C12.2 Acid-base titration
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
What are titrations used for?
A method to calculate the concentration of an acid or an alkali by determining how much acid is needed to neutralise an alkali (or vice versa)
Titrations can also be used to prepare soluble salts from the reaction between dilute acid and an alkali solution
List the apparatus needed for a titration
25 cm3 volumetric pipette
Pipette filler
50 cm3 burette
250 cm3 conical flask
Small funnel
0.1 mol / dm3 sodium hydroxide solution
Sulfuric acid of unknown concentration
A suitable indicator
Clamp stand, clamp & white tile
List the method for a titration
Use the pipette and pipette filler and place exactly 25 cm3 sodium hydroxide solution into the conical flask
Using the funnel, fill the burette with hydrochloric acid placing an empty beaker underneath the tap. Run a small portion of acid through the burette to remove any air bubbles
Record the starting point on the burette to the nearest 0.05 cm3
Place the conical flask on a white tile so the tip of the burette is inside the flask
Add a few drops of a suitable indicator to the solution in the conical flask
Perform a rough titration by taking the burette reading and running in the solution in 1 – 3 cm3 portions, while swirling the flask vigorously
Quickly close the tap when the end-point is reached
The endpoint is when one drop causes a sharp colour change
Record the volume of hydrochloric acid added, in a suitable results table as shown below
Make sure your eye is level with the meniscus
Repeat the titration with a fresh batch of sodium hydroxide
As the rough end-point volume is approached, add the solution from the burette one drop at a time until the indicator just changes colour
Record the volume to the nearest 0.05 cm3
Repeat until you achieve two concordant results (two results that are within 0.1 cm3
What are indicators used for?
Indicators are used to show the endpoint in a titration
List common acid-base indicators (neutral colour, colour in acid, colour in base)
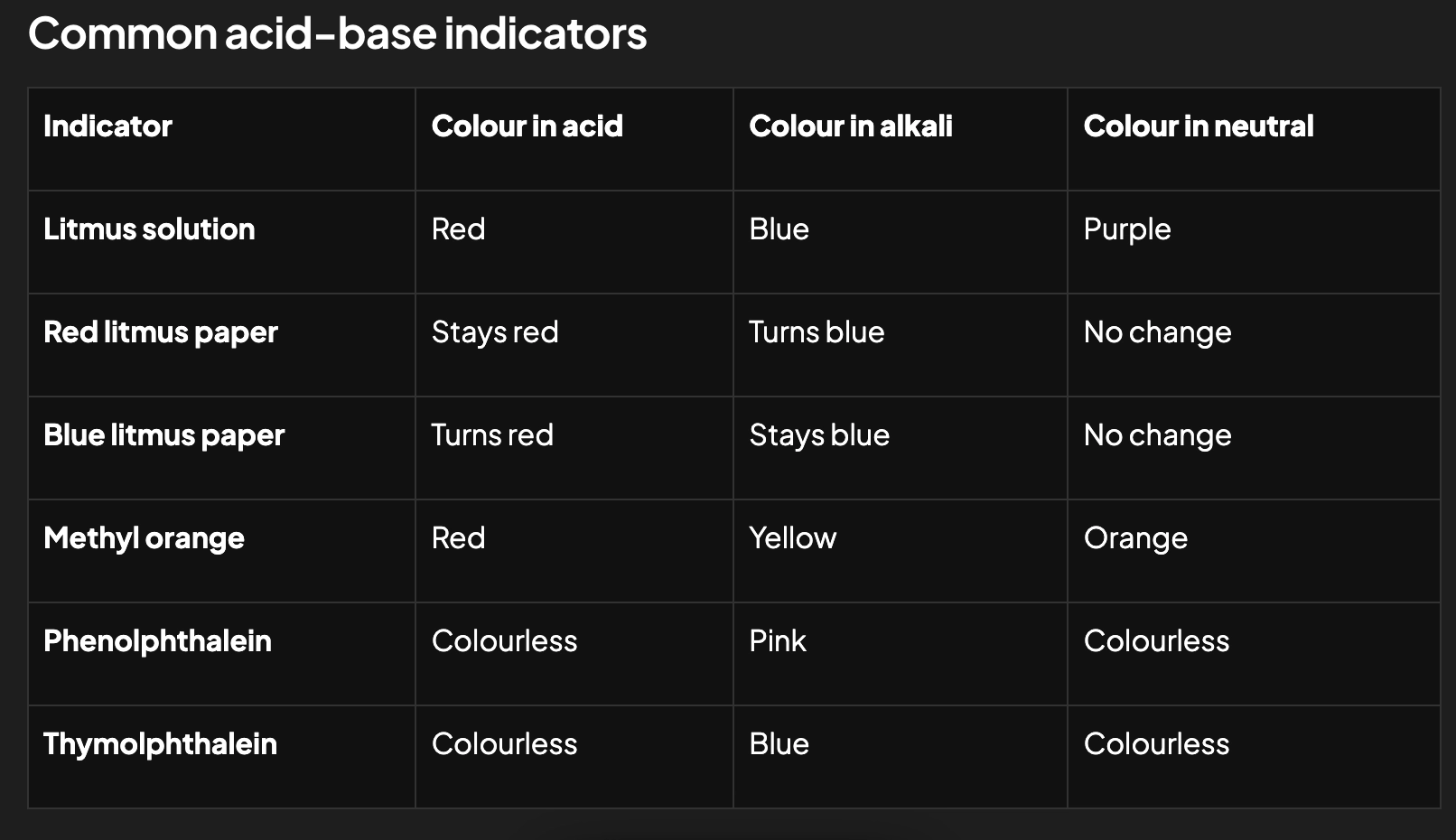
What indicator to use with a strong acid and a weak base?
