CHEM-1411: Chapter 2 - Atoms, Molecules, and Ions
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
Law of Conservation of Mass
The total mass of substances present at the end of a chemical process is the same as the mass of substances present before the process took place.
Law of Multiple Proportions
When two elements, A and B, form more than one compound, the masses of B that combine with a given mass of A are in the ratio of small whole numbers
Postulates of Dalton’s Atomic Theory
All matter is composed of indivisible, indestructible atoms, with each element featuring unique mass and properties
Cathode Rays
Streams of negatively charged particles were found to emanate from cathode tubes, causing fluorescence'
J.J. Thomson discovered in 1897
The Electron
Thomson’s measurements allowed for calculation of the charge/mass ratio of the electron.
1.76×108 coulombs/grams
Millikan Oil-Drop Experiment
Once the charge/mass ratio of the electron is known, the determination of either the charge or the mass of an electron yields the other
Robert Millikan determined the charge on the electron in 1909 was 1.602×10-19C. Electron mass is now known
Radioactivity
the spontaneous emission of high-energy radiation by an atom
Three types of radiation:
Alpha: α particles - positively charged
Beta: β particles - negatively charged, like electrons
Gamma: γ rays - uncharged
Discovery of the Nucleus
Ernest Rutherford shot α particles at a thin sheet of gold foil and observed the pattern of scatter of the particles
Subatomic Particles
Proton: Positive (1+) - 1.0073 (Mass (amu)
Neutron: None (neutral) - 1.0087 (Mass (amu)
Electron: Negative (1-) - 5.486×10-4 (Mass (amu)
Atomic Number
The humber of protons in the nucleus of an atom
Since atoms have no overall charge, the number of protons equals the number electrons in an atom
Atoms of an Element
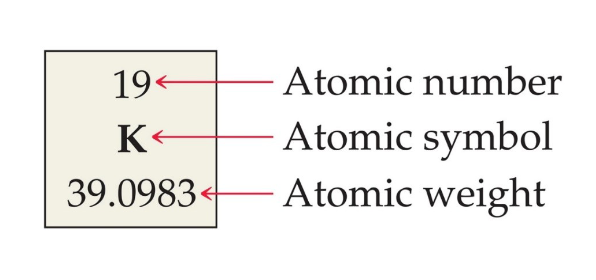
Atomic number = number of protons (above the element’s symbol)
Mass number = protons and neutrons (below the element’s symbol)
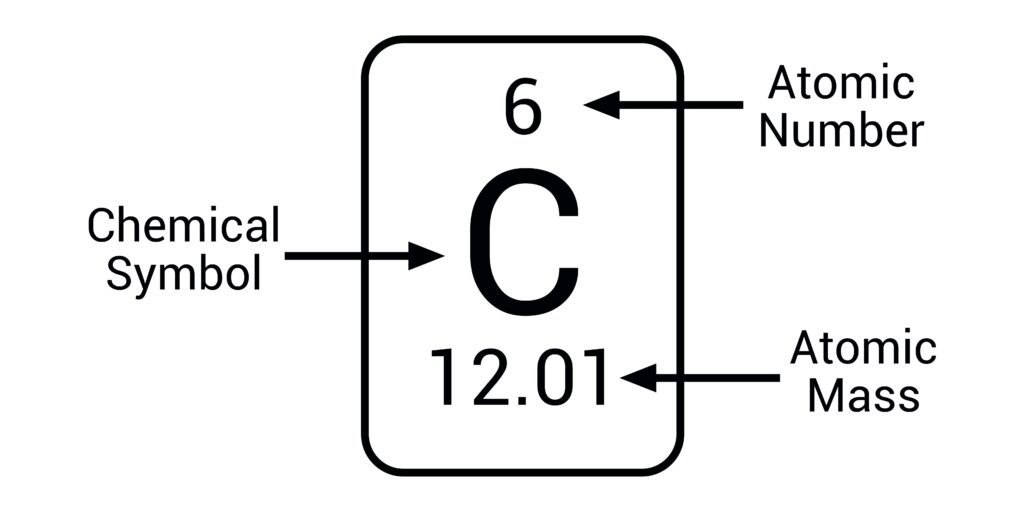
Isotopes
Atoms of the same element with the same number of protons but different numbers of neutrons (different masses).
