The shapes of molecules and ions
1/33
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
How does electrons in molecules exist?
In pairs in volumes of space called orbitals
What do pairs of electrons do?
Repel each other
What do electron pairs do to minimise repulsion?
Take up positions as far apart as possible from each other
What does the shape of a simple molecule depend on?
The number of pairs of electrons that surround the central atom
How do you work out the shape of any molecule?
Draw a dot-and-cross diagram to find the number of pairs of electrons
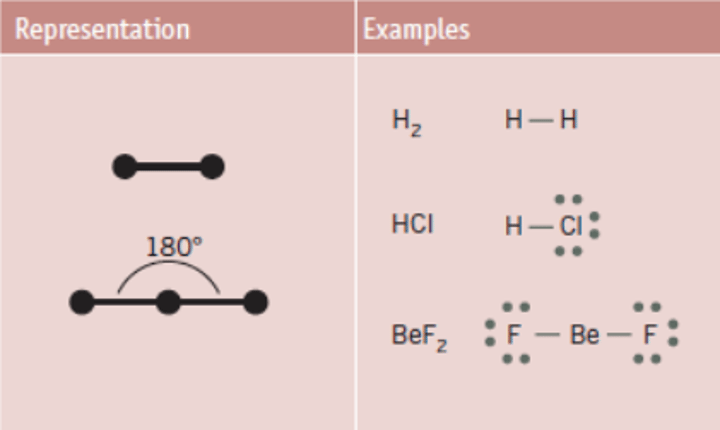
What is the bond angle of a linear molecule?
180°
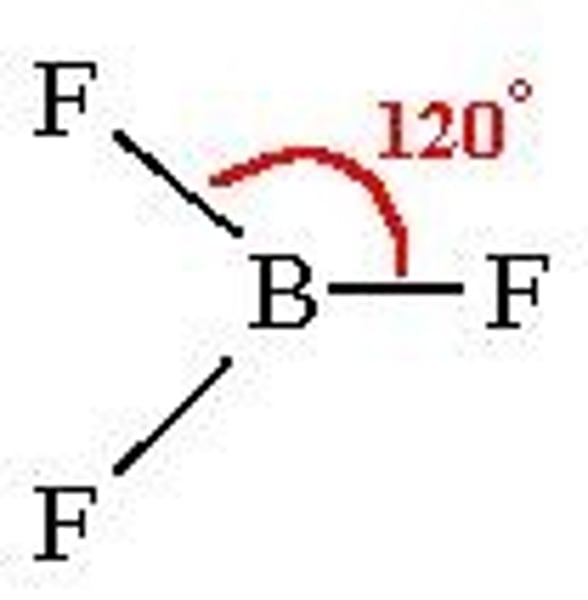
What is the bond angle of a trigonal planar molecule?
120°
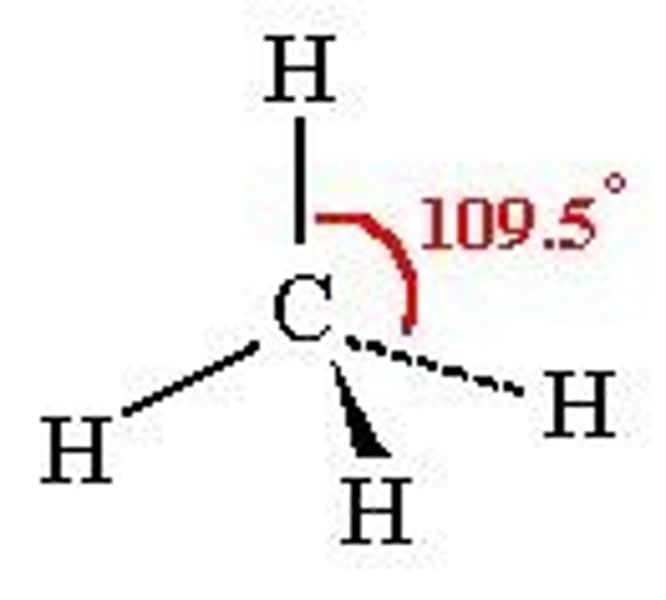
What is the bond angle of a tetrahedral molecule?
109.5°
Do the charges of ions affect the shape?
