chem cooked
1/88
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
89 Terms
Debrogile wavelength
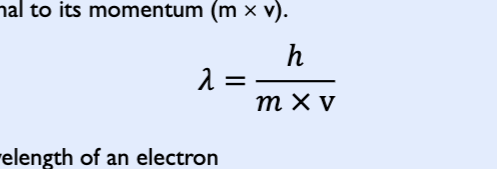
quantum numbers
principle quantum number, n
angular momentum quantum number, l
magnetic quantum number, m,l
spin quantum number, m,s
The uncertainty principle
The particle nature (location) and wave nature(motion) can be observed simutaneoulsly

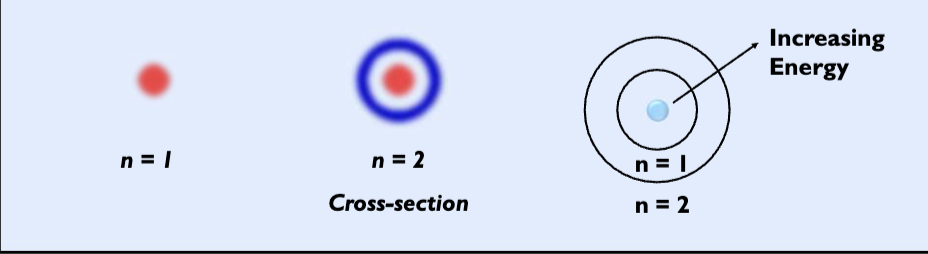
principle quantum number n
a positive integer 1,2,3…
determines the overall size and energy of an orbital
describes the distance of the orbital from the nucleus.
as n increases, energy increases, size of orbital increases, energy differences between orbitals decreases
The angular momentum quantum number, l
Determines the shape of the orbital
possible integers from 0 to (n-1)

Radial nodes
The value of the wavefunction 𝜓 is zero at this distance for this orbital.
Such a value of radius r is called a radial node.
The number of radial nodes in an orbital is n – ℓ – 1.
For a 1s orbital (n = 1, ℓ = 0): # nodes = 1 – 0 – 1 = 0
• For a 2s orbital (n = 2, ℓ = 0): # nodes = 2 – 0 – 1 = 1
• For a 3s orbital (n = 3, ℓ = 0): # nodes = 3 – 0 – 1 = 2
magnetic quantum number
If an orbital has an angular momentum l ≠ 0, then this orbital can point in different directions.
The magnetic quantum number, m ℓ, specifies the orientation of the orbital in space.
– One s-orbital for ℓ = 0
– Three p-orbitals for ℓ = 1
– Five d-orbitals for ℓ = 2
– Seven f-orbitals for ℓ = 3
Spin quantum number
The first three quantum numbers, n, ℓ, and mℓ, define the region of space where an electron is most likely to be located. An electron can only “spin” in one of two quantized states.
The spin quantum number, ms, describes two possible states. ms = +½(spin up) or –½ (spin down)
The Pauli exclusion principle
An electron in an atom is completely described by four quantum numbers: n, ℓ, mℓ, and ms.
No two electrons in the same atom can have exactly the same set of all four quantum numbers
no more than two electrons can occupy the same orbital.
• If two electrons are located in the same orbital, they must have opposite spins.
Orbital with quantum numbers
Each set of n, ℓ, and mℓ describes one and only one orbital.
Orbitals with the same value of n are in the same principal energy level.
• Orbitals with the same values of n and ℓ are said to be in the same
sublevel.
– One principal energy level includes multiple sublevels (ℓ = 0, 1, … n–1)
degeneracy
For H atoms, energies of all the orbitals with the same n are the same. Energy levels in H with the same principal quantum number, n, are called degenerate energy levels.
Energies of electron orbitals
In atoms with more than one electron, degeneracy is eliminated by electron–electron interactions.
• Orbitals in different subshells have different energies.
• Orbitals in the same subshell are degenerate and have the same energy.
Quantum numbers summary

