Nuclear Chemistry
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Nucleon
a proton or neutron
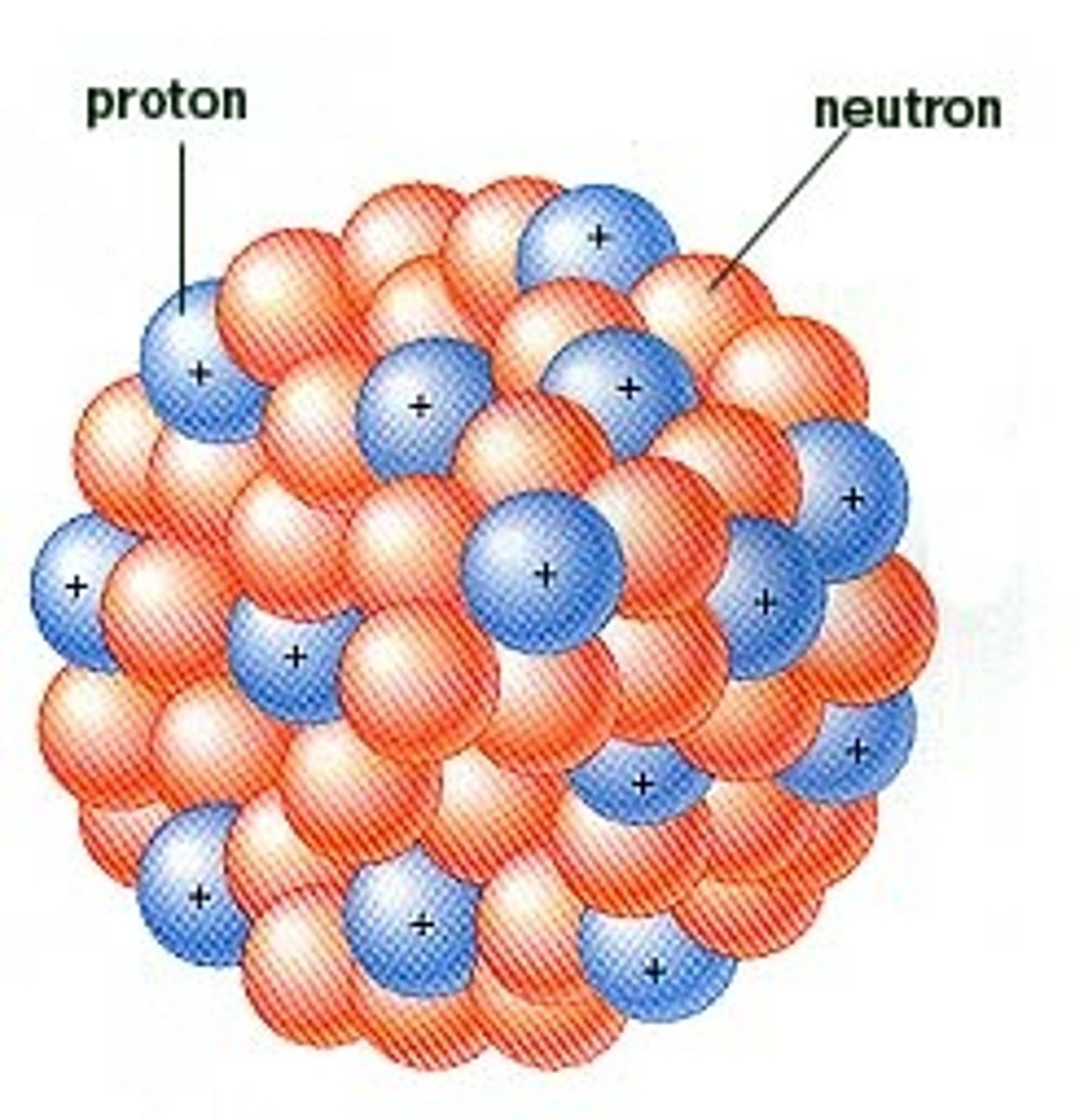
Nuclide
a specific nucleus
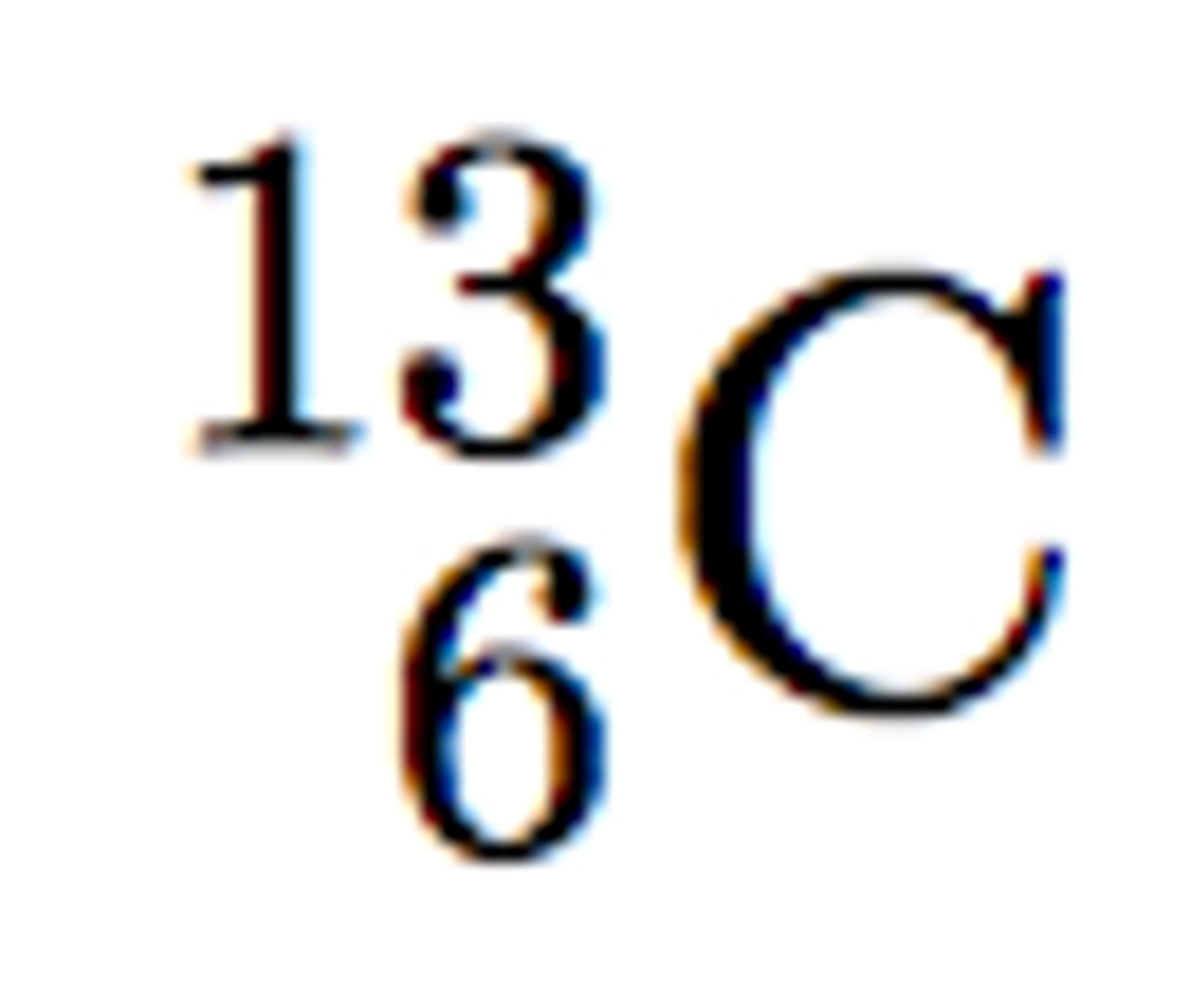
Nuclear Binding Energy
the energy released when a nuclide is formed
Magic Numbers
2, 8, 20, 28, 50, 82, 126; atoms with these numbers of protons or neutrons are more stable