No
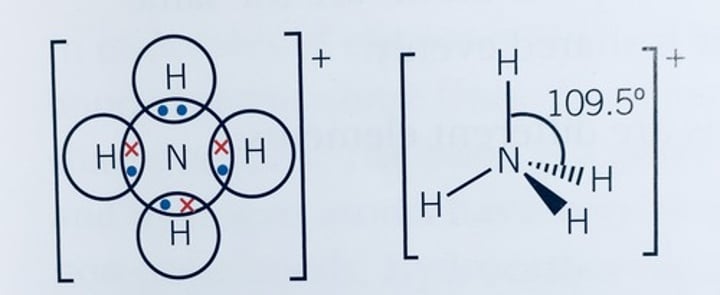
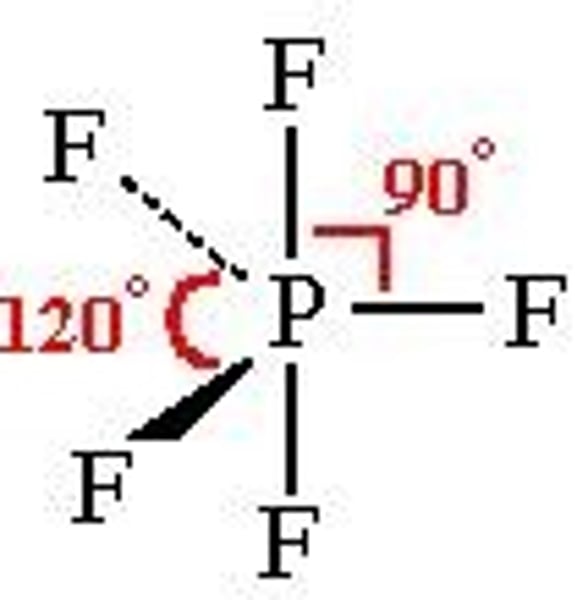
What are the bond angles of a trigonal bipyramid molecule?
90° and 120°
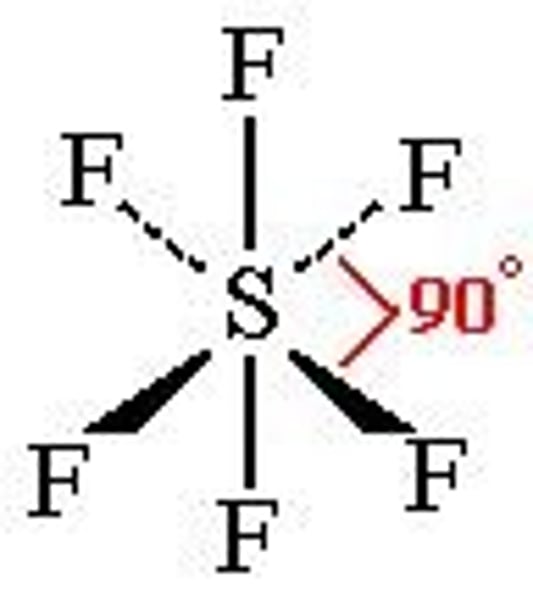
What is the bond angle of an octahedral molecule?
90°
Are lone pairs apart of the covalent bond?
No
How do lone pairs affect bond angles?