Columb’s law
describes the attractions and repulsions between charged particles.
Nucleus (+) and electron (-)
The closer these two particles are together, the more negative (lower) the energy will be: r↓ E↓
− The higher the amount of charges, the more negative (lower) the energy will be: q1 and/or q2↑ → E
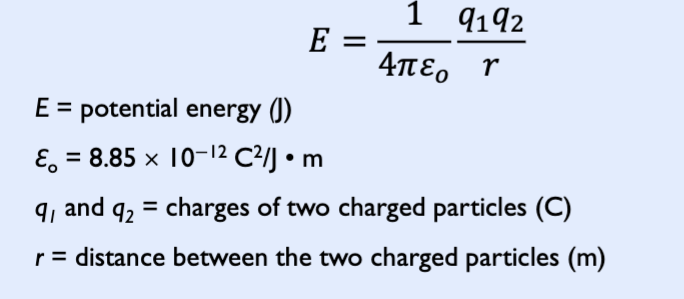
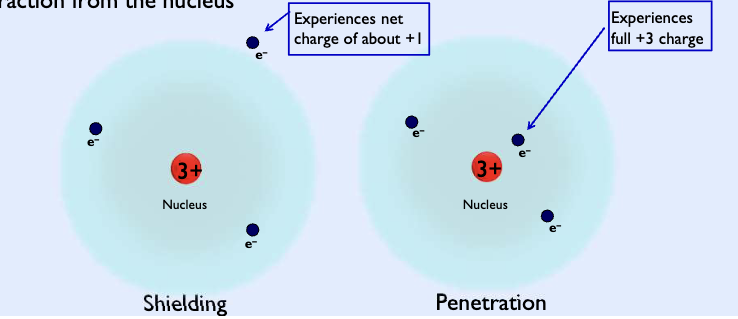
Shielding
In a multi electron atom, each electron experiences attraction to the nucleus and repulsion to other electrons.
Electrons closer to the nucleus (n =1.. n =2…) other electrons (n>=3) from experiencing the full charge of the nucleus and thus reduce the attraction to the nucleus

Penetration
As an outer electron undergoes penetration into the region occupied by the inner electrons, it experiences a greater nuclear charge and more attraction from the nucleus
Take home message,
The effects of shielding and penetration combine to produce effective nuclear charge (the effective nuclear charge “felt” by the outermost electron).
Shielding causes the electron to experience a lower positive charge from the nucleus, and therefore a higher (less negative) energy according to columb law
Penetration allows outer electron to experience a greater nucleus charge, and therefore a lower (more negative) energy according to columb law
Aufbau principle
an atom is built up by progressively adding electrons starting from the lowest energy level
Electrons filling order: 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s
Pauli exclusion principle
orbitals can hold no more than 2 electrons each
When 2 electrons occupy the same orbital, their spins are opposite
Hund’s rule
When filling orbitals, that have the same energy, electrons fill them singly first with parallel spins. once the orbitals of equal energy are half full, the electrons start to pair El
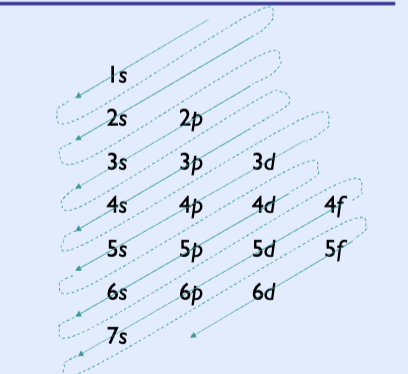
Electron configuration
Goes by arrows up: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s,
s: 2
p:6
d:10
f:14
Inner electron configuration
an abbreviation of the electron
configuration is to use the symbol of the previous noble gas in a square
brackets [ ] to represent all the inner electrons
Na = 11 electrons = 1s22s22p63s1 = [Ne]3s1
Outer electron configuration of first 18 elements
Elements in the same column, have similar properties, he number of electrons in the outermost principal energy level (highest n value) remains the same

