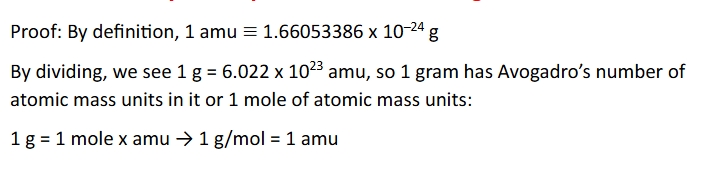CHEM1400 Chapters 4-5
1/97
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
98 Terms
There is a correlation between excited atoms and
their atomic number (Z) (frequency increased with Z)
Electron configurations
Hydrogen, H, is the simplest element with an electron configuration in its ground sate showed as 1s^1
1 on the left is the principle quantum number n=1
S is the s orbital
The superscript 1 shoe that a single electron is in the 1s orbital
Ground state
lowest energy configuration of electrons in orbitals
Electrons in orbitals of many-electron atoms
For H the energy is solely determined by the principal quantum number n. But for many-electron atoms the angular momentum quantum number l along with n are needed to determine the energy, because the electrons feel repulsive forces that can drive up energy
For any given n, energy increases with given l
The way electrons are spatially organized
matters in determining the energy
unless an external EM field is applied the ml doesn’t affect the energy of an electron in an orbital
Pauli Exclusion Principle
fundamental particles that have half-integer spins, notably the electron (ms= ± ½ ) that are in the same system (atoms) have to have a unique set of quantum numbers they cannot have the same set of quantum numbers as another electron in the same system
Set of quantum numbers
n, l, ml, ms
for H two valid sets of quantum numbers describes its one electron in the ground state (1, 0, 0, +1/2) OR (1, 0, 0, -1/2) we usually opt for the spin up electron
He has the electron configuration of 1s² in its ground state
its two electrons have to have two different sets of quantum numbers
(1, 0, 0, +1/2) AND (1, 0, 0, -1/2)
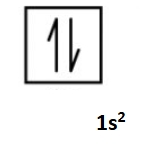
The Aufbau Principle
building up, we do the following scheme when deducing electron configurations of atoms across the periodic table
two electrons per orbital max, one is spin up and one is spin down
If an orbital is filled, the next electron goes into the lowest possible orbital
Li is 1s²2s^1
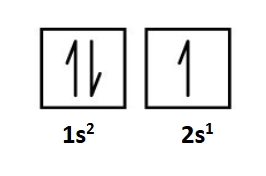
Degenerate
the orbitals have the same energy
Hund’s rule
more complicated for the 6th element C with adding the 6th electron
By building-up the ground-state electron configuration is 1s²2s²2p²
His rules are rooted in quantum mechanics
Lowest energy of a configuration when putting electrons in degenerate orbitals is when the electrons have the same spin
The requirement of the electrons having the same spin by Hund’s rule means they have to be in two different orbitals
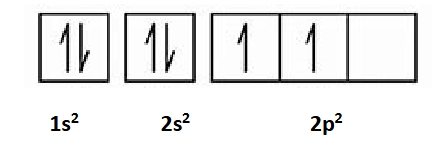
Diamagnetism
When an atom has all its electrons paired, they are repelled by magnetic fields, Helium in its ground state
Paramagnetism
unpaired electrons, like H, drawn into magnetic fields
Noble gas core also called noble gas notation
when writing in electron configuration, you can use this shorthand and start with the noble gas that is before the element you are writing
for Na the electron configuration is 1s²2s²2p^63s^1 but it can be shortened to [Ne]3s^1
Valence electron
the outermost shell, important for determining chemical reactivity and properties elements exhibit
