Nucleophilic substitution (3.3.3.1) + elimination (3.3.3.2)
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Why do haloalkanes have a polar bond?
In a haloalkane molecule there is a C-X bond which is polar as the halogens (except iodine) are more electronegative than C. This means the shared pair of electrons between the C and X are closer to the X, thus the X is δ- and the C is δ+. This means haloalkanes have a permanent dipole if they are asymmetrical.
How does the C-X bond strength change down the group?
As the halogen atom gets larger down the group, the C-X bond length increases and bond strength weakens, making it easier to break
How can haloalkanes be classified?
They can either be primary, secondary or tertiary
Primary
The C in the C-X bond has 2 or 3 Hs bonded to it
Secondary
The C in the C-X bond has 1 H bonded to it
Tertiary
The C in the C-X bond has 0 Hs bonded to it
What is the mechanism for nucleophilic substitution of haloalkanes?
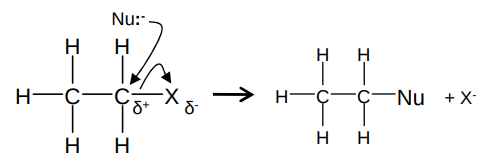
The nucleophile can be any negatively charged ion. e.g. hydroxide ions and cyanide ions
It happens to primary and secondary haloalkanes
What is the mechanism for the reaction between a halogenoalkane and NaOH and what are the conditions?
It forms an alcohol and sodium halide. e.g. for bromopropane and NaOH:
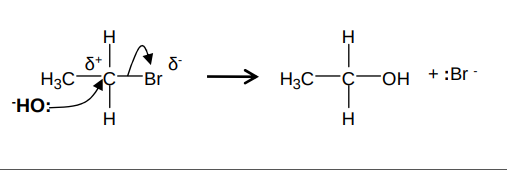
In a warm, aqueous solution. It occurs under reflux
What is the mechanism for the reaction between a halogenoalkane and H2O and what are the conditions?
It forms an alcohol and hydrogen halide. The mechanism is the same as for NaOH.
It is under a low temperature and a low concentration of -OH ions and is done under reflux
What is hydrolysis and how does the rate of hydrolysis in haloalkanes change down group 7?
Hydrolysis - a reaction which involves water or aqueous hydroxide ions to break bonds in a molecule
The rate of hydrolysis increases down group 7 as the average bond enthalpy of the C-X bond decreases down the group due to the halogens having more occupied shells so the shared pair is further away from the X nucleus so there is a weaker attraction between the bonding pair and X nucleus.
What is the mechanism for the reaction between a halogenoalkane and NaCN and what are the conditions?
It forms a nitrile and sodium halide e.g. for bromopropane and NaCN:
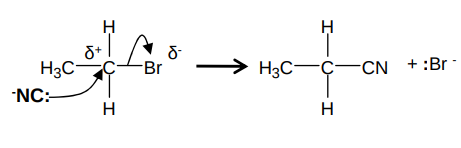
The NaCN is dissolved in ethanol/water mixture and it occurs under reflux
What is the mechanism for the reaction between a halogenoalkane and NH3 and what are the conditions?
It forms an amine and ammonium halide. e.g. for bromopropane and NH3:
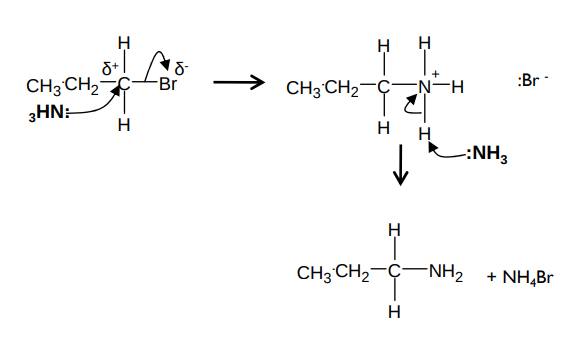
It needs excess ammonia dissolved in ethanol and heated under pressure
What is an elimination reaction?
The removal of a small molecule from the organic molecule
What is the mechanism for the reaction between a halogenoalkane and hydroxide ions and what are the conditions (elimination)?
It forms an alkene and water e.g. for bromopropane and -OH ions:
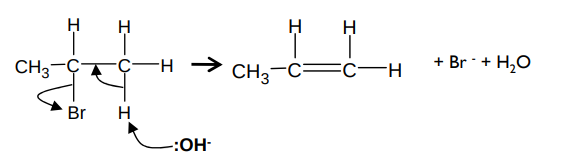
It occurs in ethanolic conditions and under high temperature
It tends to happen to secondary and tertiary haloalkanes
How can you tell the difference between an elimination and nucleophilic substitution by hydroxide ions?
Reagents/conditions | Elim | Sub |
Classification | Secondary/tertiary | Primary/secondary |
Base | High conc of OH- | Low conc of OH- |
Solvent | Ethanol | Water |
Temperature | High | Low |