Valence electrons
The electrons in the outermost principal energy level. key to chemical properties, instrumental in bonding; easy to lose or share C
Core electrons
Electrons in complete principal energy levels and those in complete d and f sub levels
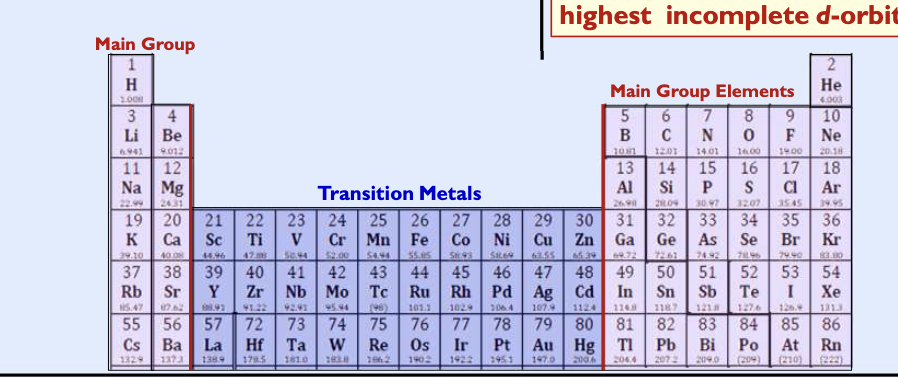
Main group eleemtns
elements that end in s block and p block, valence electrons are only in highest s and p orbitals
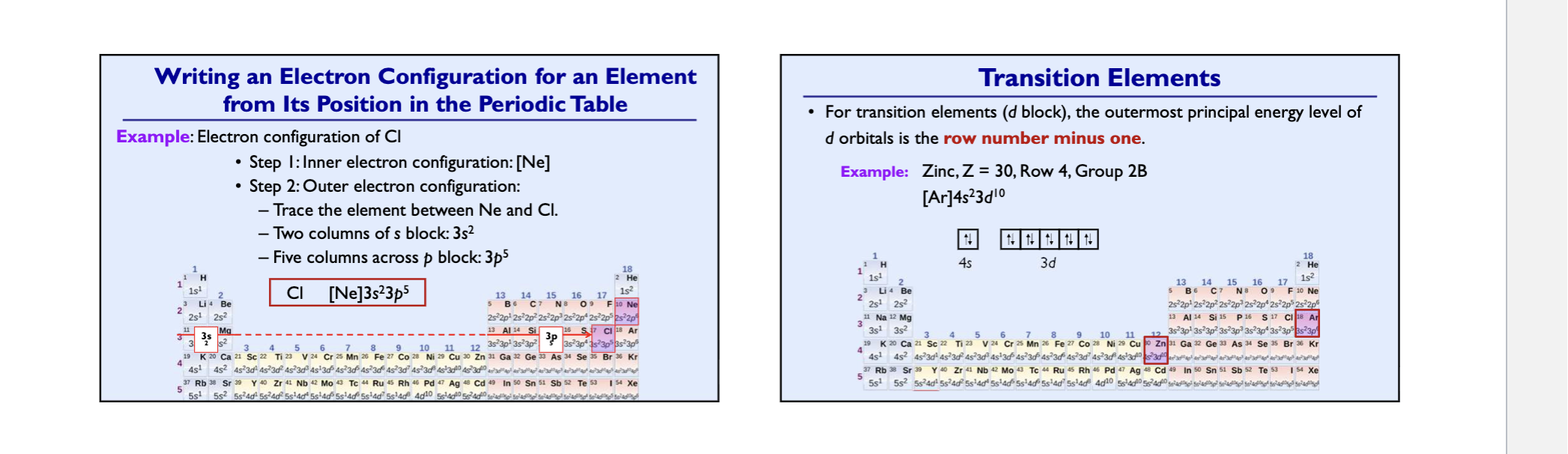
Transition metal elements
elements that end in d block
valence electrons are the two s- electrons + those electrons in the highest incomplete d-orbital
MAJOR
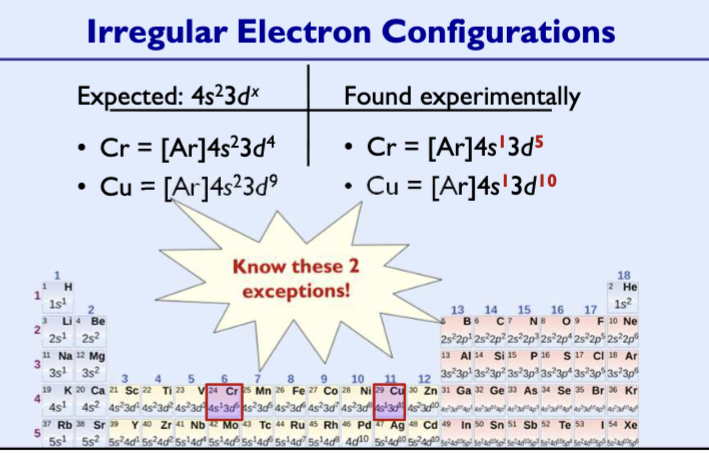
Irregular Electron Configurations
Some of the transition metals have the ns orbitals only partially filled or doesn’t fill at all before filling the (n−1)d orbitals.
– Due to extremely small energy difference between ns and (n-1)d orbitals
Electron configuration of ions
ions are formed when atoms gain or lose electrons
a cation forms when one or more electrons are removed from an atom
For main group elements, the electrons that were added last are the first electrons removed.
• For transition metals, the highest ns electrons are lost first, and then the (n – 1)d electrons are removed.
• An anion (negatively charged ion) forms when one or more electrons are added to a parent atom.
• The electrons are added in the order predicted by the Aufbau Aprinciple.
atomic radius main group
atomic radius increases down a column. Atomic radius decreases down the line
Valence electrons occupy large orbitals
More protons → higher effective nuclear charge → e–more attracted to the nucleus → smaller atomic radius
Atomic radius for transition metals
Increases down a column
roughly stays the same down the line because Moving from left to right, electrons are added to (n−1)d orbital, not the highest n orbital
– Effective nuclear charge on the ns electrons is approximately the same (Zeff = Z – S)
Nonbonding atomic radius (van der waals radius)
½ distance between two non bonding atoms that are in direct contact,
Bonding atomic radius (covalent radius)
nonmetals: ½ distance between two bonded atoms
metals: ½ distance between two adjacent atoms in a crystal of metal
atomic radius general
set of average bonding radii determined by measuring a large number of elements and compounds
ionic radii
ionic radius is used to measure describe size of an ion
A cation always has fewer electrons and the same number of protons as the parent atom.
A cation is always smaller than the atom from which it is derived.
• An anion always has more electrons and the same number of protons as
the parent atom.
An anion is always larger than the atom from which it is derived.
Trends in ionic radii - cations
cations are much smaller than their corresponding neutral atoms due to removal of outermost electrons
adii of cations increase down a column, and decrease across a period to the right