S block
the far left of the table, Group 1 and 2, the alkali and alkali earth metals, valence electrons are in s orbitals
all metals
P block
right side of the table, groups 13 through 18, the s subshell is filled, and the remaining valence electrons are filling orbitals in the p subshell
metals, non-metals, and metalloid compounds
Main group elements
elements in the s and p blocks
D block
groups 3 through 12, transition elements, valence electrons are beginning to fill the d subshell
Where are there anomalies in electron configuration
in the d block
Chromium, Cr (Z=24)
[Ar]4s^13d^5
this happens because there is a slight energy advantage in having a half-filled d subshell
![<p>[Ar]4s^13d^5</p><p>this happens because there is a slight energy advantage in having a half-filled d subshell </p>](https://knowt-user-attachments.s3.amazonaws.com/9a4f87bd-b3ed-4c59-8a41-10a9984dec07.jpg)
Copper Cu Z=29
[Ar]4s^13d^10
the filled ground d subshell has a slight energy advantage
Zinc Zn Z=30
[Ar]4s²3d^10
Lanthanides
should start filling the seven degenerate 4f orbitals, but in fact 6s, 5d, and 4f are so close in energy that it makes predicting the actual ground state configuration using qualitative rule sets like Hund’s rules impractical
Actinides
below the lanthanides, begin filling 5f orbitals, with complicated electronic configurations due to the closeness of energy of 5f with 6d and 7s orbitals
Effective Nucellar Charge (Zeff)
while the number of protons Z dictates nuclear charge, electrons can effectively block or shield other electrons from feeling the nucleus by exerting repulsive forces on the electron that competes with the attractive force between the nucleus and electron
0<Zeff<Z
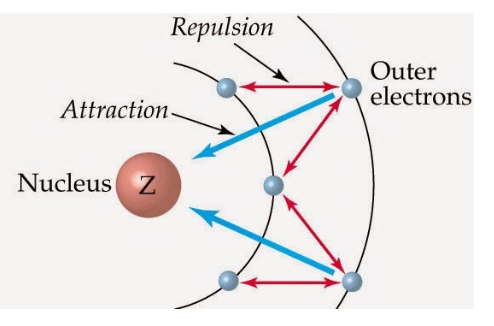
Shielding constant
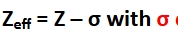
Atoms aren’t hard spheres
with discrete boundaries as predicted by quantum mechanics and shown by recent imaging studies of single atoms
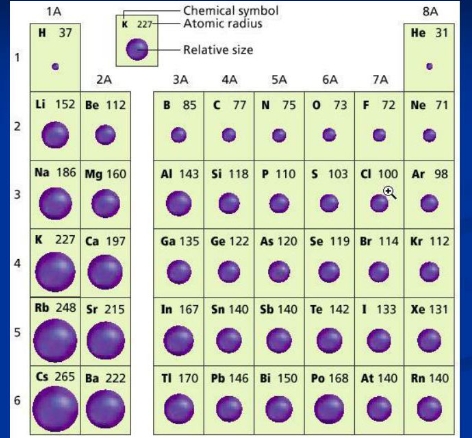
Atomic radius
has 2 definitions
Metallic radius
Covalent radius
Metallic radius
half the distance between two nuclei of 2 neighboring identical metal atoms
the metallic diameter is the distance between the two nuclei of neighboring identical metal atoms
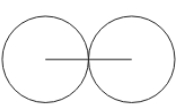
Covalent radius
the distance between two adjacent nuclei that connected by a chemical bond
the covalent diameter is the distance between the two adjacent nuclei that are connected by a chemical bond
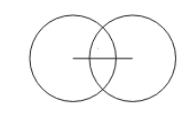
Atomic radius
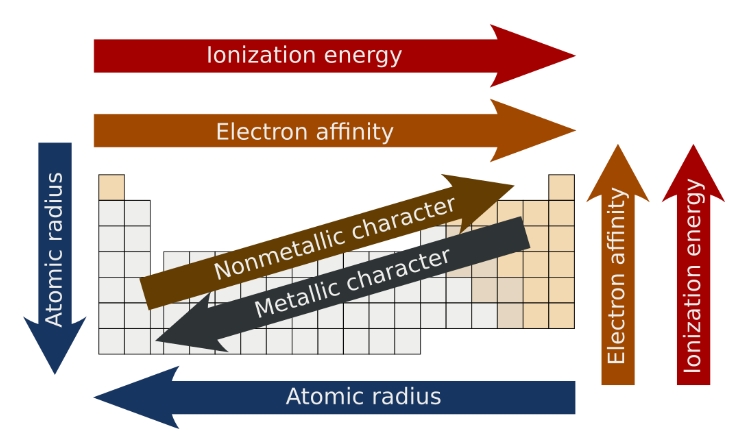
increases down a group because n increases as you move down a group which increases the distance of electrons in the outermost shell are from the nucleus
it decreases from left to right on the periodic table. Across the period this is mainly due to Zeff increasing from left to right
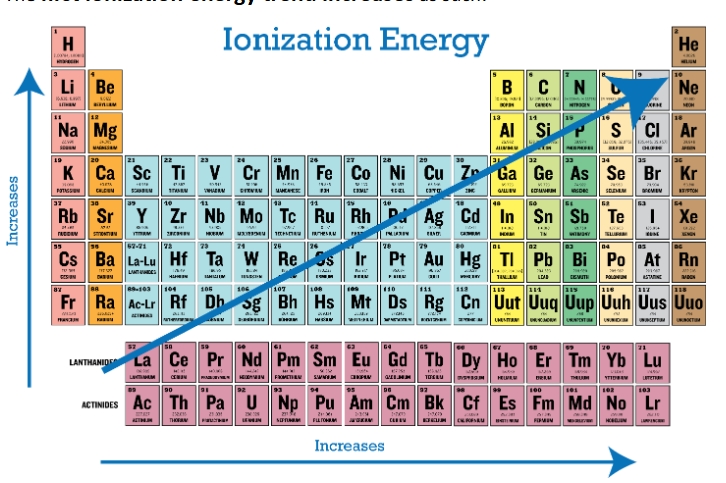
Ionization energy
IE minimum energy to remove an electron from an atom in the gas phase
across the period IE1 increases with Zeff and in general the electrons are easier to remove the further you go down a group so IE1 decreases

