Reagents
1/60
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
61 Terms

Hydration of an Alkene (Alkene to alcohol)
Preparation of Ethers (SN2 Primary alcohol to diethyl ether)
Acetal deformation (Acetal to carbonyl)
Oxidation of Cyanohydrin (Cyanohydrin to hydroxyCarboxylic acid (racemic) (CN --> ROHCOOH))
Preparation of Carboxylic Acids (Nitrile to carboxylic acid)
Ester to Carboxylic acid and alcohol
Amide to carboxylic acid and ammonium
Nitrile to amide then, another dose of H3O+ and heat, then to carboxylic acid and ammonium
Tautomerization (Ketone to resonance stabilized cation to enol
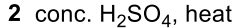
Elimination (Dehydration) - E1 tertiary alcohol to alkene, E2 alcohol to alkene
Elimination Reactions at Benzylic positions (Benzylic Alcohol to Benzylic position with an alkene attached to it)
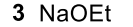
Elimination Reactions of Benzylic Halides (Benzylic Halide to Benzylic position with an alkene attached to it)
Enolate formation with a weak base (Ketone to Enolate)
Claisen Condensation (Ester to (enolate intermediate to stabilized enolate) to get the beta-ketoester)
Acetoacetic Ester Synthesis ((Ethyl Acetoacetate) Acetone to (to highly stabilized enolate intermediate to) (not isolated enol to) substituted derivative of acetone)
The Malonic Ester Synthesis (Diethyl malonate to (highly stabilized intermediate to monosubstituted diethyl malonate to monosubstituted deithyl malonic acid) to (enol not isolated to) substituted derivative of acetic acid)
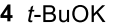

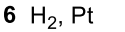

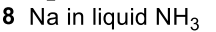


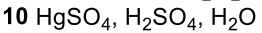


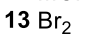


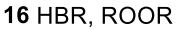
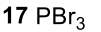
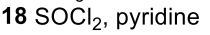
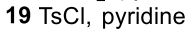
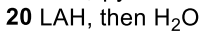
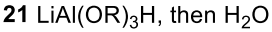
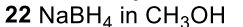
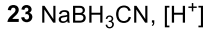
Reductive Amination (Ketone to Imine to Amine)
- Treat with Ammonia to get a primary amine
- Treat with a primary amine to get a secondary amine
- Treat with a secondary amine to get a tertiary amine
- Reductive amination can only get you to tertiary amines, so to get a quarternary, you need to alkylate (with an alkyl halide).

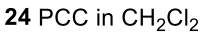
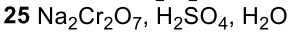
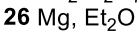

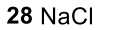
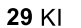
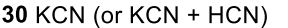
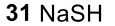

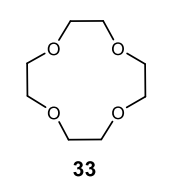
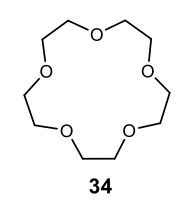
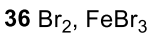
Benzene to resonance stabilized sigma complex/arenium ion to monobrominated benzene
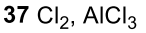
Benzene to resonance stabilized sigma complex/arenium ion to monochlorinated benzene

Benzene to nitrobenzene

REVERSIBLE Benzene to Benzene-sulfonic acid
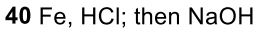
Nitrobenzene to Aniline
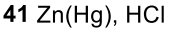
Reduction of Aryl Ketone (This is how to attach a primary cause if you do acylation and then this you can attach a primary alkyl group)

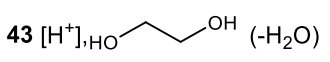

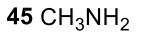
Primary amine
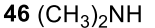
Secondary amine

Primary amine
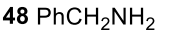
Primary aryl amine
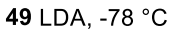
Ketone to less sub enolate
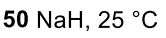
Ketone to more sub enolate

Primary aryl amine to aryl Diazonium Salt

Aryl Diazonium to benzylic Cl
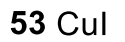
Aryl Diazonium to benzylic I

Aryl Diazonium to benzylic CN

Aryl Diazonium to Fluorobenzene
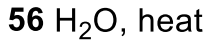
Aryl Diazonium to Phenol
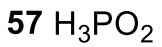
Use of hypophorphorus acid to hydrogenate (Aryl Diazonium to Benzene)
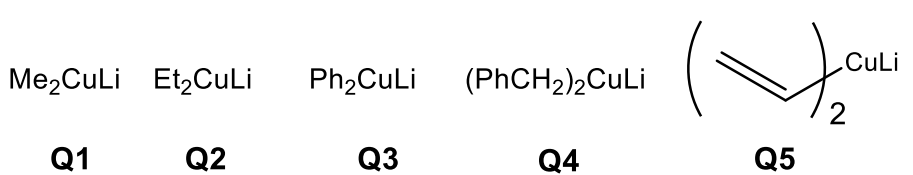
Grignard
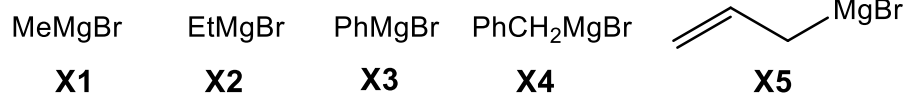
Gilman
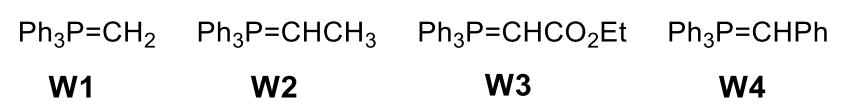
Wittig