Trends in ionic radii - anions
anions are much larger than their neutral atoms due to the addition of electrons to the valence shell
Radii of anions increase down a column, and decrease across a period to the right

radii of isoelectronic ions
ions have the same number of electrons, but different number of protons.
– The ionic radius decreases with an increasing nuclear charge.
– For a given number of electrons, a greater nuclear charge results in a smaller atom or ion
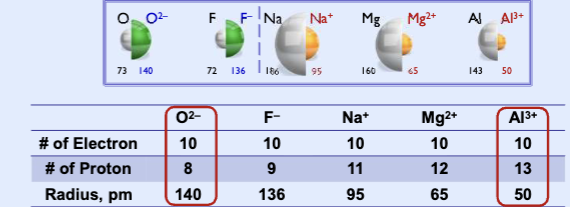
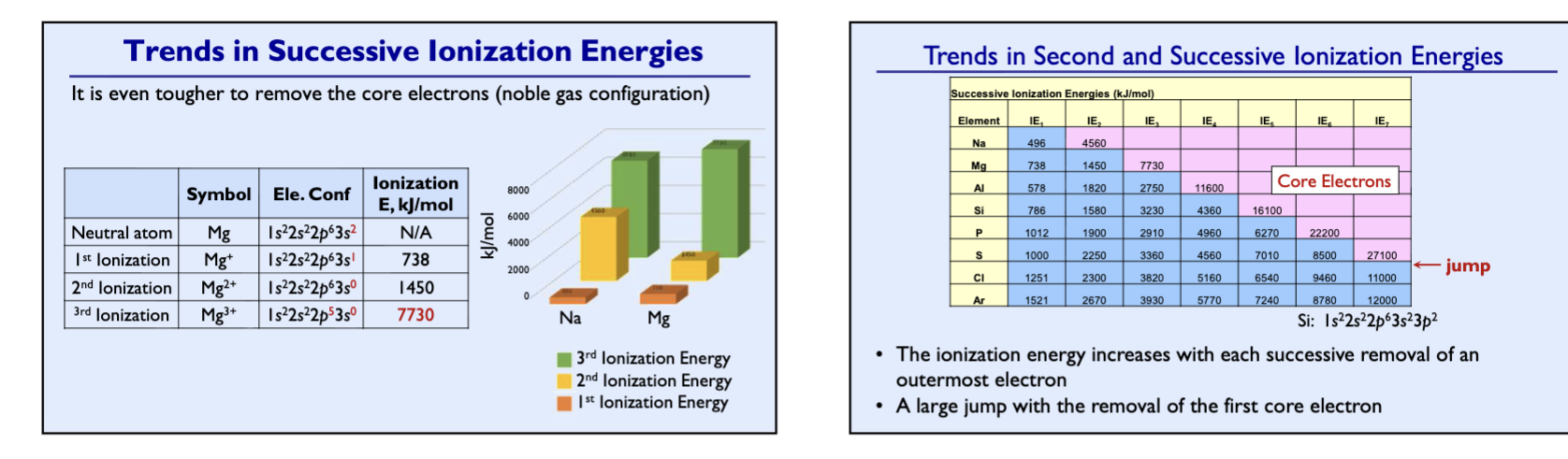
Ionization Energy (IE)
the energy required to remove an electron from the atom or ion in the gaseous state (kJ/mol). Positive values, endothermic process, require energy
First ionization energy (IE1): the energy required to remove the first electron
Second ionization energy (IE2): the energy required to remove the second electron
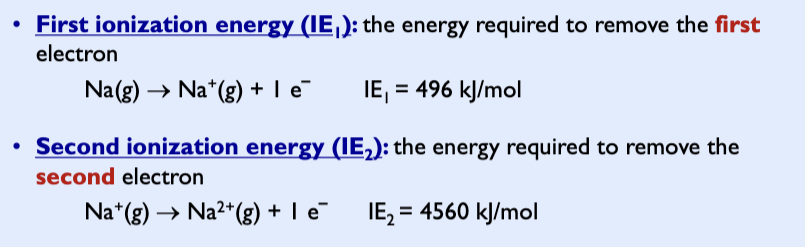
First ionization energy
Usually increases across a period to the right
higher effective nuclear charge, stronger interaction between nucleus and electron, more energy needed to remove electron
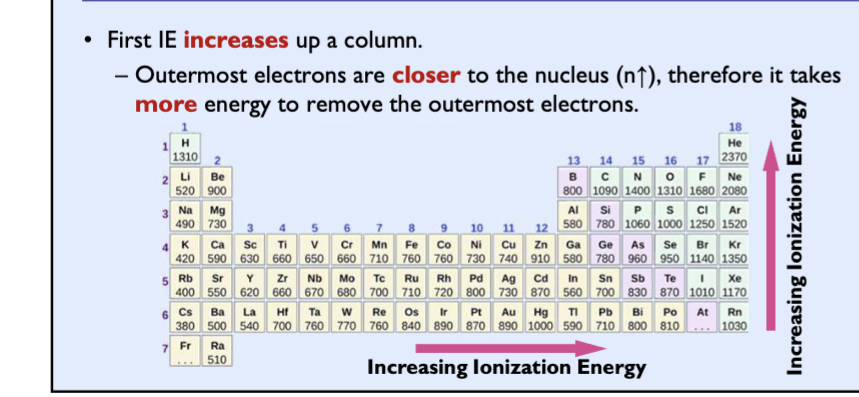
Exceptions for ionization
ionization energy, transition metals
very little difference in energy across transition metals.
trends in successive ionization energy 1

