Chemistry Atoms Unit 1.2
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
List the four types of radiation
Alpha (α) particles, Beta (β) particles, Positrons (β+),Gamma (γ) radiation
describe Alpha (α) particles
Two protons and two neutrons (Helium Nucleus)
Describe Beta (β) particles
High speed electrons
Describe Positrons (β+)
High speed anti-electrons. An anti-electron has the same mass as an electron but a different electrical charge: positive.
Describe Gamma (γ) radiation
High energy electromagnetic radiation.
Describe the Deflection of radioactive particles in an electric field
Alpha particles are weakly attracted to the negative plate of an electric field.
Beta particles move immediately towards the positive plate of the electric field.
Gamma rays are unaffected by an electric field.
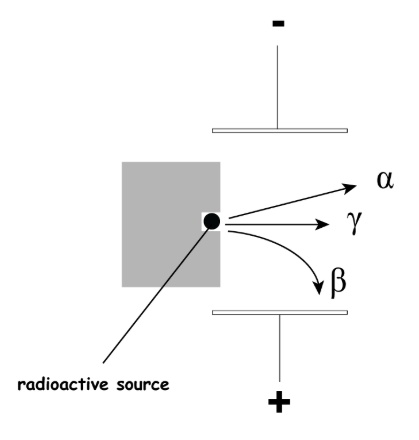
Give the order of the radioactive particles from the highest penetrating power to the lowest penetrating power.
Gamma (Only stopped by lead), Beta (stopped by aluminium), alpha (stopped by paper)
Give the order of the radioactive particles from the highest ionising power to the lowest Ionising power.
Alpha, beta, gamma
define ionising
Ionising is what radioactive particles do. it means to remove remove an electron as they collide with an atom.
describe electron capture
Electron capture occurs when a proton-rich nucleus absorbs an inner electron. This electron combines with a proton to form a neutron.
Give an equation for what it looks like when an alpha particle is emitted.
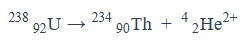
Give an equation for what it looks like when an beta particle is emitted.
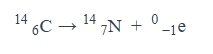
Give an equation for what it looks like when a positron particle is emitted.
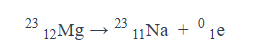
Give an equation for what it looks like electron capture occurs.
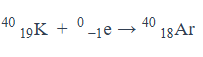
What is half life?
the time it takes for the mass of a radioactive substance to fall to half of its original value.
how does radiation effect living cells
When the DNA of a cell is damaged, it can become cancerous. A cancerous cell starts to divide uncontrollably, causing serious health problems such as tumours
What are the effects if alpha radiation is swallowed or inhaled
Its the most dangerous due to it being the most ionising. Therefore, it will cause the most damage to living cells.
What are the effects if beta and gamma radiation is swallowed or inhaled
They are not as dangerous due to them being less ionising. Therefore, they are more likely to pass through the human body without causing any damage to living cells.
What are the effects of alpha radiation outside the human body?
Its not as dangerous due to it being the least penetrating. Therefore, it cannot reach any living cells beneath the layer of dead cells on the surface of human skin.
What are the effects of beta and gamma radiation outside the human body?
They are more dangerous sources because they can penetrate the dead cells on the surface of the skin and damage the living cells beneath.