For each lone pair, the bond angle of 109.5° reduces by 2.5°
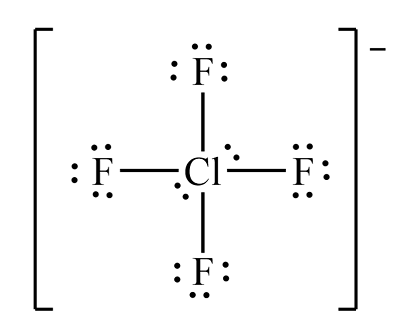
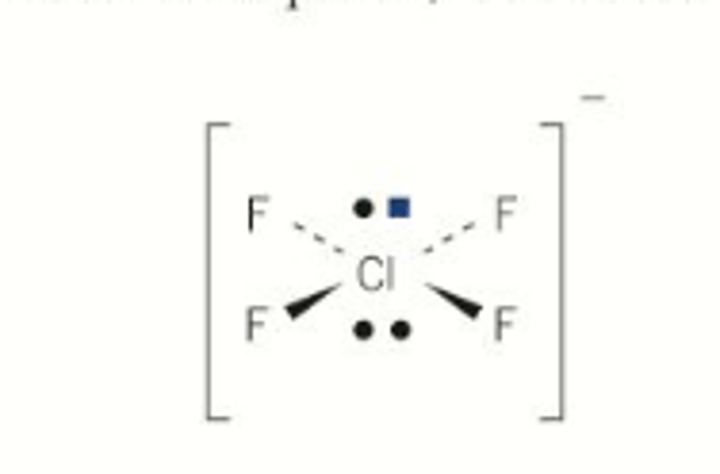
Chlorine tetrafluoride ion
Example of a square planar molecule
Repulsion between electron pairs

What is the shape of the molecule if there are:
2 electron pairs
2 bond pairs
0 lone pairs
Linear
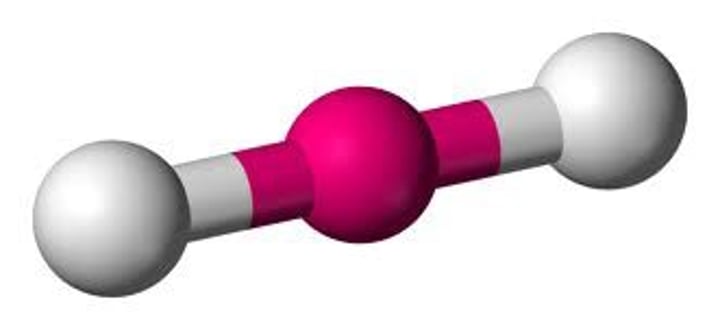
Bond angle between linear molecule
180°
What is the shape of the molecule if there are:
3 electron pairs
3 bond pairs
0 lone pairs
Trigonal planar
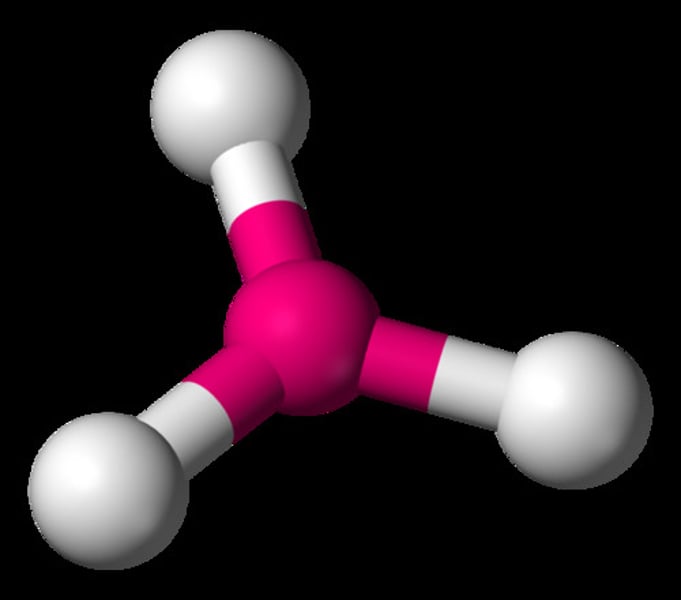
Bond angle between trigonal planar molecule
120°
What is the shape of the molecule if there are:
