs ultraviolet light from the sun
What can ultraviolet light cause in humans?
Cancer
What would have happened if the ozone layer didn’t exist?
Life as we know it wouldn’t have evolved to be what it is today
What reaction takes place when CFCs react with ozone?
Free radical substitution
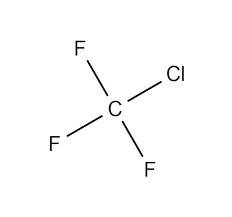
What is the first step which happens when CFCs rise to the ozone layer? What is the equation? (use CClF3 as an example)
Initiation - the UV light breaks the C—Cl bond, resulting in the formation of two chlorine atoms - two chlorine free radicals
CClF3 (U.V light) → •CF3 + •Cl
What do the chlorine free radicals do to the ozone in the stratosphere? What is this step called? What are the two equations for this?
Decomposes it - this step is called propagation
•Cl + O3 → ClO• + O2
The chlorine monoxide free radical, ClO•, then reacts with another ozone molecule:
ClO• + O3 → 2O2 + Cl•
What is significant about the propogation steps?
They repeat because the chlorine free radical is regenerated
What does the chlorine free radical effectively act as?
A catalyst - resulting in a high rate of reaction
What would be the last step in the mechanism? How do you determine the equation for this step?
Termination
Looking at the two propagation steps:
•Cl + O3 → ClO• + O2
ClO• + O3 → 2O2 + Cl•
The free radicals repeat and so can be cancelled out, ultimately resulting in 2 molecules of O3 decomposing to form 3 molecules of O2
2O3 → 3O2
What did research bring chemists to find out about the cause of the depletion of the ozone layer?
Chlorofluorocarbons were causing the depletion of the ozone layer
What did chemists then do to reduce the use of chlorofluorocarbons?
They developed an alternatives for refridgerants and coolants that did not contain CFCs
What did governments then legislate after being influenced by the scientist’s research?
For CFC’s to be phased out and be replaced by safer compounds