Pharmacovigilance and Yellow Card
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
What is the term used to describe a response to a medicinal production which is noxious and unintended?
ADRs
What are some issues relating to increased ADR prevalence?
Not enough info from clinical trials, increased polypharmacy and comorbidities
What does DoTS stand for?
Dose, Time and Susceptibility
What does Dose mean for ADRs?
ADR can occur at doses below therapeutic doses, at therapeutic range and at high doses
What does Time mean for ADRs?
Can be time-independent so requires monitoring throughout, time-immediate due to rapid administration, after first dose or early on, intermediate - risks after a few weeks or late e.g., long-term use, delayed e.g., can avoid, after withdrawal e.g., withdrawal syndromes
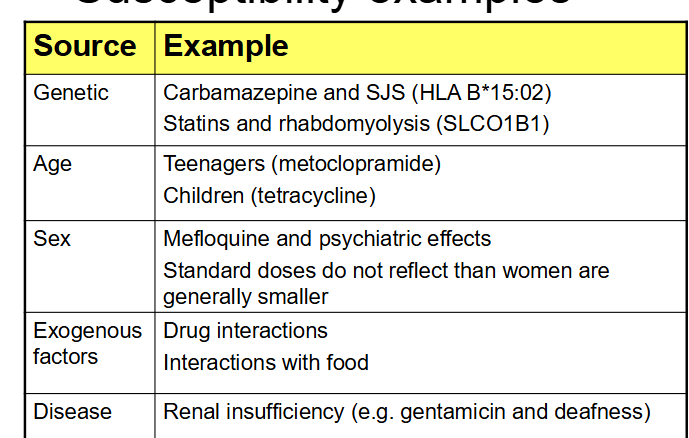
What does Susceptibility mean in DoTS for ADR reporting?
Genetic factors, Age, Sex, Physiology altered, Exogenous factors, Disease
What does a black triangle drug mean?
Intensive monitoring is needed - new active substance, biologic, new indication or conditional approval
Why is reporting necessary for new drugs available on the market?
Real world patients e.g., not ideal candidates like in clinical trials have varying ages, comorbidities, polypharmacy which can lead to unknown side effects, lots of harm could occur
Why should we report ADRs?
KEY for patient safety, allows continuous monitoring of old and new drugs, provides data on ADRs to new drugs, provides data in special patient groups
What is the scheme that collects reports of suspected problems or incidents e.g., ADRs, medical device adverse incidents, defective medicines, counterfeit/fake medicines and E-cigarettes?
Yellow card scheme
What are medical devices?
Regulated by MHRA and may include BP monitors, urine dipstick, CGM, healthcare apps
What should be reported on the Yellow card scheme?
Serious reactions to established drugs EVEN if well recognised - report suspicion, not necessarily proven causality
What is a serious ADR?
Any reaction which results in/prolongs hospitalisation - can be fatal, life-threatening, disability, cause congenital abnormalities and medically significant
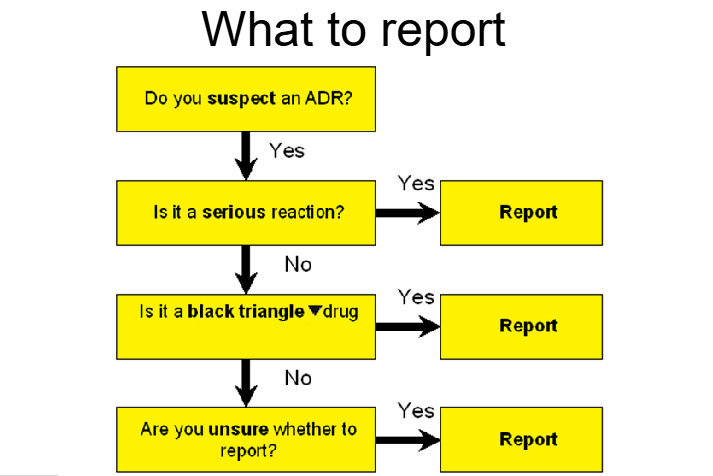
What is the diagram showing what to report for ADRs?
What is the Yellow card biobank?
Understands genetic basis of variable drug responses and spontaneous reports are made + new initiatives
What is the future for pharmacovigilance?
Safety connect reports where there is personalised information on the reports, more information sharing with MHRA, focus increase on patient experience, AI to analyse COVID reports, active surveillance implementation, pharmacogenomics