Chem
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
mixture
two or more substances that are not chemically bonded
separation techniques
filtration: removes large insoluble partacles form liquid, eg sand from water
Evaporation leaves behind crystals of a dissolved substance (if heated gently)
Distilation: condensing evaporated solvent and collecting it, (fractional distisation is same but with multiple liquids with diff boiling points
change in state:
heat is required to overcome electrostatic forces of attraction, between partacles to melt/ evaporate
charge and mass of an atom
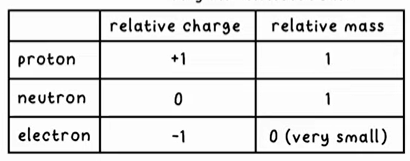
Atomic and mass numbers
Protons + neutrons = mass number
Atomic number (bottom) = number of protons
electron order
2,8,8,2
metals electrons
metals always donate electrons for an empty outer shell to form pos ions
non metals
always accept electrons to gain full outer shell, can form negative ions or share electrons
Alkalai metals, halogens, noble gasses
G1: alkali metals (all donate outer electon to become +1) , get more reactive as goes down group, as outer electon is further away from nucleus, more easily donated
G7: halogens bacially opposite, boiling points increases down group
G8/0: noble gasses, unreacive as full outer shell
transition metals
can donate diff numbers of electrons
metallic bonding
form a lattice structure (grid of ions) surrounded by delocalized electrons(can move)
ioic bonding
metal atoms donate electrons to non metals to form ions
dot + cross diagrams show electrons on outer shells, metals end up with empty shells, non metal with full outer shell, 8 electrons, CHARGES OF ALL IONS IN AN IONIC COMPOUND MUST ADD UP TO ZERO
forms lattice structure of repeating units of positive and negative ions - forms a crystal (high melting and boiling points, as bonds are strong)
covalent bonding
non metal bonding, atoms share electrons to get a full outer shell, every one consits of a pair of shared electrons, dot + cross diagram can be used , number of electrons reqiured = number of bonds formed,
giant covalent bonds,
forms giant structures that consist of repeating units of atoms, very high melting points as covalent bonds are strong,
alloys are mixtures of diff sized atoms = layers cannot slide over eachother, therefore they are harder,
moles and relative atomic mass
conservation of mass: total mass of reactants= tatal mass of products in reaction, even if gas leaves
moles = grams/relative atomic mass
limiting reactants
less amount of moles, means that it’s the limiting factor,
oxydatiion and reduction, OILRIG
oydation=loss reduction=gain (electrons) nutralistion- makes salt,
crystalisation
heat liquid gently so water evaporates, leaving slat behind
ph scale
stong acid lower ph weak acid high ph
electrolosys
involves passing an electical current through substance to cause oxydiation and reduction at anode and cathode
exothermic and endothermic
endo - cold
exo - hot
bond energies