N/Z ratio for small atoms
1:1
N/Z ration for large atoms
1.5:1
Decay Series
series of decay in which radioactive element is decomposed in different elements until it produces one stable atom
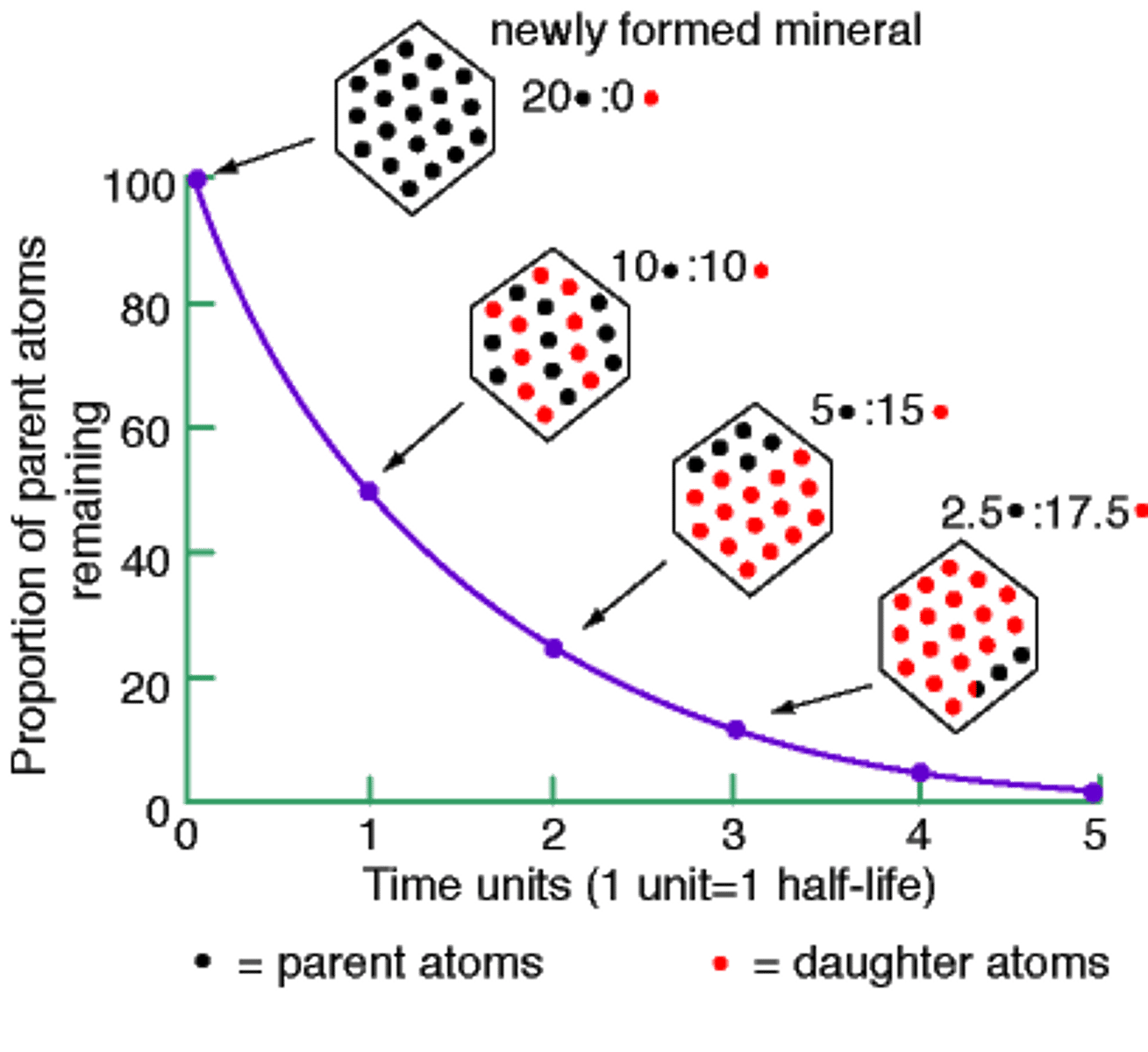
Half-life
the time it takes for one half of a radioactive sample to decay
Alpha Paticle