trends in successive ionization energy 2
Electron affinity
the energy given off when an atom or ion in the gaseous state gains an electron (reverse of ionization: removal of an electron)
Usually negative (exothermic); an atom or ion usually releases energy when it gains an electron.

Trends in electron affinity
In general gets more negative to the right.
Exception: Group 5A elements generally show more positive electron affinity than expected because of the repulsion between two paired electrons in p orbitals
Metallic character
conduct heat and electricity
malleable (permenantly shaped without breaking or cracking)
ductile (pulls into wires)
shiny and reflect light
loses electron in chemical reaction (low ionization energy, oxidized, form cations in solutions)
metallic character decreases across a period, increases down a column

Metals versus nonmetals
Metals tend to form cations by losing outermost electrons (low ionization
energy).
• Nonmetals tend to form anions by gaining electrons (high electron aff
trends
C
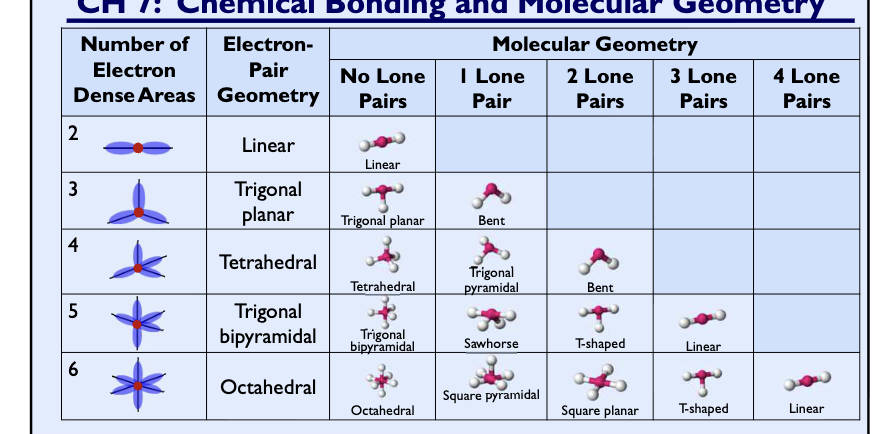
Chem geometry
Lewis model theory
simple model, dots, dashes, and chemical symbols
Valence bond theory 2
Treats electrons in a more quantum-mechanical manner but doesn’t look at electrons and molecules as a whole
Chemical bonds
They form because they lower the potential energy between the charged particles that composes atoms
when the potential energy of the bonded atoms is less than the potential energy of the separate atoms
Bonds ionic and covalent
metal and nonmetal (ionic) electrons transferred
nonmetal and nonmetal (covalent) electrons shared
Ionic bonds
formed between cations (metals) and anions (nonmetal) that are attracted by static forces'
Metals have relatively low ionization energies; they tend to form cations (+) by losing electrons.
Nonmetals have relatively high electron affinities (more negative); they tend to form anions (−) by gaining electrons.
Cations and anions attract each other resulting in lower overall potential law
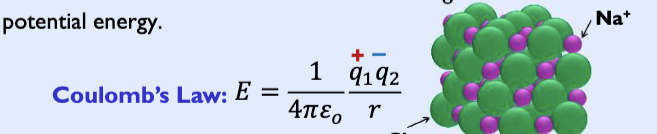
Ionic solids
crystalline structure
tend to be rigid and brittle
tends to have high melting and boiling points
ionic bonds are very strong
solids are poor conductors of electricity