Example: 13C - 6 protons, 6 electrons, 7 neutrons
Atomic Mass Unit (amu)
1 amu = 1.66054×10-24g
Atomic Weight
An average mass is found using all isotopes of an element, weighted by their relative abundances
Atomic Weight=Σ[(isotope mass)x(fractional natural abundance)] for ALL isotopes
-12C - 6 protons and 6 neutrons, has a mass of 12
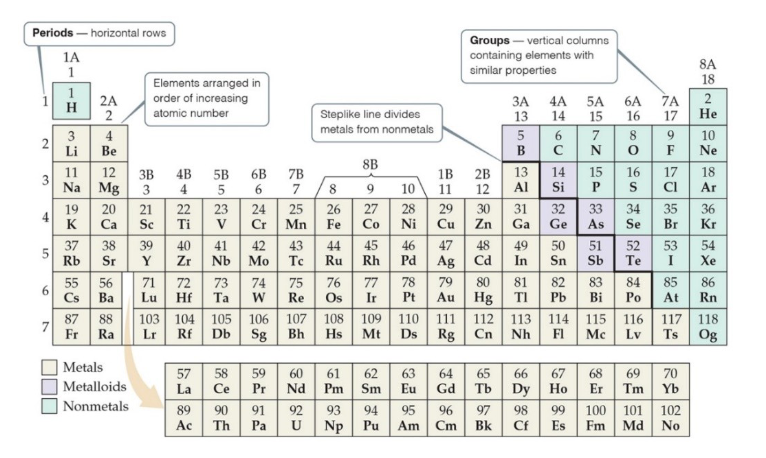
Reading the Periodic Table
Organization of the Periodic Table
The rows on the periodic table are called periods
Columns are called groups
Elements in the same group have similar chemical properties
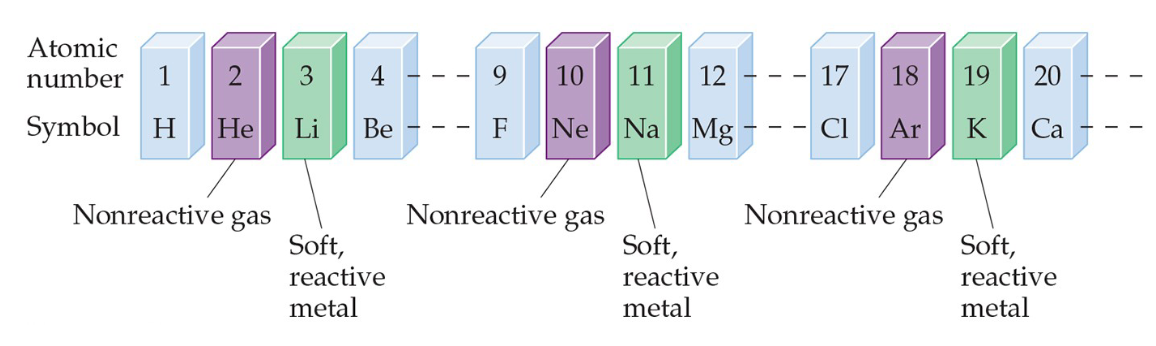
Periodicity-Rows
When one looks at the chemical properties of elements in a row, one notices a repeating pattern of properties and reactivity
Group 1A - Alkaline Metals
Li, Na, K, Rb, Cs, Fr
Group 2A - Alkaline Earth Metals
Be, Mg, Ca, Sr, Ba, Ra,
Group 6A - Chalcogens
O, S, Se, Te, Po
Group 7A - Halogens
F, Cl, Br, I, At
Group 8A - Noble gases
He, Ne, Ar, Kr, Xe, Rn
Metals
are on the left side of the periodic table
Some properties of metals include:
Shiny luster
Conducting heat and electricity
Solids (except mercury)
Nonmentals
Are on the right side of the periodic table (they include H)
They can be solid (like carbon), liquid (like bromine), or gas (like neon) at room temperature
Metalloids
Steplike purple line on the periodic table (Except Al, Po, and At)
Their properties are sometimes like metals and sometimes like nonmetals