Cations
positively charged ions like Na+

Successive ionization steps become harder
because of the coulombic attraction between the electron and the cation
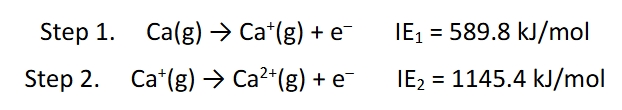
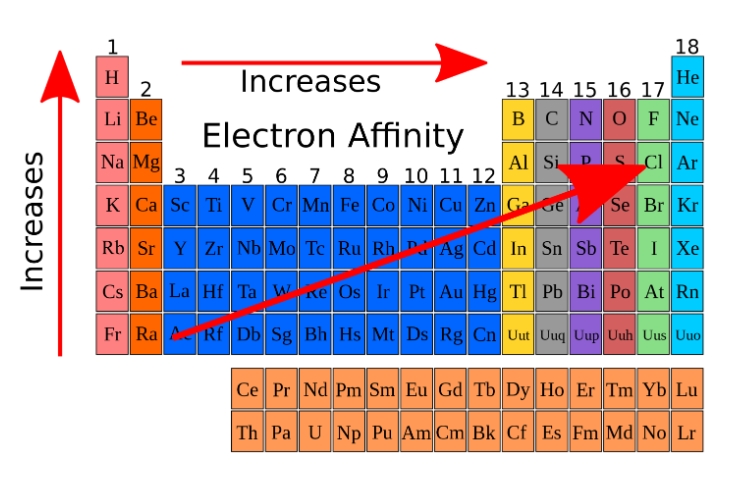
Electron affinity EA
defined as the energy released when an atom is in the gas phase accepts an electron
this means the larger positive EA energies are, the more energetically favorable the attachment is
Anions
negative ions
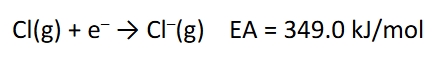
Second electron affinity EA2
gas-phase anions do not have an affinity for electrons so the EA2 values are negative
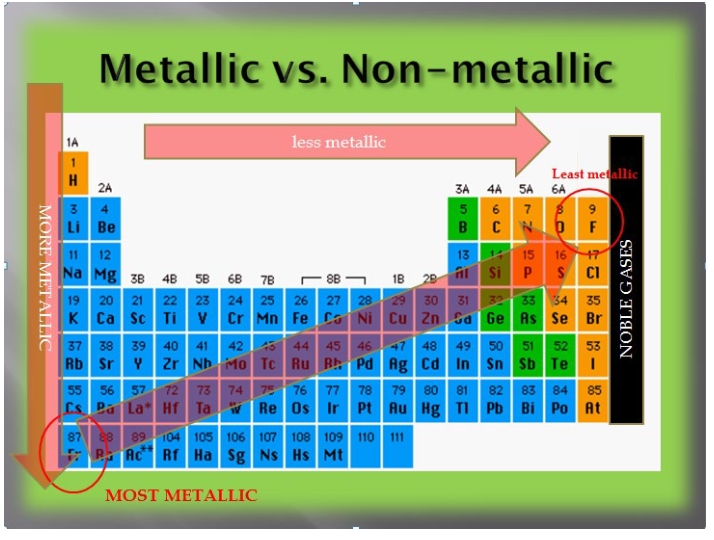
Metallic character of elements
most elements are metals with all of the s block elements, d and f block being metals and the non-metals limited to the p block (with a lot of the p block being metals and metalloids)
metals tend to be shiny, lustrous, malleable and ductile, conductive (heat and electrical), tend to form cations
Non metals characteristics
dull, not lustrous, brittle, poor or limited heat and electrical conduction (exceptions exists) ten to form anions
Metallic properties increase from right to left across the period
Francium Fr Cesium Cs are very metallic
F and O are very non-metallic
Metallic properties increase down a group because of larger n
places the electron further from the nucleus, making it easier to ionize
Summary of the periodic table characteristics
Electron configuration of ions
many common substances are made up of monoatomic ions
Monoatomic ions
cations and anions that are single atoms ex) Na+ and Cl-
Ions of main group elements
metals tend to lose electrons (form cations) and non metals tend to gain electrons (form anions) with the exception of noble gases, which tend to not be ionized under ordinary conditions
Main ground elements tend to form what type of ions
form ions that give them the noble gas electron configuration of the nearest noble gas
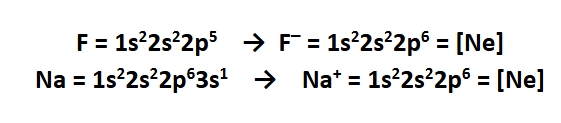
Isoelectronic
even though there are different electron configurations for regular elements like F and Na, the electron configuration of Na+ and F- are identical
Ions of the d block elements
recall from building up that 4s fills before 3d ( they are really close in energy) but as d fills it shields 4s from the nucleus as suggested by a plot of the radial distribution function, hence 4s electrons are easier to remove than 3d electrons
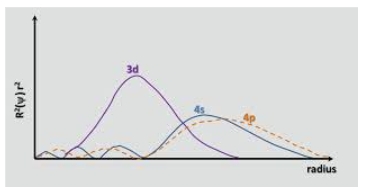
Ionic radius
the radius of an anion or cation and itis important in dictating the chemical and physical properties of ions
for cations- it is smaller than that of the parent atom because there is less electron-electron repulsion and decreases a lot when all valence electrons are lost
for anions- tend to be larger than the parent element due to increased electron-electron repulsion
Compounds
substances made of 2 or more elements and unlike a mixture can’t be separated by a physical process