most dissolve readily in water
once dissolved or melted, they’re good conductors of heat
covalent bonds
nonmetals when they share valence electrons
High ionization energy (difficult to remove electrons) high electron affinity (more negative, greater tendency to gain electrons)
shared electrons interact with nuclei of both atoms, lowering potential energy
Potential energy is the lowest when the electrons are between the nuclei based on Coulomb’s Law
– r decreases → 1/r increases → E is more negative (decreases)
covalent compounds
much lower boiling and melting points than ionic
many are liquid or gas at room temperature
usually softer than ionic solids
insoluble in water
poor conductor of electricity in any state
Electronegativity
the ability of an atom to attract bonding electrons to itself in a chemical bond
increases towards the right, decreases downward
not the same as electron affinity
dipole moment (u)
measure of bond polartiy
dipole means two electric poles (+ and -)
occurs any time there’s a separationof positive and negative charges (equal quantity)
proportional to the size of partial changes and proportional to distance between them
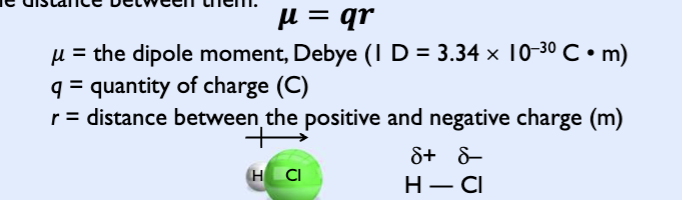
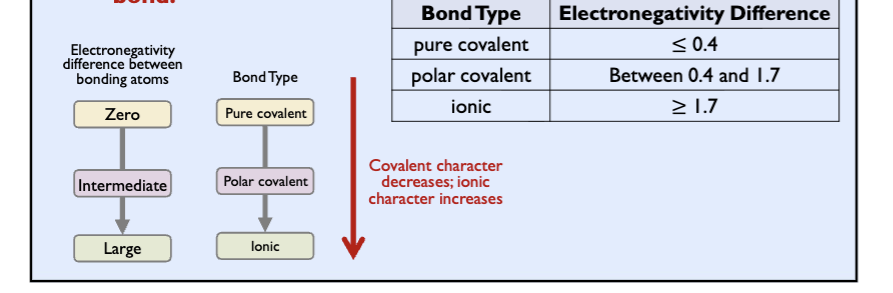
electronegativity difference and bond type
degree of polarity in a chemical bond depends on the electronegativity diff between two bonding atoms
greater the electronegativity difference, more polar the bond
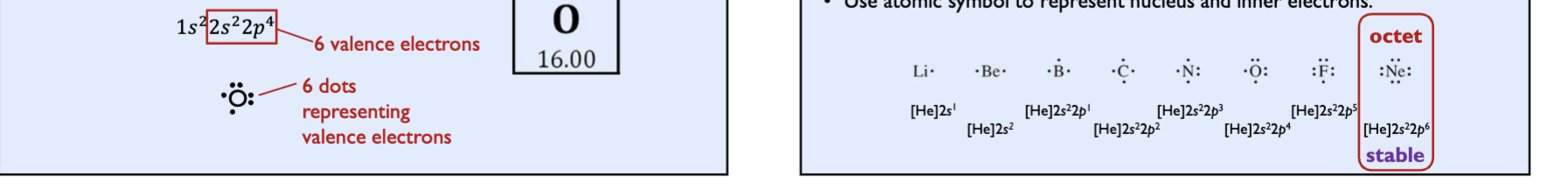
lewis bonding theory
theory emphasis valence electrons to explain bonding
valence electrons are dots.
Lewis theory covalent bonding
atoms form chemical bonds to attain stable electron configuration. stable means low potential energy
nonmetals achieve an octet of valence electrons by sharing their valence electrons
nonmetals have relatively high ionization energies (tend to form bonds by sharing electrons)

Octet rule
bonding atoms obtain stable electron configurations with eight electrons in the outermost shell
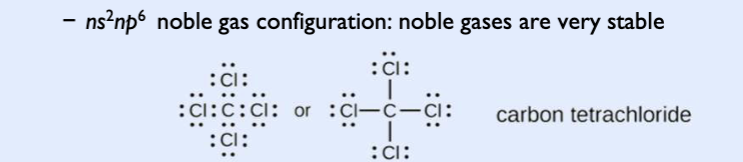
covalent bonding pairs
shared electrons would count toward each atom’s octet rule
Bonding pairs: electrons shared by atoms
lone pairs (nonbonding pairs): electrons not shared by atoms but belong to a particular atom

