C8 : Chemistry Analysis
1/56
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
57 Terms
What does it mean if a substance is pure?
It is a single element or compound, not mixed with any other substance
What properties do pure substances have that can differentiate them from impure substances?
Pure substances have specific melting and boiling points whereas impure substances don’t.
What is a formulation?
A mixture that has been designed for a specific use
How are formulations made?
Formulations are made by mixing the components in carefully measured quantities to ensure that the product has the required properties
Give examples of formulations (7)
Fuels
Cleaning agents
Paints
Medicines
Alloys
Fertilisers
Foods
What is paper chromatography used to separate + identify?
Used to separate mixtures and can give information to help identify substances
What are the 2 types of phases in chromatography?
Stationary phase
Mobile phase
What is the stationary phase + give an example? (in general terms)
Where the molecules can’t move → a solid or a really thick liquid
What is the mobile phase + give an example (in general terms)?
Where the molecules can move → a liquid or a gas
What is the stationary phase in paper chromatography?
The chromatography paper
What is the mobile phase in paper chromatography?
The solvent
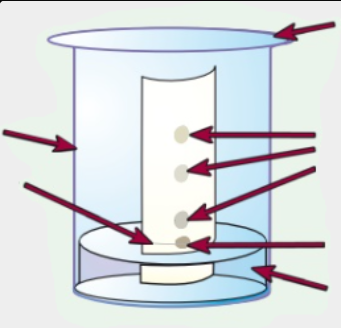
List the equipment needed for paper chromatography (6)
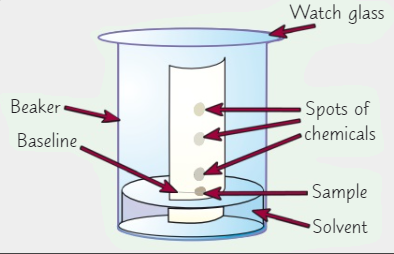
What 2 things could be used as a solvent?
Water
Ethanol
What phase do components normally separate in?
Stationary phase
What 2 things determine the amount of time substances spend in each phase?
How soluble the solvent is
How attracted they are to the paper
Which phase does a more soluble substance spend more time in + what attraction will it have to the paper + how far will it be carried up the paper?
Spends more time in the mobile phase
Less attracted to the paper
Will be carried up very high on the paper
Which phase does a less soluble substance spend more time in + what attraction will it have to the paper + how far will it be carried up the paper?
Spends less time in the mobile phase
More attracted to the paper
Won’t be carried up very high on the paper
How would you differentiate a pure substance from a impure substance using chromatography?
A pure substance would only have one dot in any solvent whereas an impure substance would have more than one
What is the result of a chromatography analysis called?
A chromatogram
What is the equation for the Rf value?
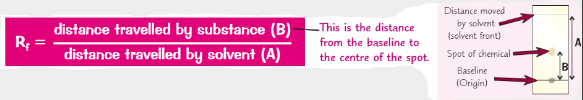
If you change the solvent what would happed to the Rf value?
The Rf value would change
Explain how paper chromatography can be used to separate and tell the difference between coloured substances (7)
Draw a horizontal pencil line 2 cm from the short edge of the chromatography paper + Mark pencil spots at equal intervals across the line but not too close to the end of the paper
Use a glass capillary tube to put a small spot of each colouring on the pencil spots. A small spot ensures that the colouring separates clearly. Label each spot in pencil
Pour about 1 cm3 of water into the beaker
Suspend the paper in the beaker so that bottom edge of the paper dips into the water + Make sure the pencil line is above the water surface
Wait for the water solvent to travel at least three quarters of the way up the paper. Remove the paper and draw another pencil line at on the dry part of the paper right next to the wet edge. This is the solvent front, the distance travelled by the solvent
Hang the paper up to dry thoroughly
Calculate the Rf values for each spot. Rf value is calculated by distance travelled by substance/ solvent front.
What is the test for chlorine?
The test for chlorine uses litmus paper. When damp litmus paper is put into chlorine gas the litmus paper is bleached and turns white
What is the test for oxygen?
The test for oxygen uses a glowing splint inserted into a test tube of the gas. The splint relights in oxygen
What is limewater known as?
Calcium hydroxide
What is the test for carbon dioxide?
The test for carbon dioxide uses an aqueous solution of calcium hydroxide (lime water). When carbon dioxide is shaken with or bubbled through limewater the limewater turns milky (cloudy)
What is the test for hydrogen?
The test for hydrogen uses a burning splint held at the open end of a test tube of the gas. Hydrogen burns rapidly with a pop sound
What can flame test be used to identify?
Metal ions (cations)
Explain how to do a flame test (3)
Dip a clean platinum wire loop into dilute HCL and then hold it in a blue flame (from a bunsen burner) until it burns without any colour
Then dip the loop into the sample you want to test and put it back in the flame
Record the colour produced
What colour flame does the compound lithium emit?
Crimson flame
What colour flame does the compound sodium emit?
Yellow flame
What colour flame does the compound potassium emit?
Lilac flame
What colour flame does the compound calcium emit?
Orange-red flame
What colour flame does the compound copper emit?
Green flame
What happens if a sample containing a mixture of ions is used for the flame test?
some flame colours can be masked
What is the test for carbonates?
Carbonates react with dilute acids to form carbon dioxide gas. Carbon dioxide can be identified with limewater
What is the test for sulfates?
Sulfate ions in solution produce a white precipitate with barium chloride solution in the presence of dilute hydrochloric acid
What is the test for halides?
Halide ions in solution produce precipitates with silver nitrate solution in the presence of dilute nitric acid
What does chloride give in the test for halides?
A white precipitate of silver chloride
What does bromide give in the test for halides?
A cream precipitate of silver bromide
What does iodide give in the test for halides?
A yellow precipitate of silver iodide
What is the test for metal hydroxides?
Adding a few drops of sodium hydroxide solution can be used to identify some metal hydroxides
What colour is the precipitate for calcium?
White
What is the ionic equation for the precipitate formation of calcium?
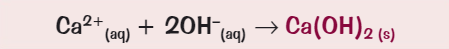
What colour is the precipitate for copper (II)?
Blue
What ionic equation for the precipitate formation of copper (II)?
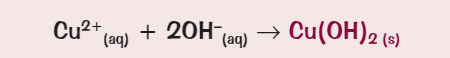
What colour is the precipitate for iron (II)?
Green
What ionic equation for the precipitate formation of iron (II)?

What colour is the precipitate for iron (III)?
Brown
What ionic equation for the precipitate formation of iron (III)?
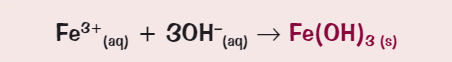
What colour is the precipitate for magnesium?
White
What ionic equation for the precipitate formation of magnesium?
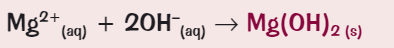
What colour is the precipitate for aluminium?
White at first, but then redissolves in excess NaOH to form a colourless solution
What ionic equation for the precipitate formation of aluminium?
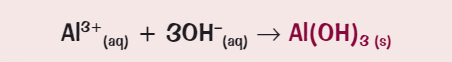
What happens during a flame emission spectroscopy? (4)
The ion is heated up
When the electrons in the ions cool down they transfer energy as light
The light passes through a spectroscope which detects different wavelengths of light to produce a line spectrum
Each ion produces a different pattern of wavelengths and has a different line spectrum

What does the intensity of the spectrum indicate?
The concentration of that ion in solution
What is the advantages of using machines such as instrumental methods such as flame emission spectroscopy? (3)
Very sensitive
Very fast
Very accurate