Lewis dot symbols
a symbolic representation of atoms, ions and molecules that’s based on principle that atoms usually react through their valence electrons, and it is designed to show electrons in a manner that is suggestive of reactivity
for the main group the main number of valence electrons surrounding an atom is equal to its group number
Anion example of lewis dot structure Cl-
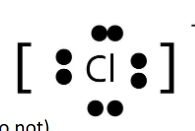
Ionic compound
when cations and anions come in proximity to each other, electrostatic attraction brings them together
cations are typically metals that tend to lose electrons (form cations) and anions are typically nonmetals that tend to gain electrons (form anions)
most are binary compounds formed derived from a metal and nonmetal
ex Na+CL- Sodium chloride NaCl
Chemical formulas/simply formulas
representations like NaCl and MgBr2, though the ionic compounds are formed of charged particles we typically leave out the charges in the formula
Ionic compounds tend to form
crystals in what we formally call lattices
Lattice energy
one of the ways we characterize ionic compounds
closely related to lattice enthalpy
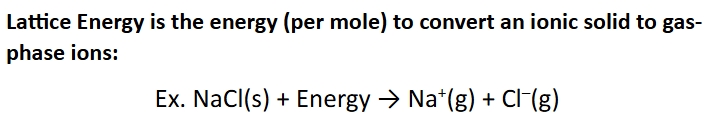
The greater the lattice energy
the more stable the ionic solid is and melting point usually increases with lattice energy
Lattice energy increases when
the bond length is shorter
*Coulomb’s law
Lattice energy increases with
increasing charge of the ions comprising the lattice
Nomenclature
naming ions, compound and other chemical entities
Naming main group monatomic ions
adding ion to the name
nothing needs to be specified for the charge because in groups 1, 2, and 13 the charge is almost always +1, +2, +3

Transition metal ions naming
more complicated than main group cations because these can form different charges

Monatomic anions naming
simply put ide at the end of the elements name followed by the word ion
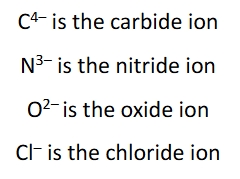
Formula units
simplest formula, the key to writing ionic compounds is adding cations and ions together in ratios that give a neutral compound, since ionic compounds tend to form a lattice (crystals)