4 electron pairs
4 bond pairs
0 lone pairs
Tetrahedral
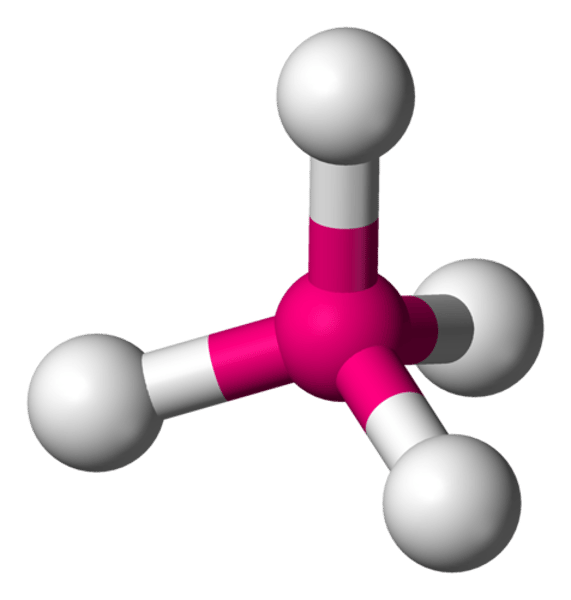
Bond angle between tetrahedral molecule
109.5°
What is the shape of the molecule if there are:
5 electron pairs
5 bond pairs
0 lone pairs
Trigonal bipydramid
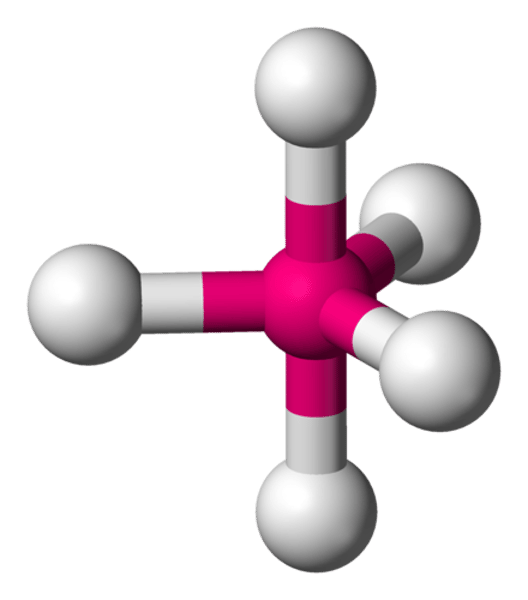
Bond angle between trigonal planar molecule
90° and 120°
What is the shape of the molecule if there are:
6 electron pairs
6 bond pairs
0 lone pairs
Octahedral
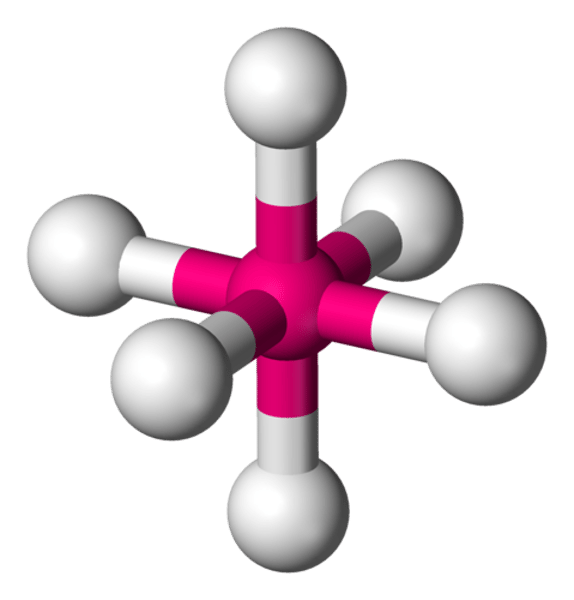
Bond angle between octahedral molecule
90°
What is the shape of a molecule if there are:
4 electron pairs
3 bond pairs
1 lone pair
Triangular pyramidal
Bond angle between a triangular pyramidal molecule