particle releaed during alpha decay; esentially the nucleus fo a helium atom; contains 2p and 2n
Beta Particle
particle released during beta decay; essentially an electron
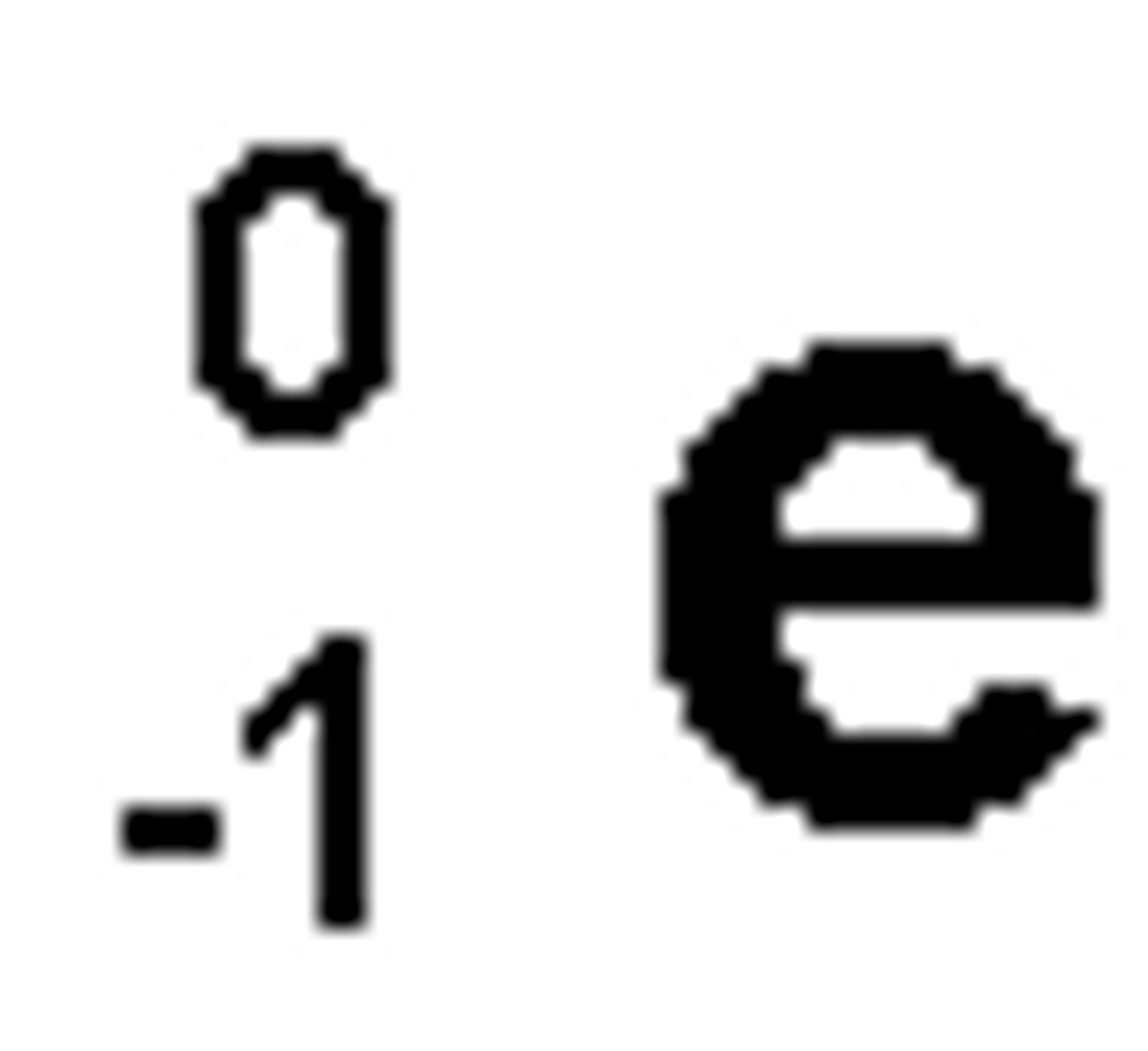
Positron
particle released during positiron emission; essentially a positively charged electron
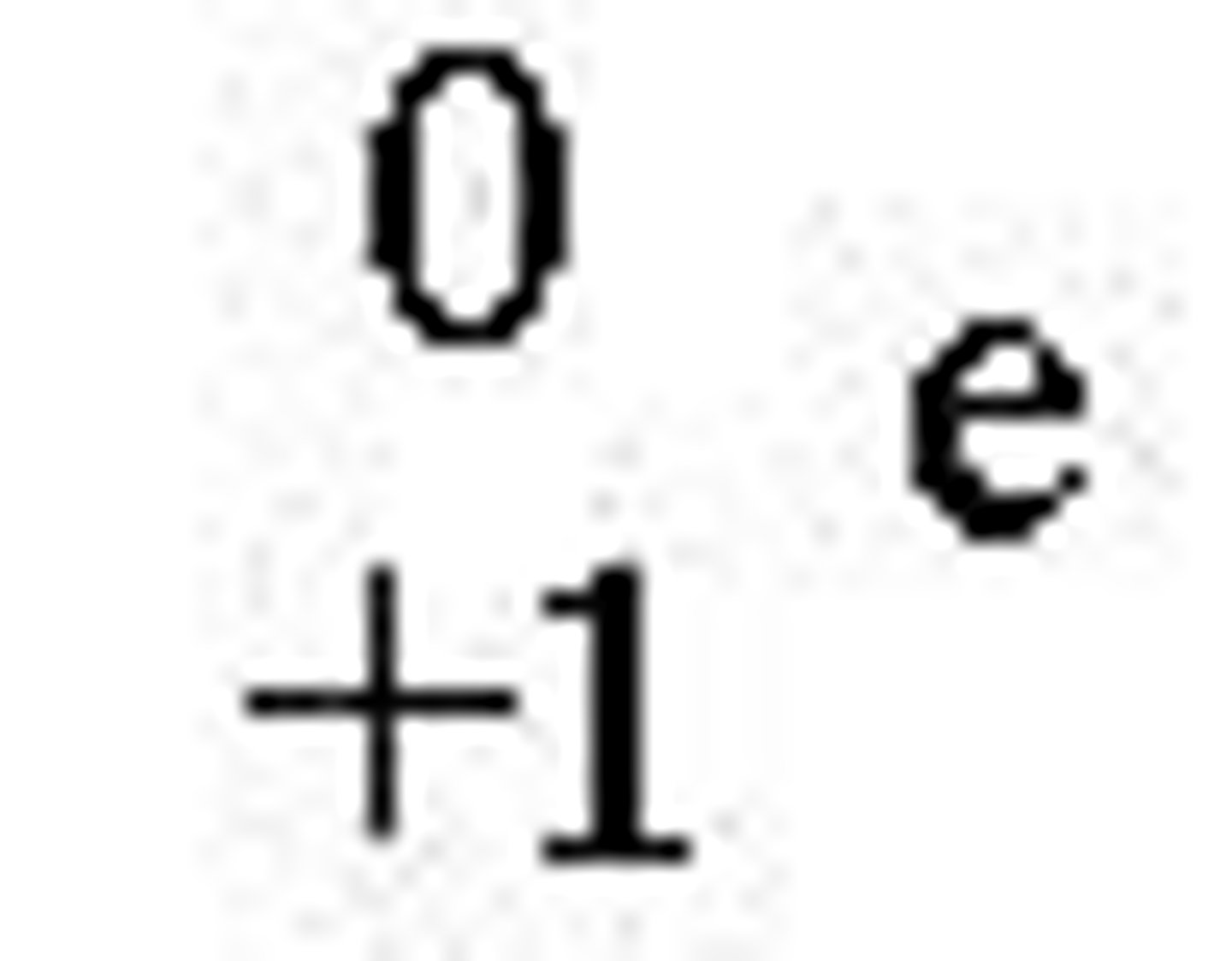
Gamma Ray
energy released during nuclear reaction; has no mass