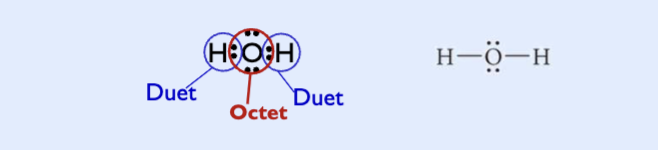
single covalent bonds
when two atoms share one pair of electrons
sharing electrons to get a duet for hydrogen and an octet for oxygen
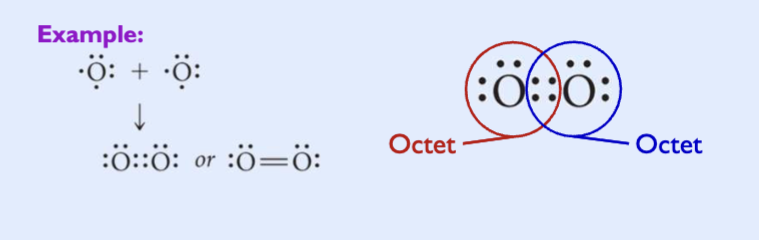
Double covalent bonds
two atoms share two pairs of electrons
two bonding pairs, four electrons
generally, double bonds are shorter and stronger than single bonds
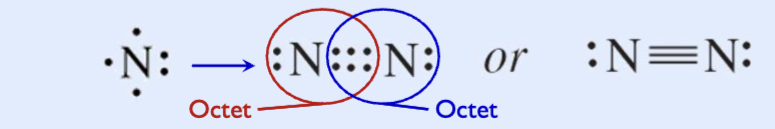
Triple covalent bonds
When two atoms share three pairs of electrons,
Three bonding pairs, six electrons
– Triple bonds are even shorter and stronger than double bonds
– The strong triple bond between nitrogen atoms makes N2 a relatively unreactive molecule
Lewis theory of covalent bonding
why particular combinations of atoms form molecules and other don’t
lewis theory of covalent bonding implies attractions between atoms are highly directional
shared electrons are most stable between the bonding atoms → each bond links one specific pair of atoms → The fundamental units of covalently bonded compounds are individual molecules (H2O, O2, HCl)
Lewis theory of covalent bonding 2
Predicts the melting and boiling points of molecular compounds are relatively low. (involves breaking the attractions between the molecules)
predicts that covalent bonded compounds should not conduct electricity (no charged particles)
predicts that the more electrons two atoms share, the stronger and shorter the bond should be
– Triple bonds are stronger and shorter than double bonds, and double
bonds are stronger and shorter than single bonds
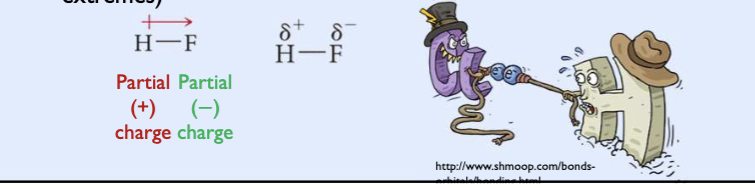
Polar covalent bonding
between unlike atoms results in unequal sharing of the electrons
one atom pulls the electrons to its side, resulting in larger electron density than the other
polar covalent bonding facts
positive sign indicates partial positive charge and lower electron density
arrow represents a partial negative charge and larger electron density
A polar covalent bond has a positive pole and a negative pole.
Lewis structure and octet rule
predicts that atoms will be most stable when they have their
octet of valence electrons.
• Some atoms commonly violate the octet rule
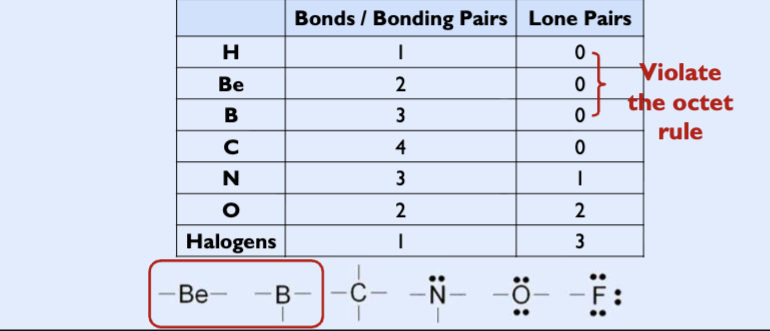
octet rule exceptions
Incomplete octets
– Molecules or ions with fewer than eight electrons around an atom
• Expanded octets
– Molecules or ions with more than eight electrons around an atom
• Odd electron species
– Molecules or ions with an odd number of electrons