109.5 - 2.5 (1 lone pair) = 107°
Molecular shape of ammonia (NH₃)
Triangular pyramidal
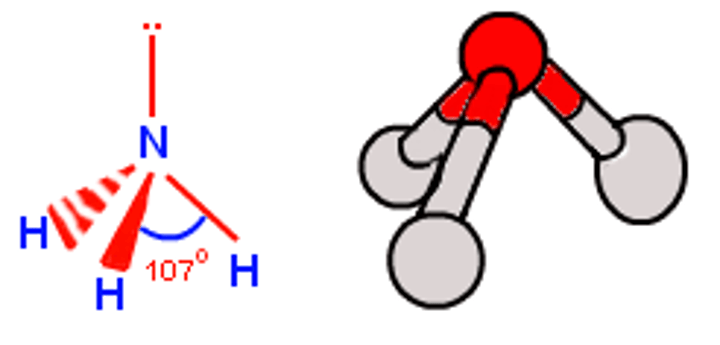
What is the shape of a molecule if there are:
4 electron pairs
2 bond pairs
2 lone pairs
Angular
Bond angle between an angular molecule
109.5 - (2 × 2.5) (2 lone pairs) = 104.5°
Molecular shape of water (H₂O)
Angular
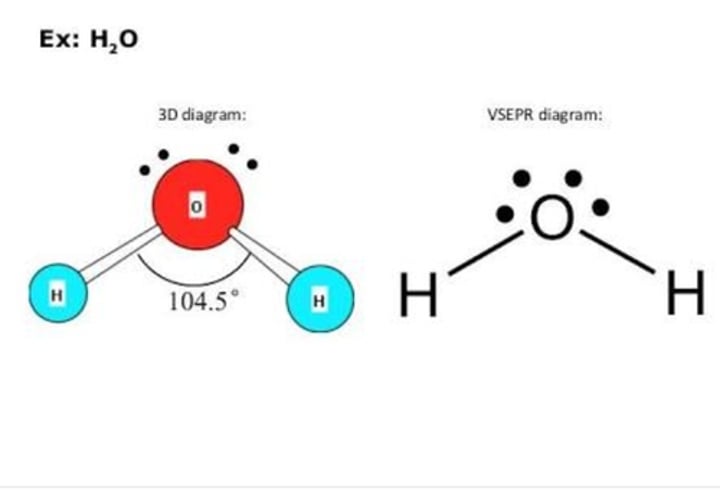
What is the shape of a molecule of there are:
6 electron pairs
4 bond pairs
2 lone pairs
Square planar
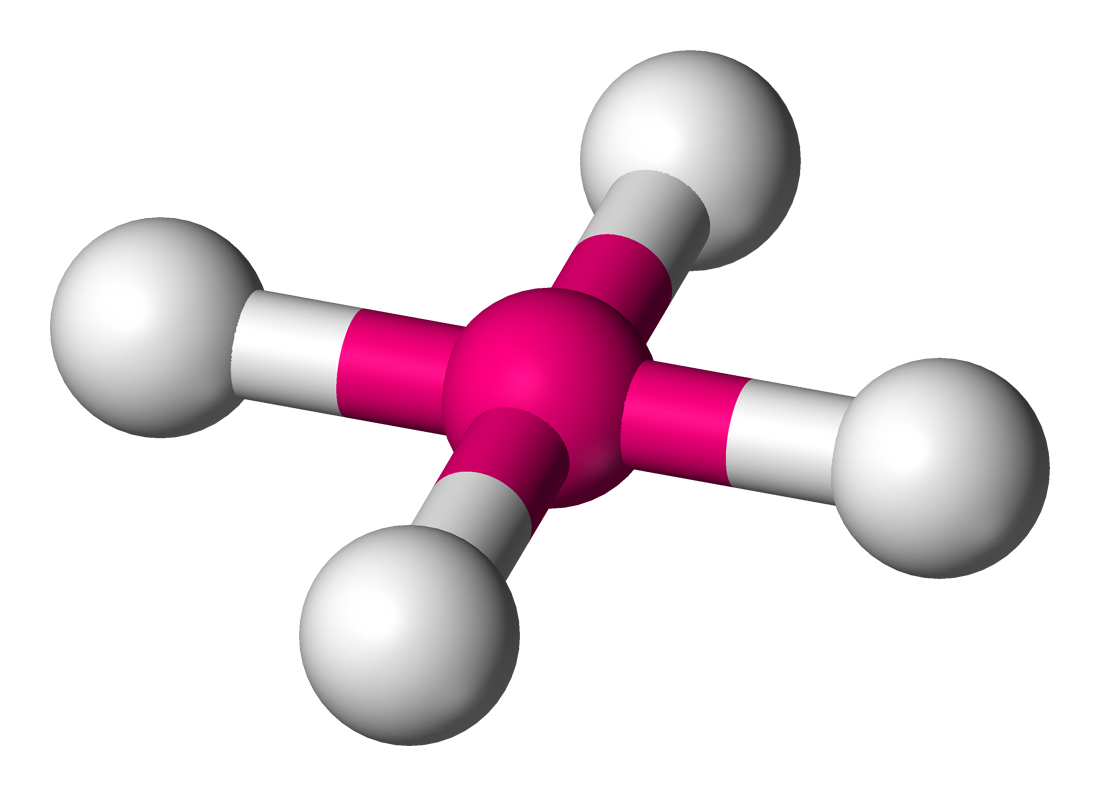
Bond angle between a square planar molecule
90
Molecular shape of chlorine tetrafluoride ion (ClF₄⁺)
Square planar