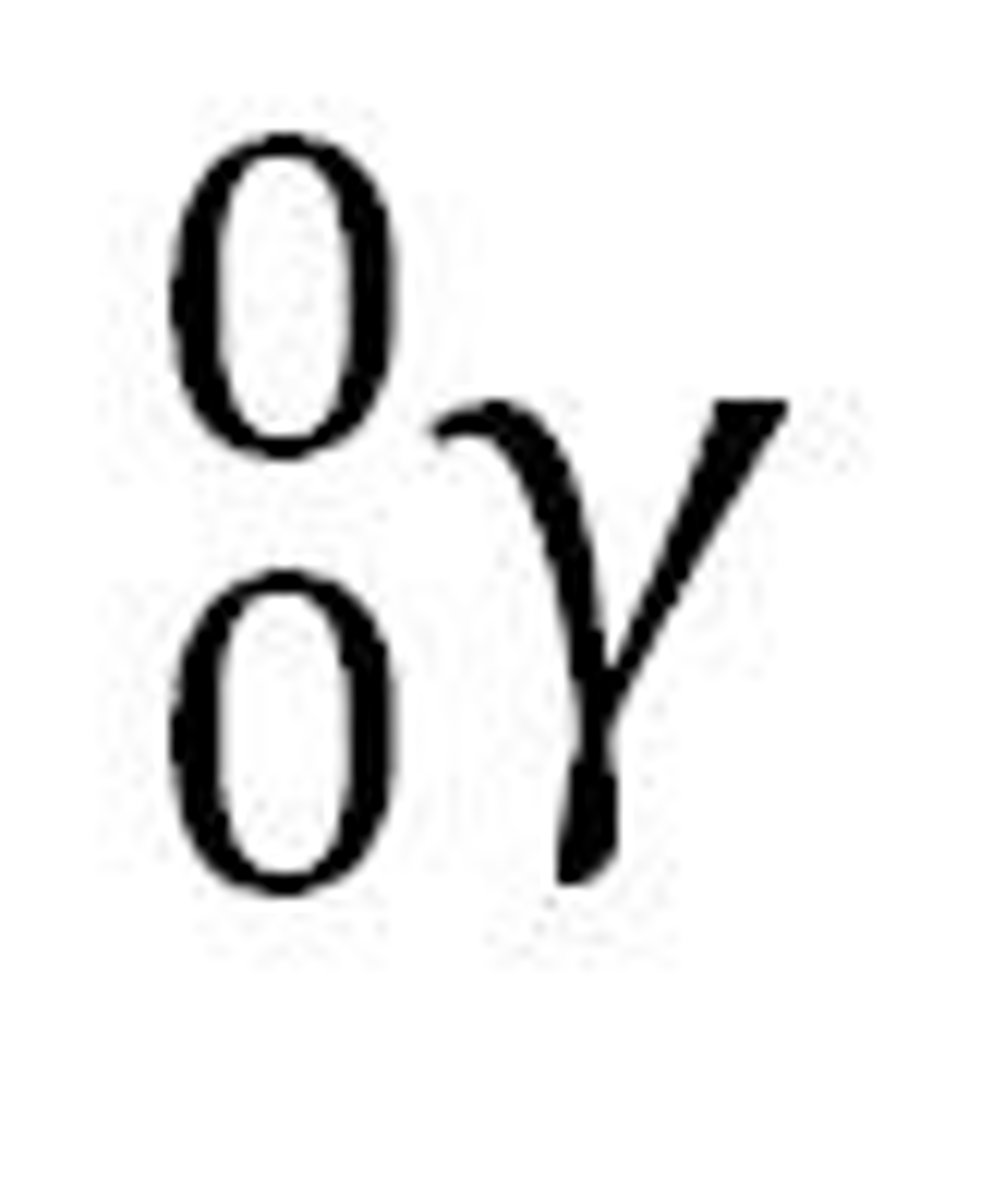
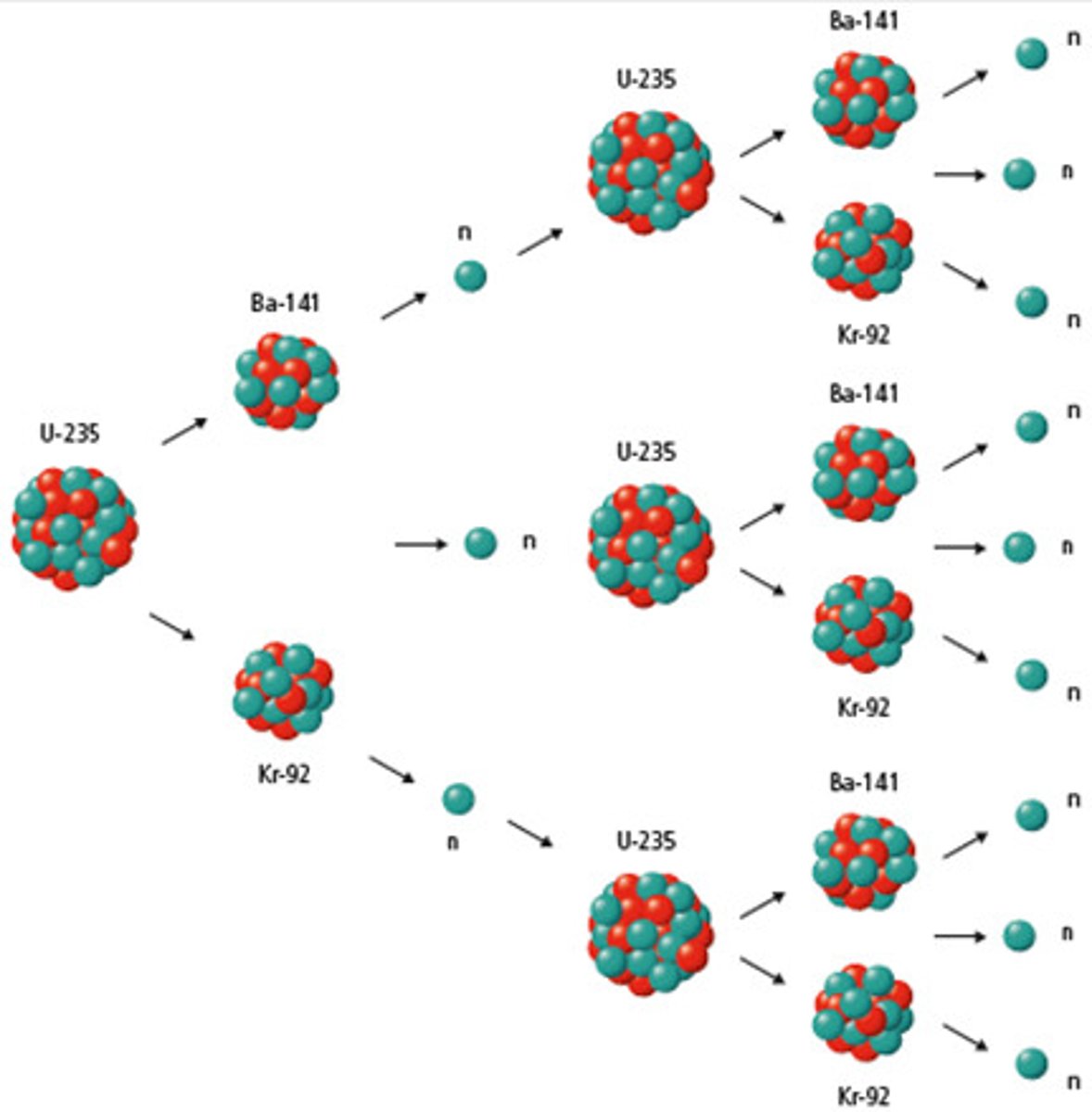
Chain Reaction
a reaction that prepetuates itself; one of the reactants in the reaction is also a product of the reaction
Critical Mass
the smallest amount of fissionable material required to sustain a chain reaction
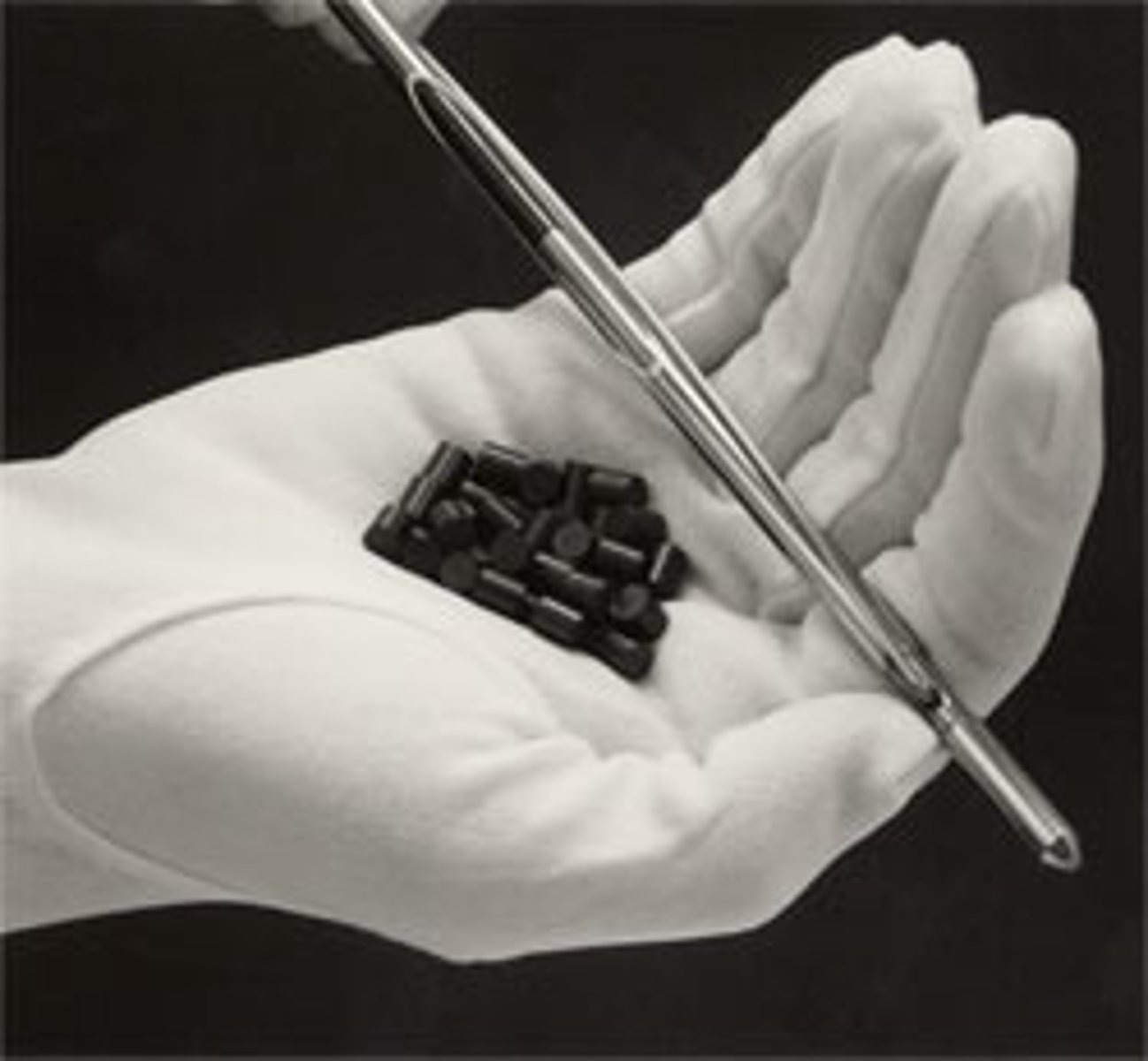
Fuel Rods
pellets of uranium arranged in long steel cylinders
Contol Rods
rods made of nonfissile material which help control the rate of fission in the reactor
Steam Geneator
uses energy from the primary loop to convert water in the secondary loop to steam
Turbine
large blades which spin when hit by pressurized steam. The roataing blades cause a shaft attached to the generator to spin.