Resonance
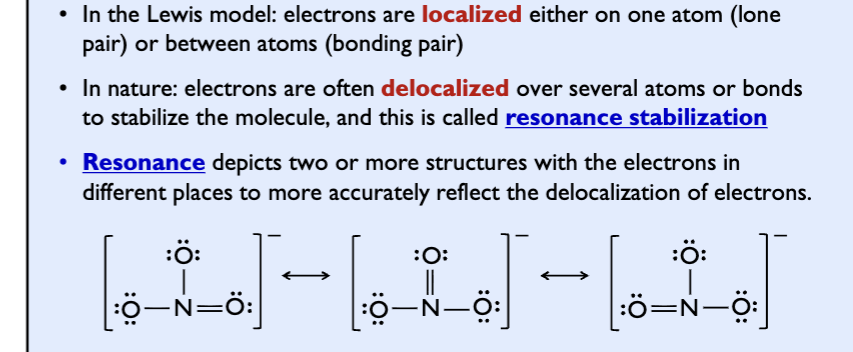
Resonance structutes
structures where more than one is valid
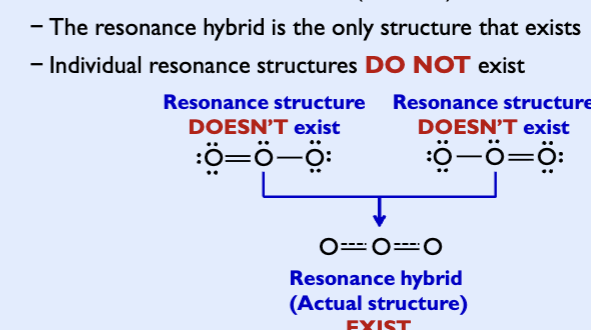
Resonance hybrid: actual structure of the molecule is intermediate between the two or more resonance strctures
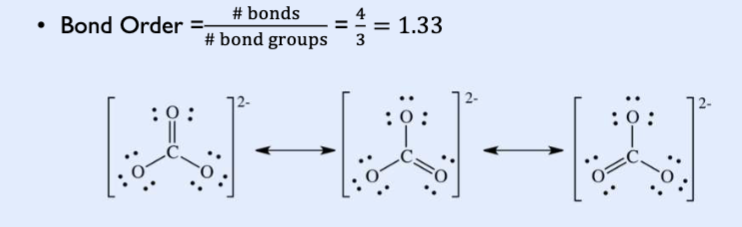
Bond order
draw the lewis structure
count the total number of bonds
count the number of bonds groups between individual atoms
# of bonds/ # bond groups.
Formal charge
The formal charge is the charge it would be if all bonding electrons were shared equally between the bonded atoms
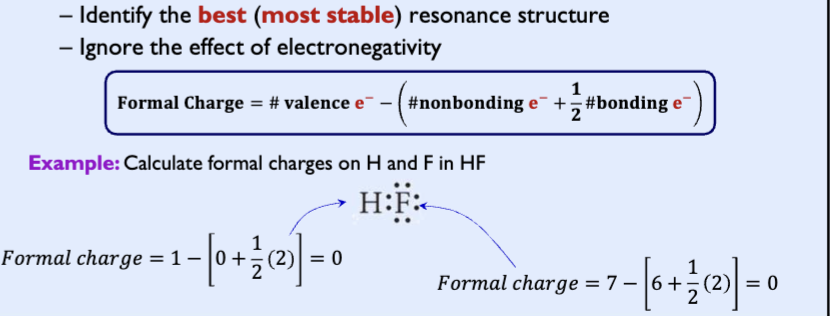
Rules of formal charge
sum of all formal charges must be 0
sum must be equal the charge of the ion
small (or zero) formal charges on individual atoms are better than larger ones
when formal charges can’t be avoided negative charge should be reside the most electronegative
Drawing renosance structures
draw the first lewis structure that maximizes octets
calculate formal charges
move electron pairs with atoms (-) towards the (+) formal charge
if (+) charge is in the 3rd row or beyond, bring in electron pairs to reduce formal charge, even if it gets expanded octet
if (+) formal charged atom is in the 2nd row only move in electrons if you move out electron pairs from multiple bond L
Lattice energy
energy associated with the formation of crystalline lattice of alternating cations and anions from gaseous ions

Trends in lattice energy
lattice energy is less negative (less exothermic, so it increases) as we move down the column
the more negative the more stable the ionic compound
Lattice energy becomes more negative (more exothermic) with increasing magnitude of ionic
charge (similar radii)
Greater charged ions as well as smaller sized ions lead to more negative lattice energy values and more stable ionic compounds.
• The effect of charge on the lattice energy is greater than the effect of
size
Bond energy
energy required to break 1 mole of the bond in the gas phase (alywas positive, specific to the two bonding atoms, higher bond energies → stronger bonds → more stable)Te
Trends in average bond energies #1
in general, the more electrons two atoms share, the stronger the covalent bond,

Trends in average bond energies #2
the shorter the covalent bond, the stronger the bond (stronger attraction → stronger bonds)

Golden rule
bond breaking is endothermic (heat change breaking) = +
bond forming is exothermic, (heat change forming) = -
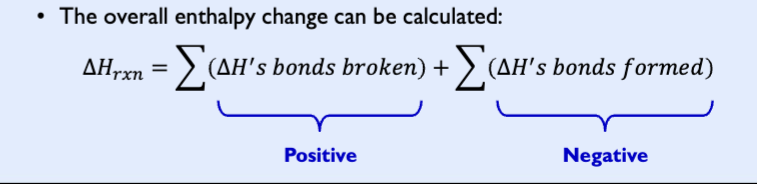
Endothermic reaction
(ΔHrxn < 0):
− when bonds formed give off more energy than bonds broken require
− weak bonds break and strong bonds form

endothermic
(ΔHrxn > 0)
− When bonds broken require more energy than bonds formed give off
− strong bonds break and weak bonds form