Methyl orange (pH range 3.1-4.4)
What indicator to use with a strong acid and a strong base?
Bromythol blue (pH range 6-7.6) or phenolphthalein (pH range 8.2-10)
What indicator to use with a weak acid and a strong base?
Phenolphthalein (pH range 8.2-10)
What is a titrant?
A substance of known composition and concentration that is used to react with an analyte (in burette)
What is an analyte?
A substance that is being investigated (in the flask)
What is the equivalence point?
The point in a titration when there are equivalent amounts of titrant and analyte so the two substances can react completely with nothing left over
How to calculate pH of a strong acid given molarity?
-log[H+]
How to calculate pH of a strong base given molarity?
14 + -log[H+]
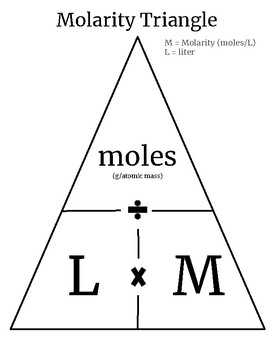
How to calculate moles given molarity and volume?
mL to L → divide by 1000
L to mL → multiply by 1000
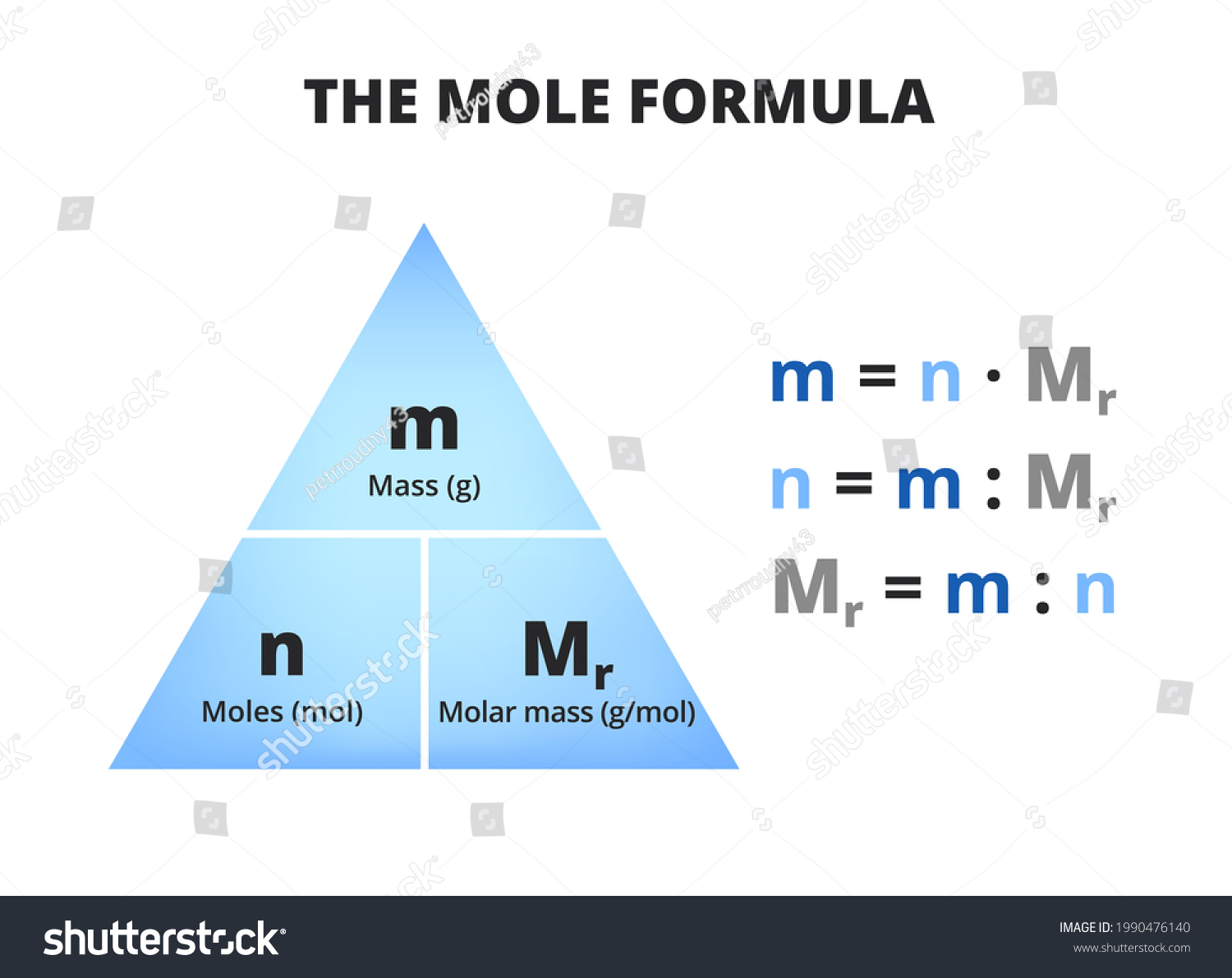
How to calculate moles given mass and relative mass?
Relative mass: find element on periodic table, add their atomic masses
If there is a subscript, multiply
If there is a coefficient, it is not affected