Generator
consists of a huge mass of coiled wire spinning within a magnetic field to create an electric current
Pressure Vessel
heavily shielded structure contains fuel rods, control rods, and coolant
Containment Building
acts as protective barrier from radiation emitted from within the pressure vessel
Cooling Tower
extract the excess heat gained in th condensor loop before water is discharged into the ocean

Nuclear Fission
reaction in which a heavy nucleus splits spontaneously or on impact with another particle, with the release of energy
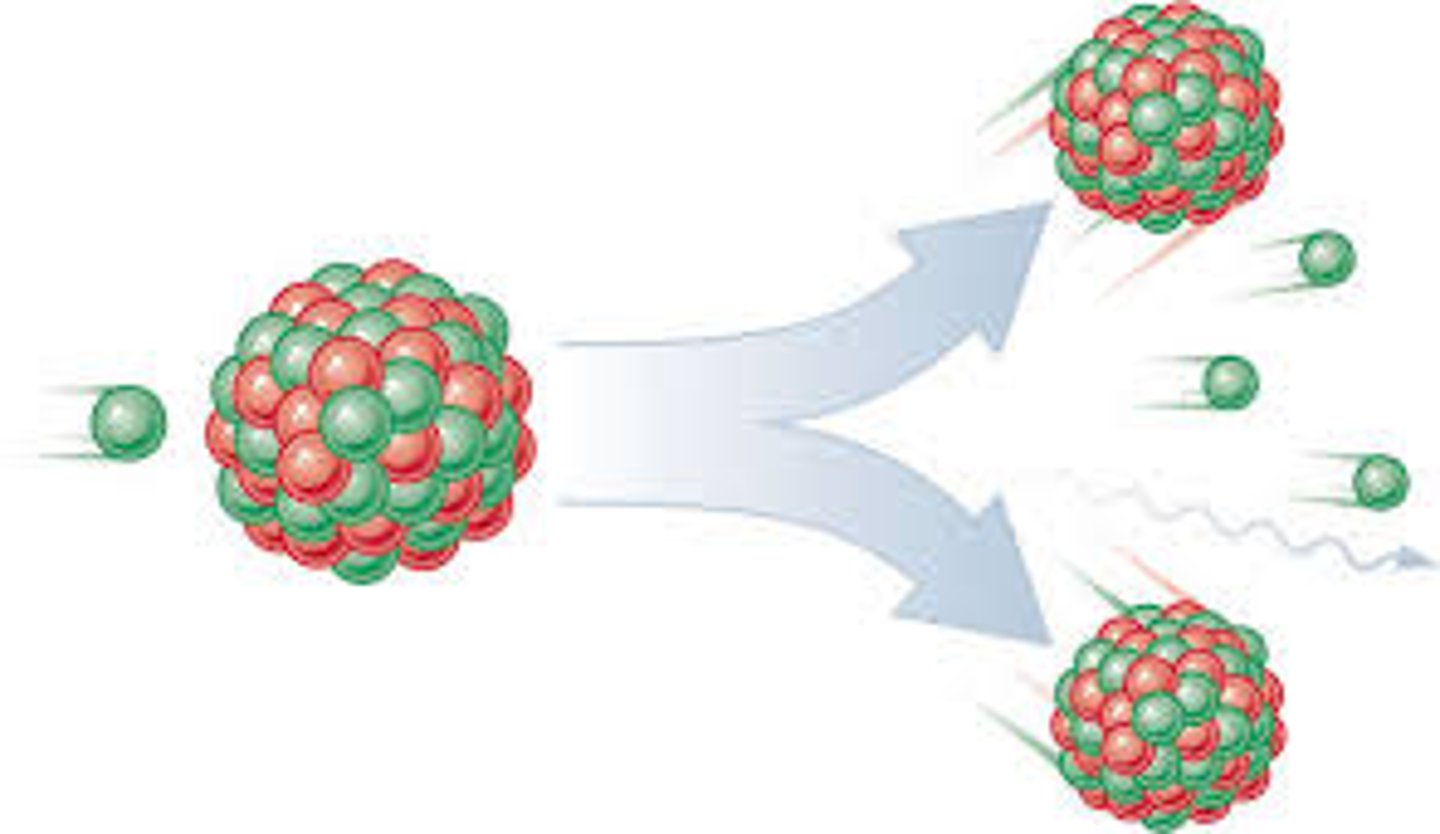
Nuclear Fusion
reaction in which atomic nuclei of low atomic number fuse to form a heavier nucleus with the release of energy.