Transition metals combining
the same rule for making the ionic compounds is the same as for ionic compounds derived from metals from the main block. However, the naming has to account for the possibility cation derived from the transition metal has more than one common possible charge

Ionic compounds tend to form
between metals (readily form cations) with nonmetals (readily form anions) and involve the complete transfer of the electrons from the former to the latter
Covalent bonding
involves a sharing of an electron and is very between nonmetal elements
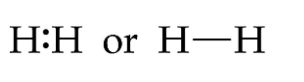
Molecules
a molecule is a neutral combination of two or more atoms held together by a covalent chemical bonds
Molecules can be elements
H2, O2, O3, N2, Cl2
there are 7 common diatomic elements H2, N2, O2 and the halogens F2, Cl2, Br2, and I2
Molecules can be compounds
made of different types of atoms with simple examples being NO, H2O, and NH3
Diatomic Molecules
have two atoms
H2, O2, NO
Homonuclear diatomic molecules
H2 and O2
Heteronuclear diatomic molecules
NO, has two different atoms
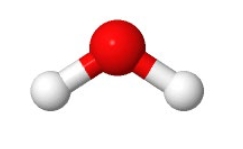
Polyatomic molecules
have more than two atoms with the most common ex being H2O
most important organic and biological molecules
Molecular formulas
show the exact numbers of atoms of each element in each molecule
H2O, H2, NH3
Allotropes
special class of elements that have 2 or more distinct forms

Structural formulas
these structures not only how the composition of atoms but give the general arrangement in 2D
ex) H-O-H
Ball and stick model
Empirical formulas
formulas used for molecules that represent the simplest whole number ratio of atoms in the molecule
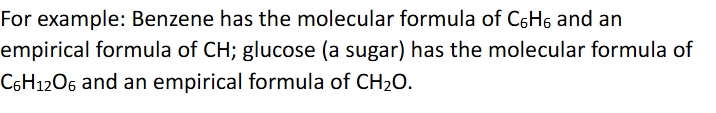
Naming molecular compounds
name the element that appears in the formula and add an ide to the name of the second element
Add Greek prefixes to specify the number of atoms in the molecular formula (and we usually omit mono for one with CO, carbon monoxide is a notable exception)

Compounds containing hydrogen naming
there are many compounds with hydrogen that don’t follow nomenclature rules
H2O - water
NH3 - ammonia
Many acids contain H and when they are in the gas phase
they are named like HCl hydrogen chloride
Many acids contain H and when they are dissolved in water
like the aqueous phase
HCl (aq) hydrochloric acid
Polyatomic ions
these are ions that consist of two or more atoms
common pitfall- students tend to pull apart these ions into component atoms. Under ordinary chemical conditions, like the reactions you do in lab, and we’ll encounter in these classes, these ions generally stay intact
Polyatomic ions combining
polyatomic ions often combine with monatomic ions to form ionic compounds, put the polyatomic cation first followed by the anion name
Polyatomic cations can also react with polyatomic anions
Neutral ionic compound
in order to form this, there has to be 3 ammonium ions to one phosphate ion. This compound would be named in accord with ionic compounds (not molecular compounds): ammonium phosphate
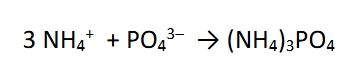
Oxoanions
class of polyatomic anions that contain one or more O atom and very common in the lab and nature. Two important series are look at picture

Chlorates are a more complex series

Oxoacids
acids derived from oxoanions that when dissolved in water result in the formation of hydronium ions and oxoanions. These are commonly used in labs, including gen chem, so the nomenclature is important

A series of acids and ions derived from the phosphate ion POsuper script 3- subscript 4

Two really important oxoacids for labs, including gen chem

Molecular and formula masses
use the periodic table with molar masses(amu) listed. Molecular mass (aka avg molecular mass or molecular weight) is the sum of atomic masses (in amu) for all the atoms in a molecule
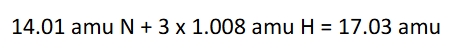
Formula mass aka formula weight
calculated the same way as molecular mass, but the term applies to formula units for ionic compounds
Percent composition (ex. percent mass) of compounds
n denotes the number of the element in question within the compound
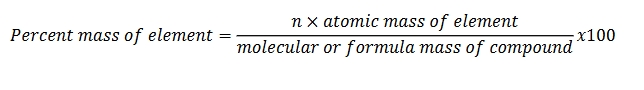
Molar mass
defined as the mass in grams of a substance per mole
1amu=
1g/mol