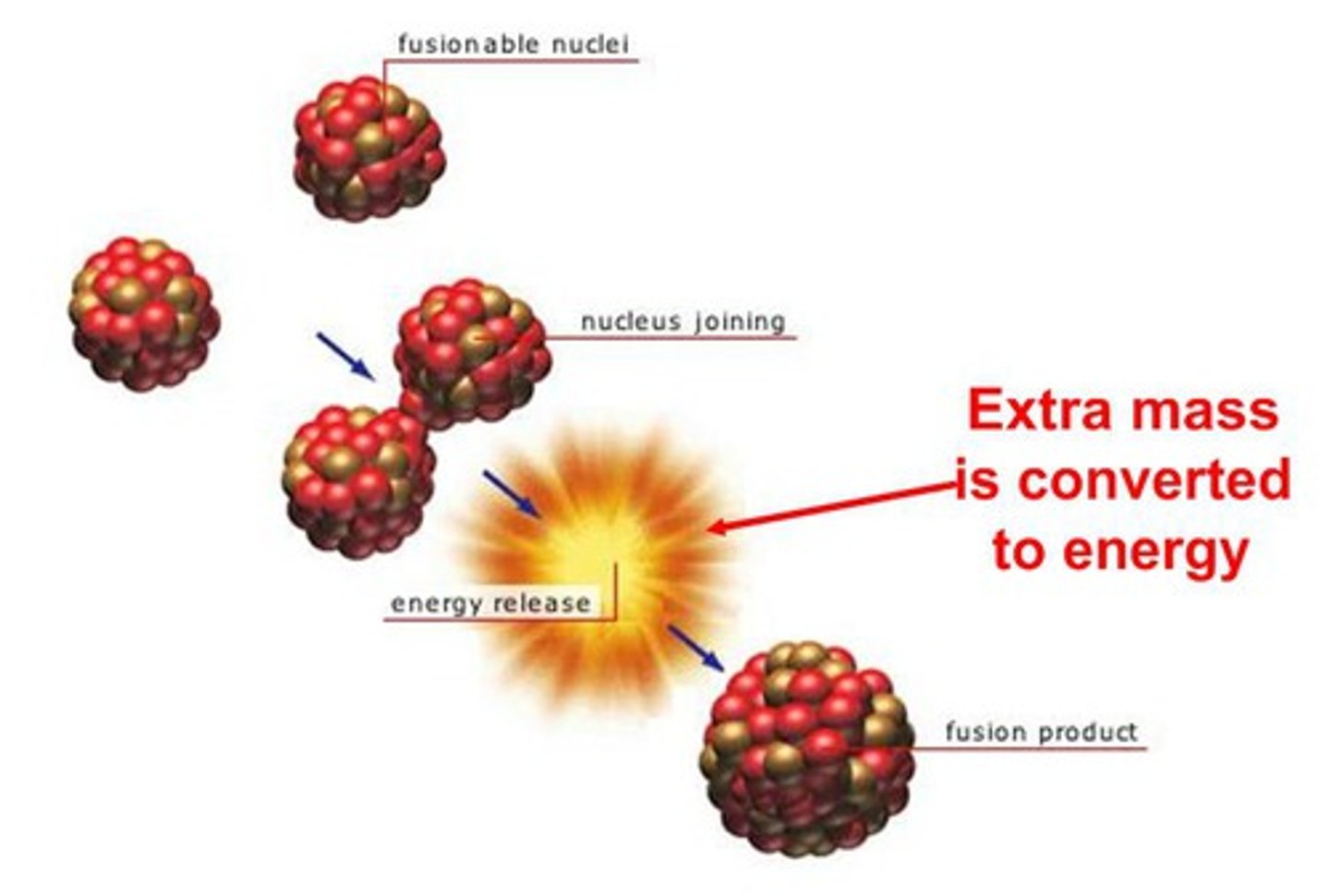
Mass Defect
the difference between the sum of the mass of the parts of a nuclide and the mass of the nuclide itself; the sum of the parts is always larger than the mass of the nuclide
Mass number
# p + # n; the top number in isotopic notation; 56 in this example
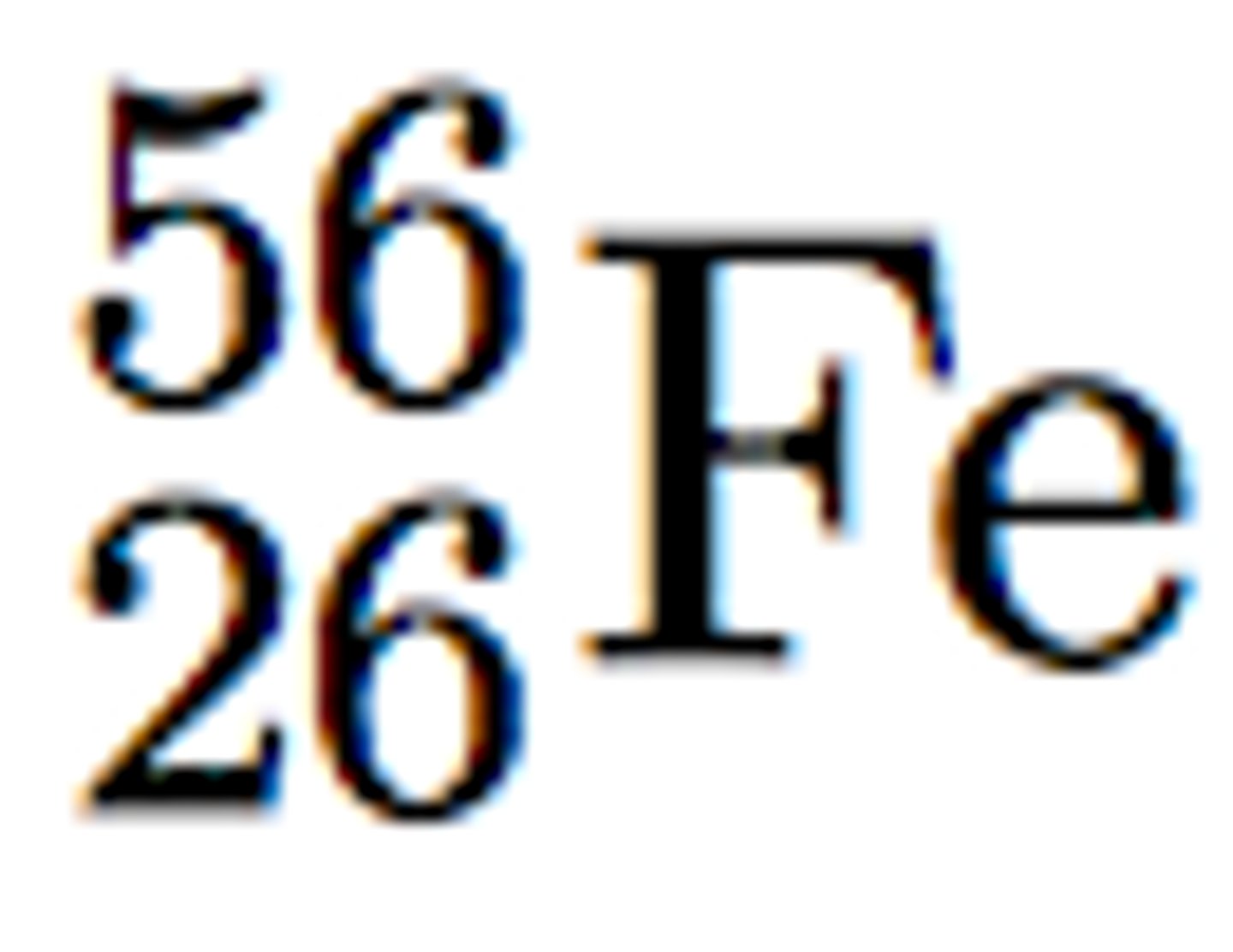
Atomic Number
# p; the bottom number in isotopic notation; 26 in this example
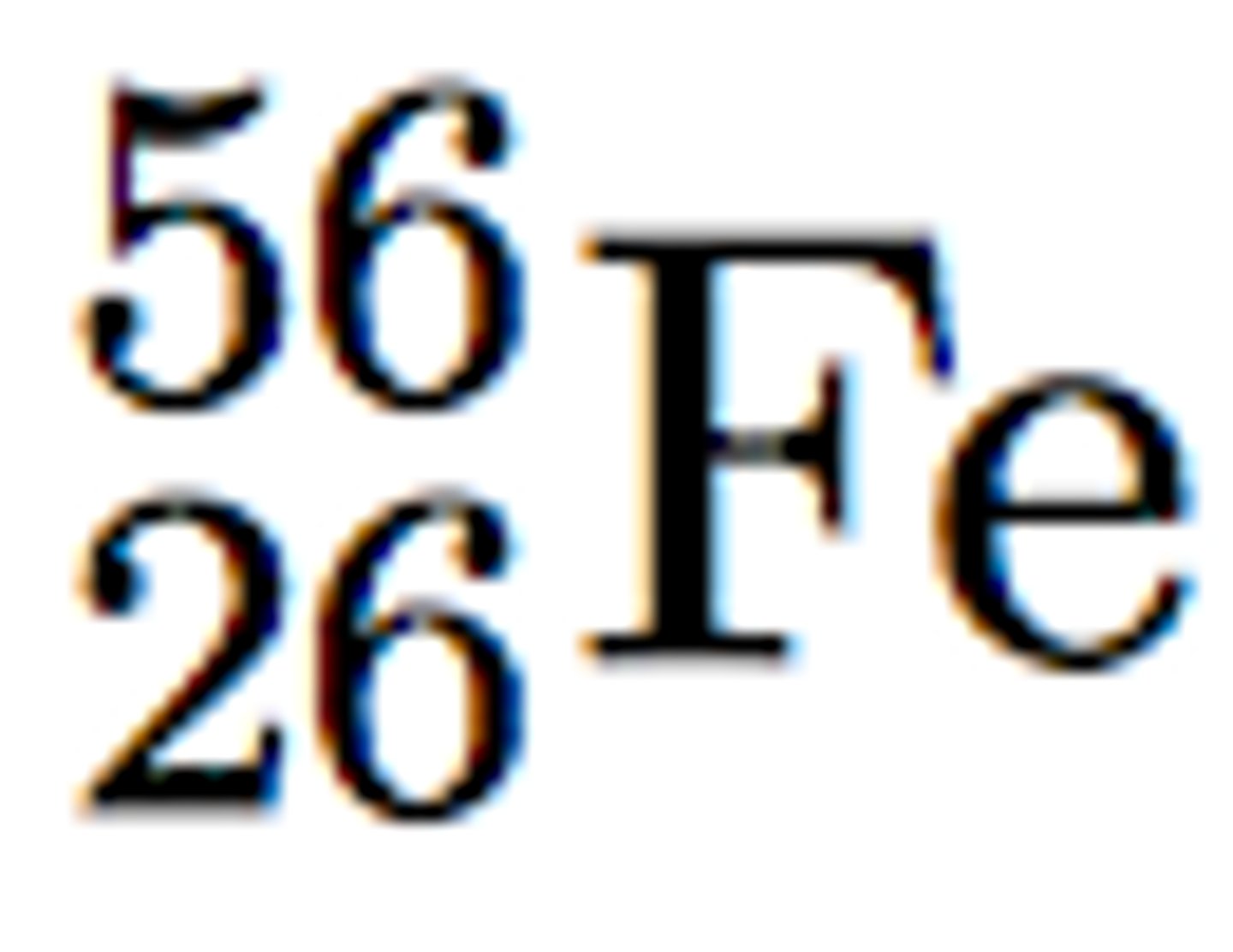
Alpha Decay
radioactive decay that produces an alpha particle and a new element; the new element's mass number decreases by 4 and the atomic number decreases by 2

Beta Decay
radioactive decay that produces a beta particle and a new element; the new element's mass number stays the same and the atomic number increases by 1
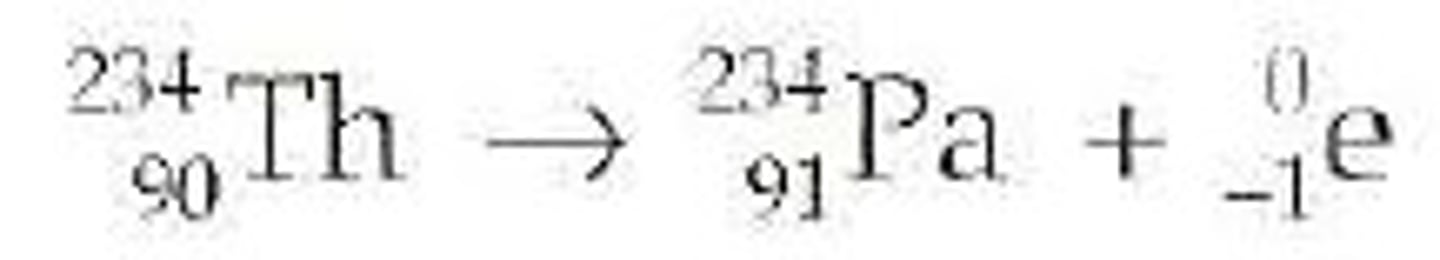
Positron Emission
radioactive decay that produces a positron, 2 gamma rays, and a new element; new element's mass number stays the same and the atomic number decreases by 1
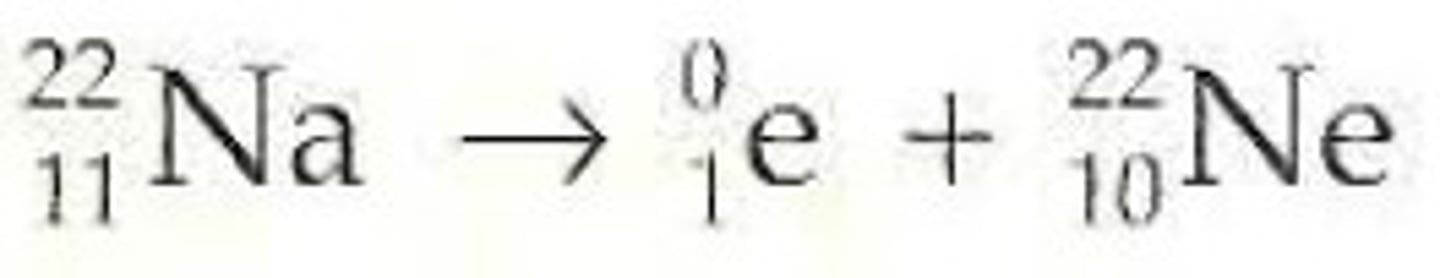
Electron Capture
radioactive decay that has an electron as a reactant and produces 1 gamma ray and a new element; new element's mass number stays the same and the atomic number decreases by 1
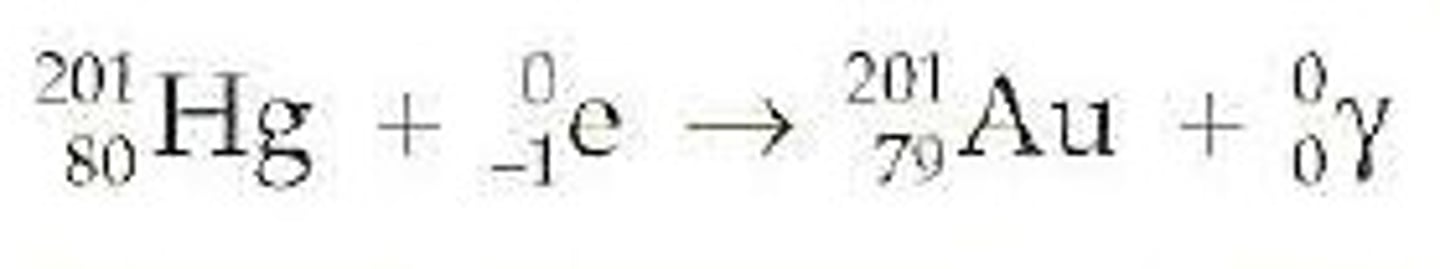
radioactive decay
spontaneous change from an unstable nuclei to a more stable one, releasing particles, electromagnetic waves or both.